Clear Sky Science · en
Site-1 protease mediated GPC processing is required for persistence of LCMV Clone 13
Why this virus story matters
Mammarenaviruses, a family that includes the Lassa fever virus and the lab workhorse LCMV, can cause deadly hemorrhagic disease and serious infections in people, yet we still lack approved vaccines or widely effective treatments. These viruses cloak themselves in a sugary protein coat that must be cut by host enzymes before the virus can spread. This paper asks a deceptively simple question with big implications: why do these viruses rely on a particular host enzyme called S1P, and what happens if we force them to use a more common enzyme, furin, instead?
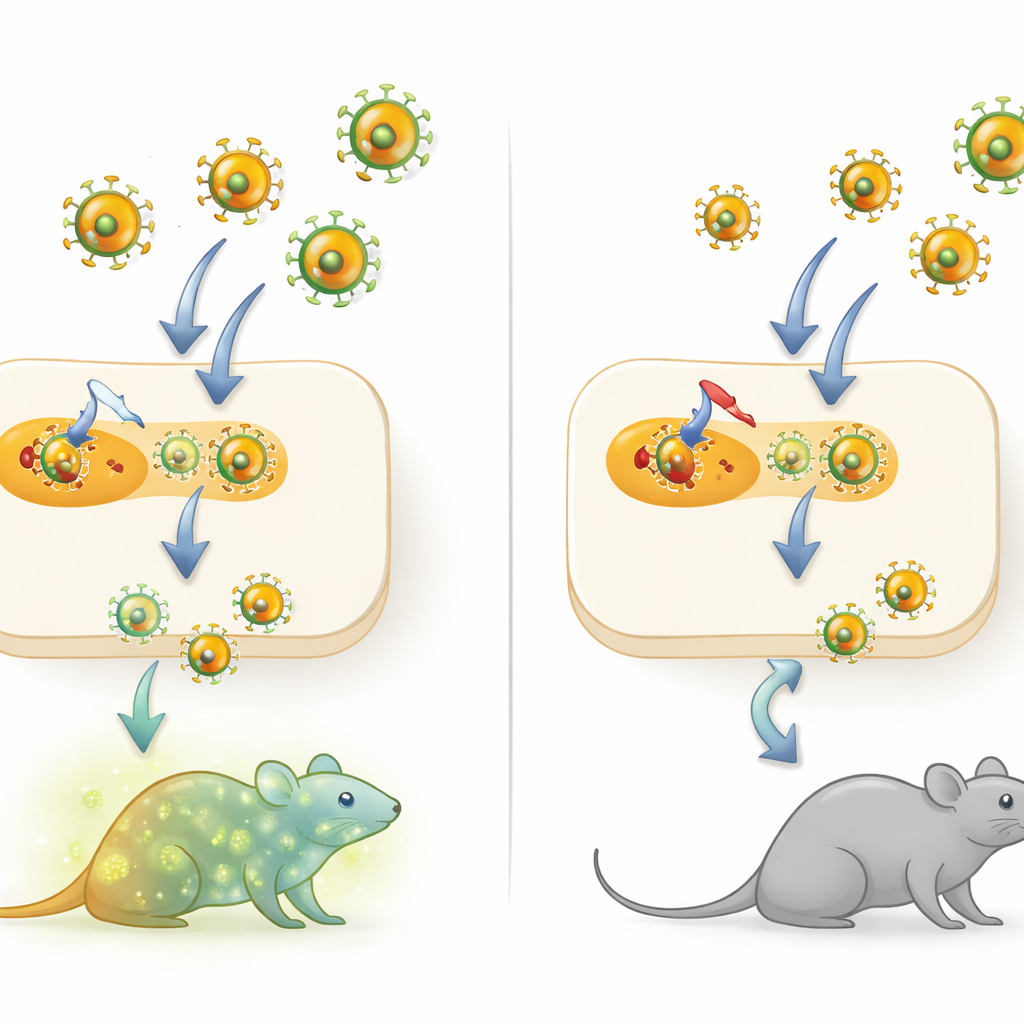
How the virus normally uses our cell machinery
Mammarenaviruses are wrapped in a membrane studded with spike-like proteins that they use to enter cells. These spikes start life as a single long chain, a precursor that must be cut into pieces before it can work. Unlike many other enveloped viruses, which depend on an enzyme called furin for this trimming, mammarenaviruses use another enzyme, S1P. The authors engineered a version of the persistent LCMV Clone 13 strain whose spike precursor could be cleaved by furin instead of S1P, creating a virus they call rCl13-RRRR, and then compared its behavior with the original virus in cells and in mice.
Same punch in the dish, weaker in the animal
In cultured cells, the furin-dependent virus looked surprisingly normal. It grew just as well as the S1P-dependent parent virus and its spike protein fused membranes efficiently, meaning the basic entry machinery still worked. Biochemical tests and the use of specific enzyme inhibitors confirmed that the modified virus really used furin, while the original virus strictly required S1P. This showed that, at least in a controlled cell culture environment, LCMV does not absolutely need S1P to assemble infectious particles.
A persistent virus turned into a cleared infection
The story changed dramatically in living mice. Wild-type LCMV Clone 13 normally establishes long-lasting, high-level infection in immunocompetent mice, a hallmark of this strain. By contrast, when mice were infected with the furin-dependent rCl13-RRRR, virus levels in the blood and organs dropped quickly to below detection and persistence never developed, even though animals clearly made antibodies showing that infection had occurred. Detailed analysis of the spleen showed that the altered virus infected different macrophage subsets and largely failed to reach specialized marginal-zone macrophages that help seed long-term infection, hinting that early tissue targeting is crucial for persistence.
Immune defenses and a built-in vaccine effect
The researchers next asked which parts of the immune system were responsible for clearing the weakened virus. When the type I interferon receptor was knocked out or blocked, rCl13-RRRR rebounded to high levels, showing that interferon is a key early defense. Depleting CD8 T cells also prevented clearance, whereas removing CD4 T cells did not, indicating that virus-killing CD8 T cells are essential. Importantly, unlike animals chronically infected with the original Clone 13, mice infected with rCl13-RRRR retained functional CD8 T cells that produced antiviral cytokines. In lethal challenge models, the furin-dependent virus was far less deadly and, crucially, a single nonlethal infection with rCl13-RRRR protected mice from later otherwise fatal exposure to wild-type Clone 13, both by intravenous and intracranial routes.
What this means for drugs and vaccines
To a non-specialist, the main message is that the choice of host enzyme used to activate a virus’s surface protein can make the difference between lifelong, immune-exhausting infection and a short, protective one. For mammarenaviruses, S1P processing of the spike appears to be a third key requirement for persistent infection, alongside known mutations that boost receptor binding and replication. Because the artificially furin-dependent virus was easily controlled in healthy mice yet still induced strong protection, targeting S1P with drugs or deliberately rewiring viral dependence away from S1P could be a powerful strategy for both antiviral therapies and the design of safer live-attenuated vaccines against dangerous mammarenaviruses such as Lassa virus.
Citation: Zhou, R., Witwit, H., Ai, T. et al. Site-1 protease mediated GPC processing is required for persistence of LCMV Clone 13. npj Viruses 4, 18 (2026). https://doi.org/10.1038/s44298-026-00184-7
Keywords: mammarenavirus, LCMV Clone 13, site-1 protease, viral persistence, live-attenuated vaccine