Clear Sky Science · en
Maximizing photon utilization in spectroscopic single-molecule localization microscopy using symmetrically dispersed dual-wedge prisms
Sharper Views of the Tiny World
Many of the most important players in biology—individual molecules inside our cells—are far too small to see with ordinary microscopes. Over the past decade, new “super‑resolution” methods have changed that, but they often force scientists to trade image sharpness for color information or long, complicated experiments. This paper introduces a clever optical add‑on that helps researchers see many kinds of molecules at once, in 3D, with better detail and less fuss.
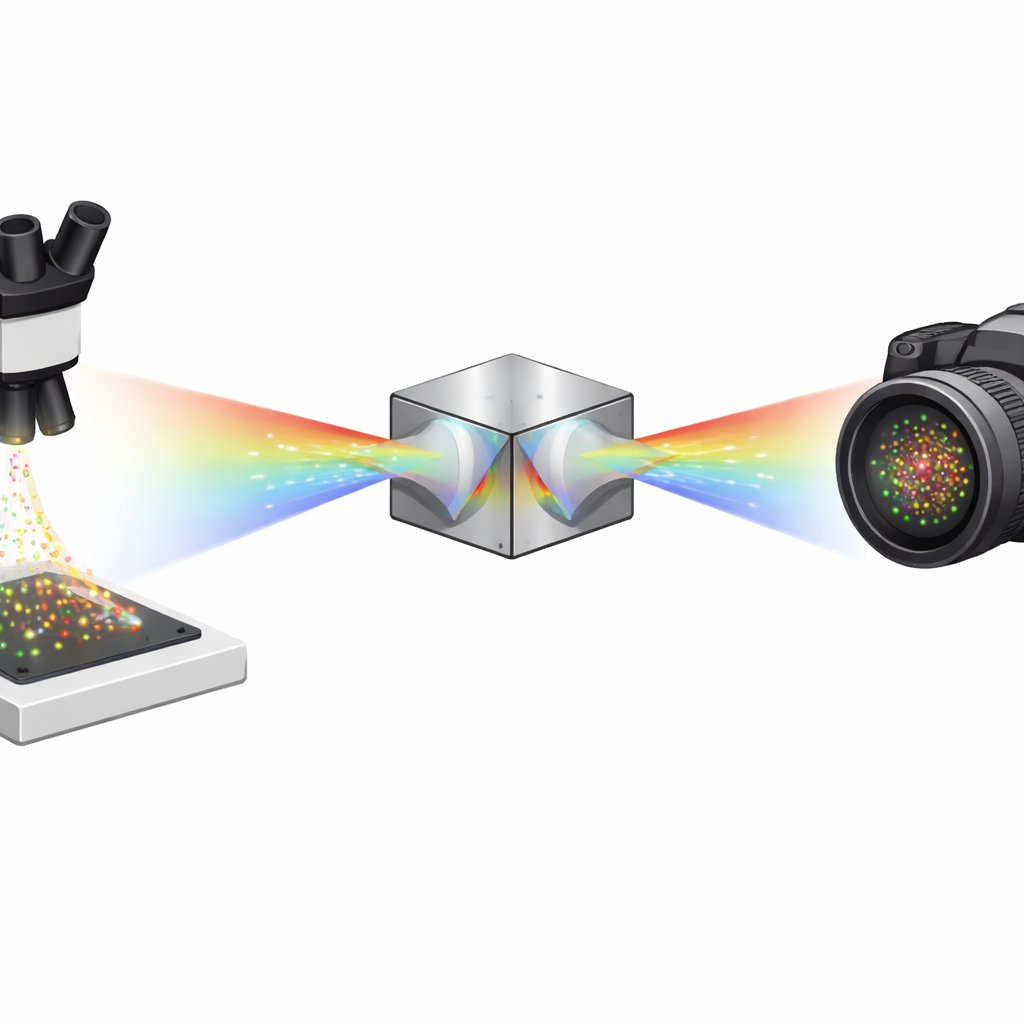
Seeing Single Molecules One by One
Super‑resolution methods like STORM and PALM work by making only a few fluorescent molecules blink at a time, then pinpointing each blink with high precision and combining thousands of such frames into a detailed image. Spectroscopic single‑molecule localization microscopy (sSMLM) goes one step further: it not only finds where each molecule is, but also measures its color spectrum. That extra spectral information is powerful, because it lets scientists use multiple dyes whose colors overlap and still tell them apart. The catch is that traditional sSMLM typically has to split precious photons between a position image and a spectrum image, which blurs the final picture and makes dim molecules hard to detect.
Using Every Photon Twice
The authors solve this problem with a compact optical module based on two identical dual‑wedge prisms and a beam splitter. Instead of sending photons into a “position” arm and a separate “color” arm, their symmetrically dispersed dual‑wedge prism (SDDWP) design creates two mirror‑image, spectrally spread copies of every blinking molecule on the same camera. Because these two images are perfectly symmetric, a simple computation can recover both the molecule’s true position (from the midpoint between the two spots) and its spectrum (from how far apart the spots lie). In effect, all collected photons contribute to both spatial and spectral information, dramatically improving how precisely the system can locate and identify each molecule.
Sharper, Clearer Colors in Three Dimensions
Using analytical models and carefully calibrated test samples, the team shows that SDDWP improves lateral (in‑plane) precision by about 27% and spectral precision by about 48% compared with their previous prism‑based system. They then extend the design to three‑dimensional imaging using a “biplane” approach, where the two spectral images are slightly out of focus in opposite directions. By analyzing how the size of each spot changes between the two planes, the system can determine how far above or below the focal plane a molecule sits, achieving axial precision on the order of 18 nanometers within a useful depth range of roughly half a micrometer. Despite the added complexity of 3D imaging, the new design maintains near‑2D levels of spectral sharpness, allowing very fine discrimination between dyes whose color spectra strongly overlap.
Color‑Separating Cell Structures and Moving Particles
To demonstrate what this means in practice, the researchers imaged fixed HeLa cells in 3D using a single red laser and three far‑red dyes that normally have overlapping spectra. They labeled peroxisomes, microtubules, and mitochondria, and showed that the system could reliably separate these structures based on their subtle spectral differences while keeping spatial detail high across depth. They also used spectrally distinct quantum dots as tiny tags to track many particles moving at once in a viscous solution. By treating each particle’s spectrum as a unique “fingerprint,” the SDDWP setup could correctly follow hundreds of densely packed trajectories that would otherwise become hopelessly tangled when paths crossed, reducing tracking errors to only a few percent even at particle densities approaching theoretical limits.
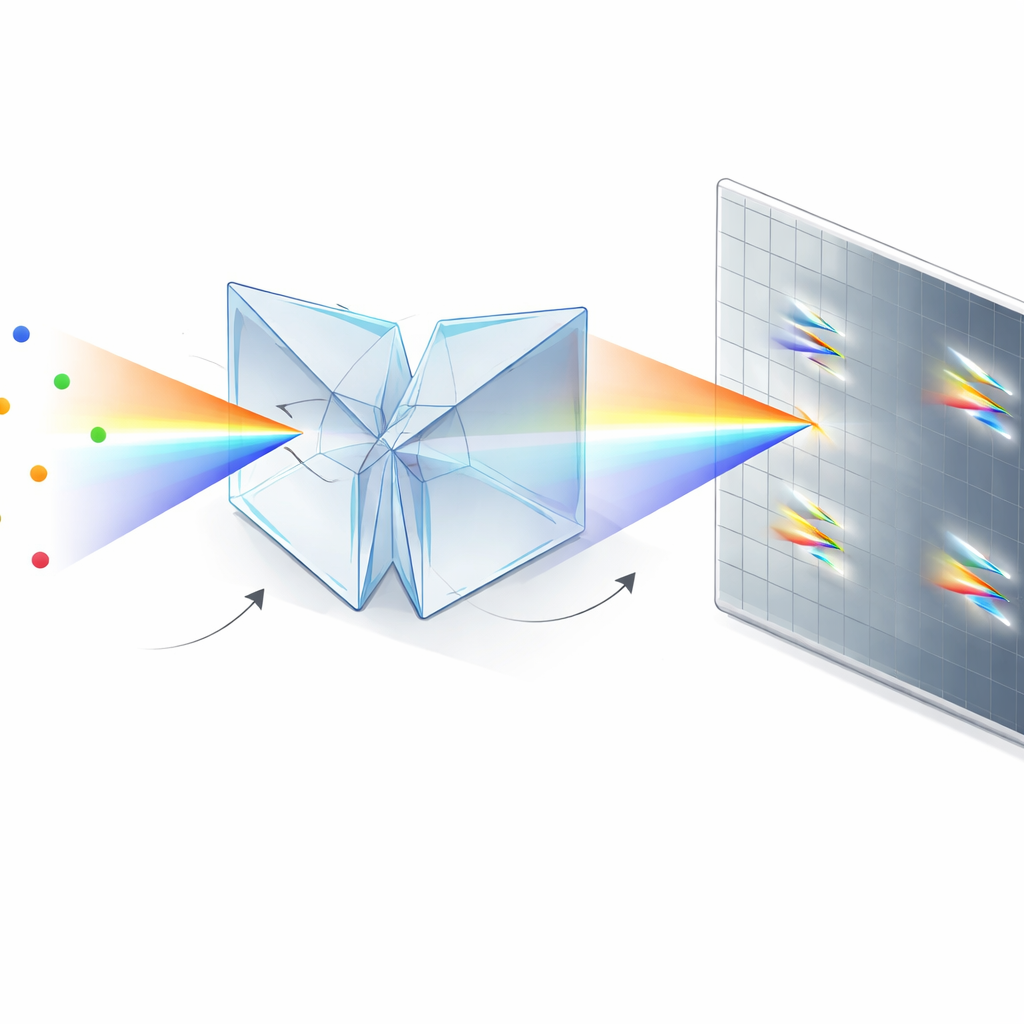
From Complex Optics to Simple Add‑On
Beyond performance, a key advantage of this approach is practicality. The SDDWP unit is a small, mostly monolithic assembly that bolts onto the side port of a standard inverted fluorescence microscope and requires only modest alignment. Its prism‑based design wastes far fewer photons than diffraction gratings, and it is mechanically stable enough to stay calibrated over long periods with only routine checks. This makes it a realistic upgrade path for many existing single‑molecule labs.
What This Means for Future Microscopy
By rethinking how light is split and recombined, this work shows that it is possible to get both sharper positions and clearer color information from the same limited pool of photons. In everyday terms, it lets scientists tell apart more kinds of molecules in crowded, three‑dimensional environments, and track many labeled particles at once, without sacrificing image quality. As the technique is adopted and adapted—potentially even for live‑cell imaging with gentle red light—it could become a versatile tool for exploring how complex molecular assemblies and organelles are organized and how they move inside living cells, all at nanometer scales.
Citation: Yeo, WH., Brenner, B., Shi, M. et al. Maximizing photon utilization in spectroscopic single-molecule localization microscopy using symmetrically dispersed dual-wedge prisms. npj Imaging 4, 20 (2026). https://doi.org/10.1038/s44303-026-00152-z
Keywords: single-molecule microscopy, super-resolution imaging, spectral imaging, 3D cell imaging, single-particle tracking