Clear Sky Science · en
Accelerated and localized synucleinopathy in a hybrid mouse model: implications for positron emission tomography studies
Why this research matters
Parkinson’s disease is best known for tremors and movement problems, but deep inside the brain it begins with tiny changes that are hard to see. Doctors urgently need ways to spot those changes early and to test new drugs quickly. This study describes a refined mouse model that reproduces key features of Parkinson’s-like brain damage in just a few weeks and in a very specific brain region, tailored for use with advanced brain scans called PET imaging. 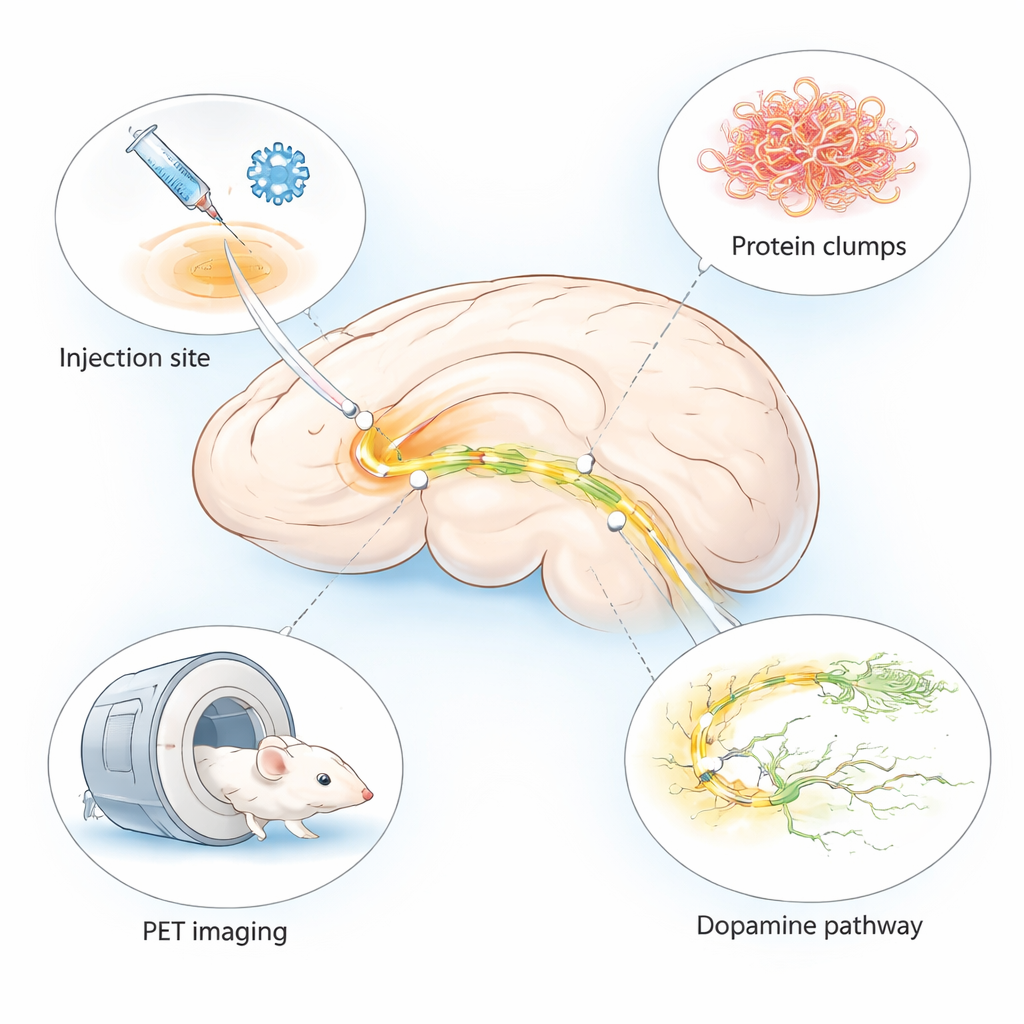
Building a faster, more focused disease model
Many existing animal models of Parkinson’s disease take months to develop symptoms or fail to show the full range of brain changes seen in patients. The authors combined two established approaches into a single “hybrid” model. They injected a harmless virus that makes brain cells produce human alpha‑synuclein—a protein that clumps in Parkinson’s—together with pre‑formed fibrils, which act as seeds that trigger further clumping. Both were delivered into a small, movement‑related region called the substantia nigra on one side of the mouse brain. This design produces local but intense disease‑like changes on the injected side, while the other side remains relatively healthy for comparison.
Tracking protein clumps and dying nerve cells
Within just two weeks of injection, the mice showed high levels of human alpha‑synuclein and its abnormal, phosphorylated form in the targeted brain area. Under the microscope, the researchers saw structures resembling the Lewy bodies and Lewy neurites that are hallmarks of Parkinson’s disease. Over the next several weeks, these abnormal deposits increased. At the same time, nerve cells that produce the chemical messenger dopamine—crucial for smooth movement—started to disappear. Markers of these cells dropped to roughly 60 percent of the level seen on the untouched side by week five, confirming a clear, progressive loss of the dopamine system.
Inflammation, fading synapses, and tired power plants
The brain’s immune cells, called microglia, also switched into a highly active, inflammatory state around the damaged region. Stains for several inflammation‑related proteins were much stronger near alpha‑synuclein deposits than on the opposite side of the brain. Later in the process, the team detected a loss of synapses—the tiny contact points where nerve cells communicate—and signs that mitochondria, the cells’ energy factories, were not working properly. Together, these findings show that the model does not just create protein clumps; it also captures the chain reaction of inflammation, failing connections, and energy problems that contribute to nerve cell death in Parkinson’s disease. 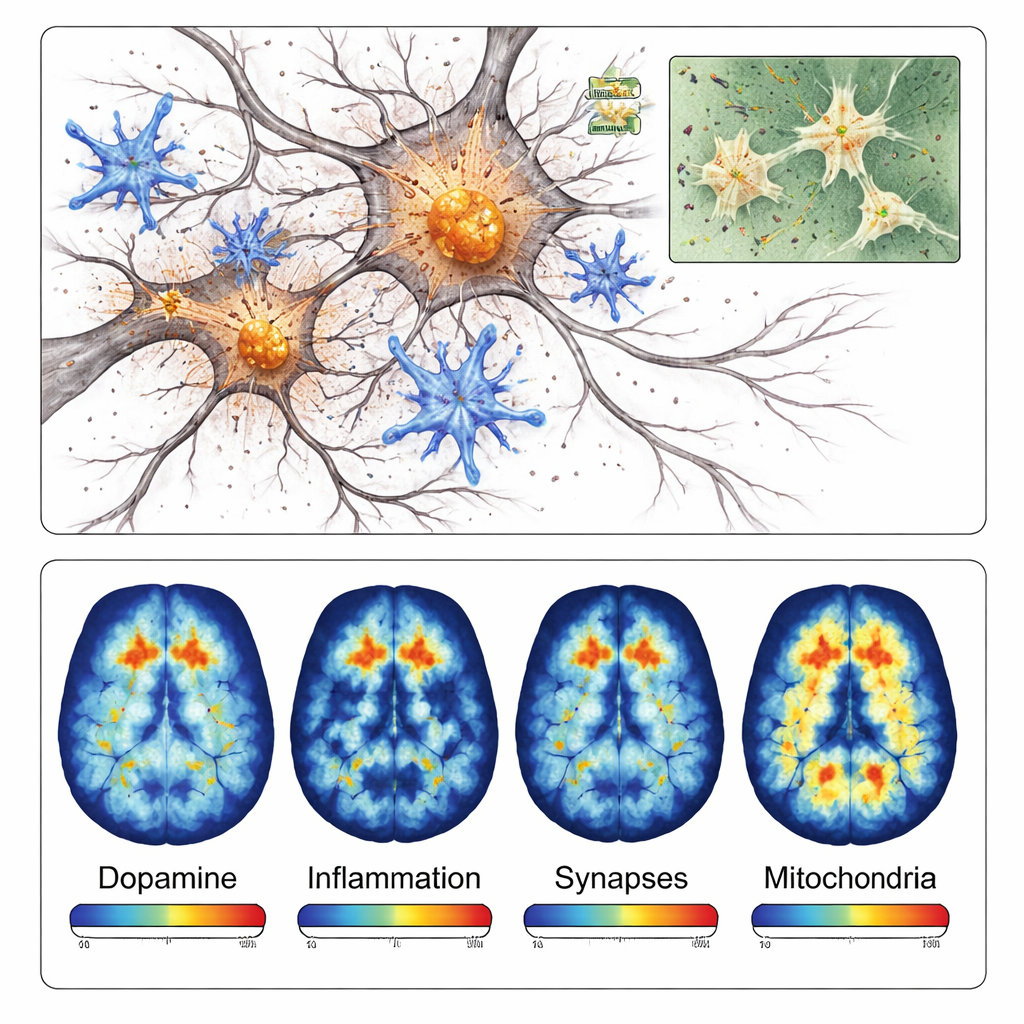
Putting PET scanners to work
Because this model is localized and develops quickly, it is especially well suited to positron emission tomography (PET), a scanning technique that can measure specific molecules in living brains. The researchers tested four different PET tracers that bind to dopamine nerve endings, inflammatory microglia, synaptic proteins, and mitochondrial enzymes. In each case, PET scans showed lower or higher signals on the injected side in ways that matched the microscope findings. For example, the tracer targeting dopamine pathways showed about 40 percent less signal on the damaged side, while the inflammation tracer showed more than 40 percent higher signal in the affected region.
What this means for patients and future treatments
This new mouse model brings together, in a short time frame, many of the key changes that occur over years in the brains of people with Parkinson’s disease. Because the damage is confined to a small, well‑defined area and can be followed with PET scans, researchers can more efficiently test new imaging tracers and potential therapies. While no mouse can fully reproduce the human condition, this approach offers a powerful, practical tool to bridge the gap between basic laboratory work and human studies, ultimately helping to refine diagnosis and accelerate the search for better treatments.
Citation: Xia, C.A., Tsai, HM., Diaz Garcia, S. et al. Accelerated and localized synucleinopathy in a hybrid mouse model: implications for positron emission tomography studies. npj Imaging 4, 7 (2026). https://doi.org/10.1038/s44303-026-00138-x
Keywords: Parkinson’s disease, alpha-synuclein, synucleinopathy, PET imaging, mouse model