Clear Sky Science · en
Ordinary differential equation models of SARS-CoV-2 replication dynamics and antiviral drug efficacies
Why Numbers Matter for a New Virus
When COVID-19 emerged, doctors could see what the virus did to patients, but they could not easily predict who would get very sick or when a drug should be given for best effect. This review article looks at how researchers turned virus measurements from patients, animals, and cell cultures into mathematical “movies” of infection inside the body. These models use equations to follow how the virus grows, how our cells and immune system react, and how antiviral drugs and vaccines can tip the scales in our favor.
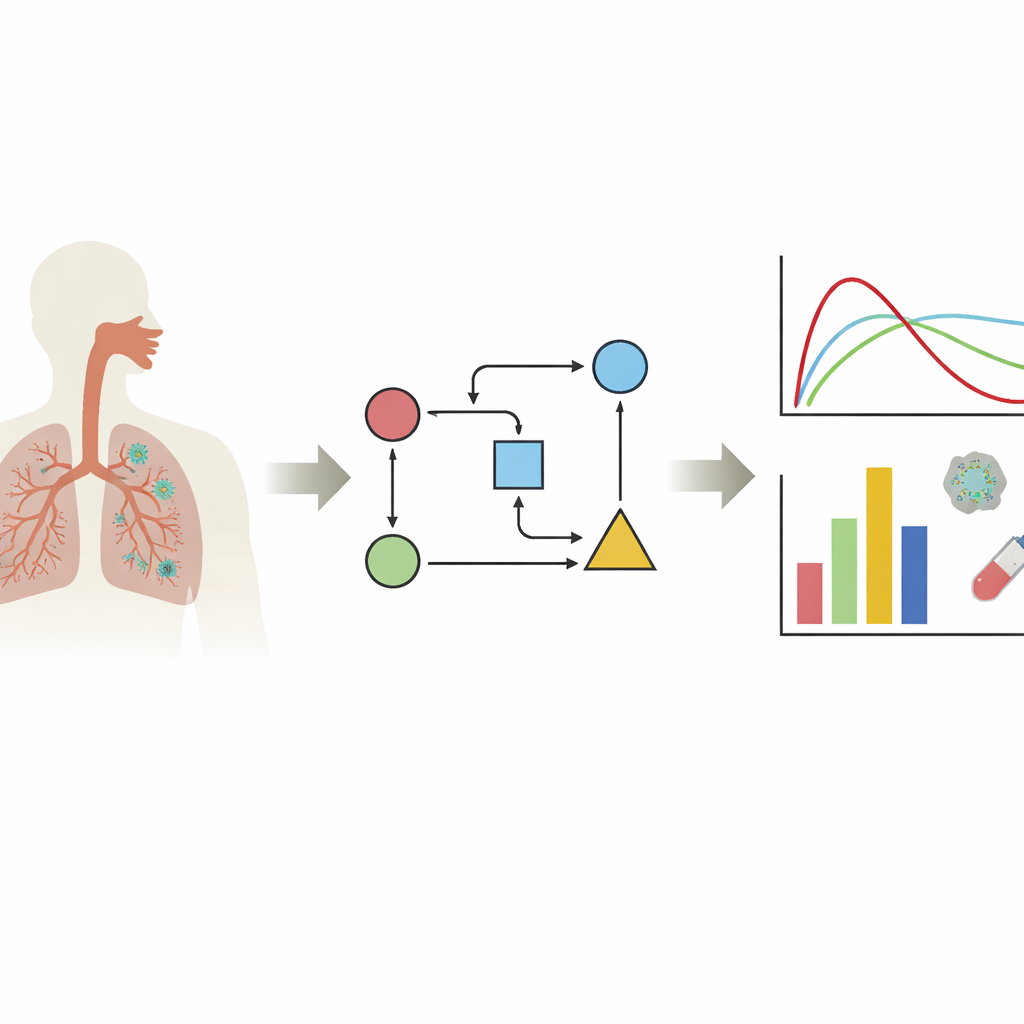
Following the Virus Inside the Body
The authors focus on a family of models called target cell–limited models. In plain terms, these models track three main actors: healthy cells that can be infected, cells that are already infected, and free virus particles. By feeding real data on viral levels into these equations, researchers can estimate hidden features of infection: how quickly the virus infects cells, how long an infected cell produces virus, and how fast virus is cleared. The review shows that most work centers on the tissues that matter most for COVID-19—the nose and the upper and lower airways—while only a few models examine other organs. One key message is that virus behavior can look very different depending on which tissue, which virus variant, and which laboratory cell line is studied, so there is no single “typical” infection curve.
What We Learn About the Immune System
Many models add layers that represent the immune system’s early defenses and its slower, more targeted response. Studies focusing on the first line of defense suggest that quick, well-timed activity from innate immune cells and signaling molecules can blunt the infection, but rarely clears it by itself. Other work highlights the crucial role of T cells and antibodies in finally bringing the virus under control. The models also capture a darker side: if the targeted immune response is late or misdirected, the same tools that should protect us can fuel damaging “cytokine storms,” in which excess inflammatory signals harm healthy tissue. In these simulations, the fine balance between helpful and harmful immune activity often decides whether disease remains mild or becomes life-threatening.
When Drugs and Vaccines Work Best
Because equations can be run again and again under different assumptions, they are powerful tools for testing treatment strategies on a computer before trying them in people. Across many studies, the models agree on one central point: antiviral drugs work best when given very early, before the virus has reached its peak and infected most of the available cells. Treatments that block the virus from copying its genetic material come out as especially promising, particularly when combined with other drugs that act through different mechanisms. Late treatment, in contrast, tends to have little impact in simulations unless several strong drugs are used together. Vaccination is consistently predicted to outperform drugs alone, mainly by priming the immune system so it recognizes the virus quickly and keeps high viral levels short-lived.
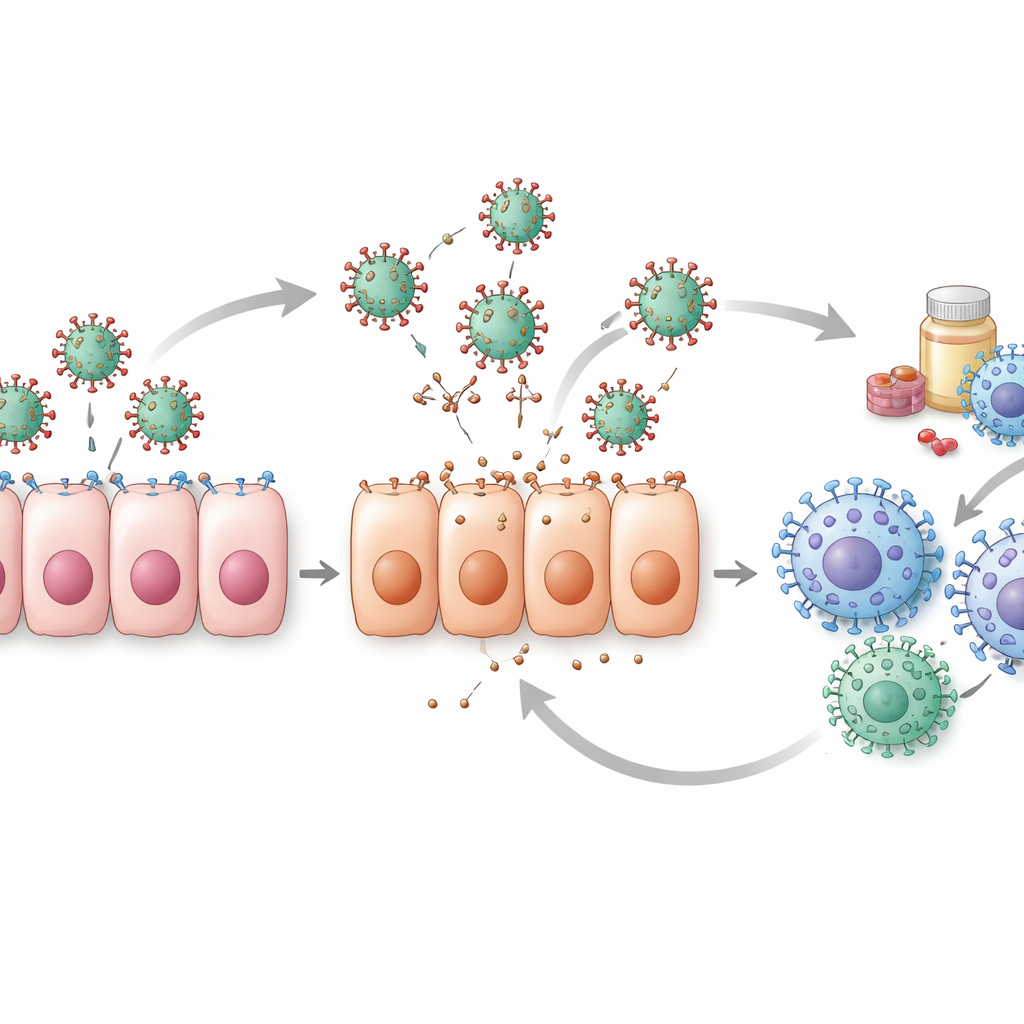
Hidden Challenges in the Data
The review also shines a light on the weaknesses behind the numbers. Most clinical datasets start only after people already feel sick, which means models often miss the crucial first days of infection when virus levels are rising fastest. Without those early measurements, different combinations of model parameters can explain the same data, making it hard to be sure which explanation is right. Only a minority of studies perform rigorous checks to see whether their parameter estimates are truly unique and reliable. Animal studies and cell-culture experiments help fill some gaps, especially for early time points, but differences between species and laboratory systems limit how directly those results can be applied to humans.
What This Means Going Forward
For a non-specialist, the bottom line is that carefully built mathematical models have greatly improved our understanding of how SARS-CoV-2 behaves inside the body and how timing, drug combinations, and prior vaccination shape outcomes. The models broadly agree that early, strong intervention—especially via vaccination and timely antivirals—gives the immune system its best chance to win, while delayed responses, whether from the body or from treatment, are linked to more severe disease. At the same time, the authors stress that these tools are only as good as the data fed into them. To prepare for future variants or new viruses, they call for richer, earlier clinical sampling and broader immune measurements, so that the “numbers behind infection” can guide faster, more accurate decisions in real-world outbreaks.
Citation: Kapischke, T., Herrmann, S.T., Bertzbach, L.D. et al. Ordinary differential equation models of SARS-CoV-2 replication dynamics and antiviral drug efficacies. npj Viruses 4, 17 (2026). https://doi.org/10.1038/s44298-026-00183-8
Keywords: SARS-CoV-2 viral dynamics, within-host modeling, antiviral therapy timing, immune response, COVID-19 vaccination