Clear Sky Science · en
A single-step radiolabeling strategy for PET, SPECT, and therapeutic radionuclides using nanoparticles as a universal chelator
Why tiny radioactive particles matter
Modern cancer care increasingly relies on seeing and treating tumors with radioactive drugs that light them up for scans and, in some cases, burn them from within. Yet building each new radioactive medicine is slow and fussy, because most designs must be custom‑built around a specific radioactive metal. This study explores a very different approach: using a single type of iron‑based nanoparticle as a “universal socket” that can hold many different medical isotopes for imaging and therapy, potentially speeding up how such drugs are created and fine‑tuned.
From many keys to one lock
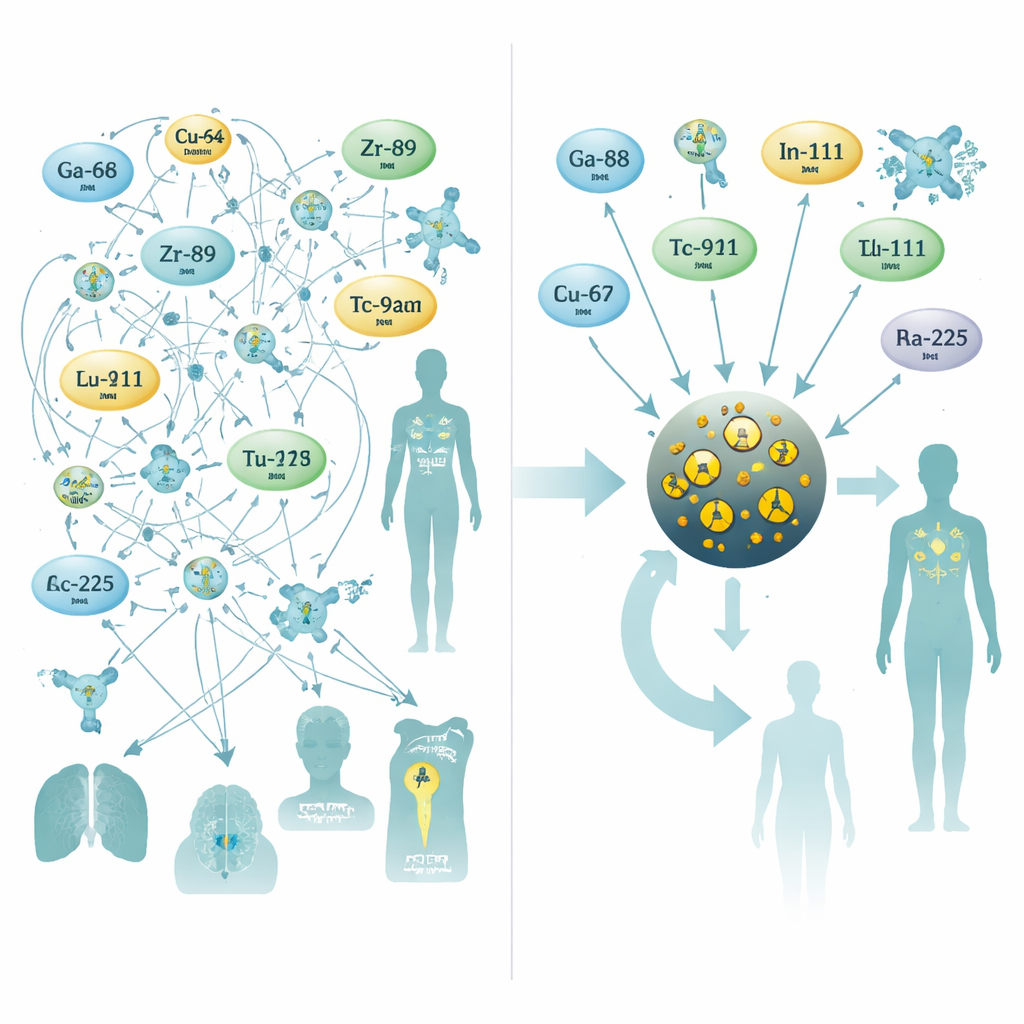
Today’s radioactive medicines usually rely on chemical claws called chelators that grip metal atoms and attach them to antibodies or other targeting molecules. Each radioactive metal tends to need its own chelator and its own recipe—conditions such as temperature, acidity, and reaction time all change. For some medically important metals, especially those used for powerful alpha‑particle therapies, there is still no ideal chelator. The authors argue that this metal‑by‑metal approach has become a bottleneck: the list of useful isotopes keeps growing, but the chemistry to handle them safely cannot keep up.
Nanoparticles as a universal socket
To tackle this, the researchers turned to very small iron oxide nanoparticles, a material already explored for MRI contrast. They designed particles with an iron‑oxide core about three nanometers wide, wrapped in a coating of citrate molecules that keeps them stable in the bloodstream. During a single, 10‑minute microwave‑assisted synthesis step, they doped the core with one of ten different radioactive metals commonly used for PET scans, SPECT scans, or internal radiotherapy. This one‑pot process produced consistently sized particles with high yields and, crucially, excellent stability in human serum—even for challenging therapy isotopes such as radium‑223 and actinium‑225, whose decay products often escape traditional chelators.
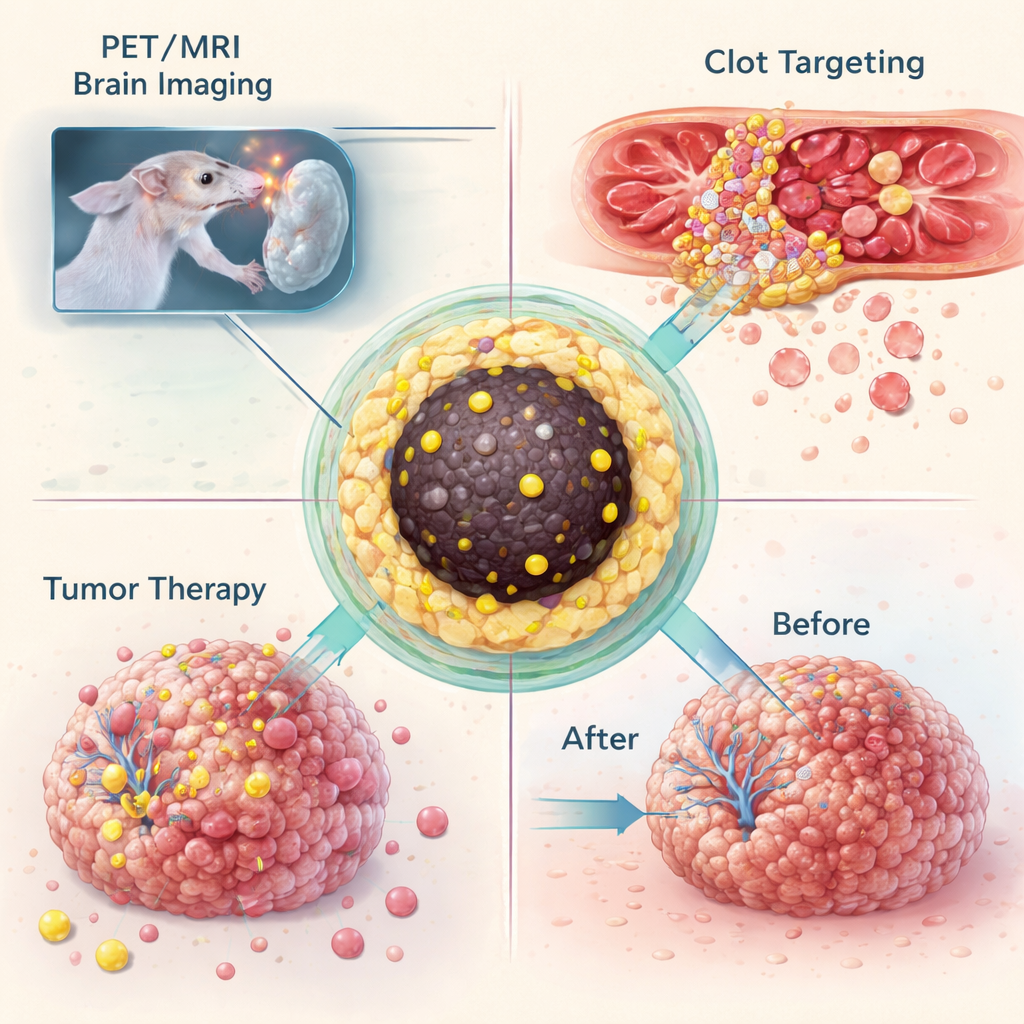
Imaging clots and brain tumors
Having shown they could trap many different isotopes in the same nanoparticle design, the team tested what these "nanotracers" can do in living animals. In a mouse model of glioblastoma, a deadly brain tumor, particles labeled with gallium‑68 were injected into the bloodstream and imaged with combined PET and MRI. The particles accumulated in tumors large enough to disrupt the blood–brain barrier, allowing the same formulation to provide both bright MRI contrast and a sensitive PET signal. In a separate experiment, the particles were chemically modified so they could snap together, via a highly selective click reaction, with an antibody that homes in on activated platelets inside blood clots. In mice with an injured carotid artery, this two‑step "pretargeting" strategy produced clear PET signals at the clot site, while control animals showed no such hotspot.
From diagnosis to treatment and safer clearance
The same platform was also used to deliver treatment. When lutetium‑177–loaded nanoparticles were injected directly into glioblastoma tumors in mice, the particles stayed largely confined to the tumor mass for at least two weeks. Over that time, untreated tumors grew to several times their original size, whereas treated tumors stopped growing, suggesting that the localized radiation dose was sufficient to halt progression. Recognizing that repeated treatments could overload the liver and spleen with iron, the team then adjusted synthesis conditions to make even smaller particles—around four to five nanometers across—that are small enough to be filtered by the kidneys. These shrunken particles still held their radioactive cargo securely but were rapidly excreted into the bladder, reducing long‑term buildup in major organs.
What this could mean for future cancer care
For non‑specialists, the key message is that the researchers have built a single, flexible nanoparticle platform that can be paired with many different radioactive metals without having to reinvent the chemistry each time. In animal models, the same basic particle can help spot brain tumors, highlight blood clots, deliver radiation directly into tumors, and even be tuned to exit the body through the kidneys. While more work is needed before this approach reaches patients, it offers a promising route to designing families of diagnostic and therapeutic agents that share the same behavior in the body, differing only in the type of radiation they carry. That consistency could simplify development, improve safety testing, and ultimately make advanced nuclear medicine tools more broadly available.
Citation: Herraiz, A., Rodríguez-San-Pedro, A., Casquero-Veiga, M. et al. A single-step radiolabeling strategy for PET, SPECT, and therapeutic radionuclides using nanoparticles as a universal chelator. npj Imaging 4, 8 (2026). https://doi.org/10.1038/s44303-026-00142-1
Keywords: radiotheranostics, nanoparticles, PET imaging, glioblastoma, targeted radiotherapy