Clear Sky Science · en
Prolonged dysregulation and pathological changes in the upper respiratory tract of SARS-CoV-2 infected hamsters
Why this research matters for everyday health
Many people recover from COVID-19 only to find that breathing problems, odd breathing patterns, or a constant cough linger for months. This study in golden Syrian hamsters helps explain why symptoms in the nose and upper airways can persist long after the initial infection has cleared, offering clues that may be relevant to long COVID in humans.
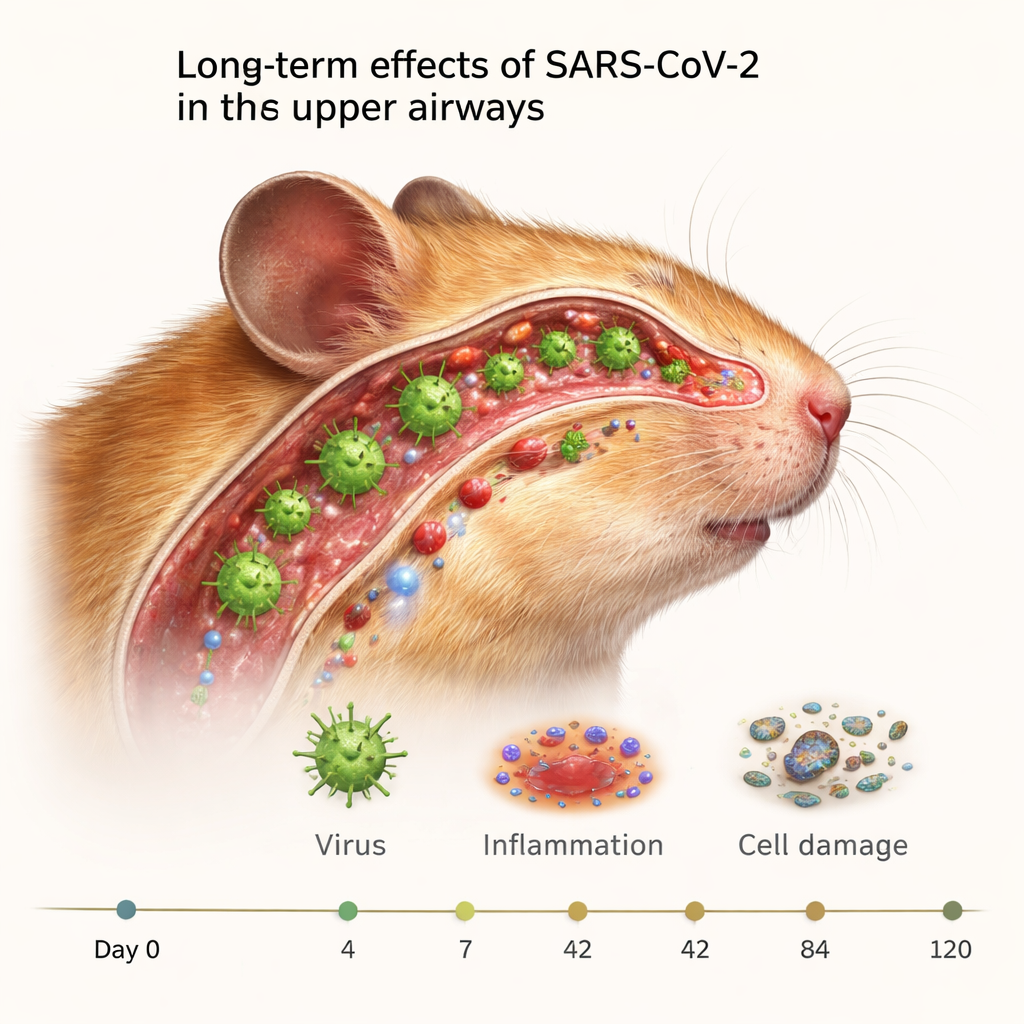
Hidden traces that refuse to go away
The researchers infected hamsters with the original strain of SARS-CoV-2 and examined their nasal tissues over four months, the rough equivalent of many human years. They focused on the nasal turbinates, the folded structures deep inside the nose that warm and filter the air we breathe. Even though standard tests showed that infectious virus disappeared from these tissues within a few weeks, pieces of the virus, including its genetic material and nucleocapsid protein, lingered in some animals up to 120 days after infection. In other words, the virus itself was gone, but its fingerprints were still embedded in the nasal lining.
Inflamed tissue that will not fully heal
Those leftover viral components were not harmless. When the team measured signaling molecules that drive immune responses, they found that many pro-inflammatory markers in the nose—such as interferons and key alarm chemicals that summon immune cells—did not quickly return to normal. Instead, the inflammatory signal gradually rose and peaked almost three months after infection, and even at four months it was still much higher than in uninfected, age-matched hamsters. Microscopic images confirmed that the nasal lining remained abnormally crowded with immune cells and that the delicate surface layer of cells was still being damaged or lost in a fraction of animals long after the acute illness.
Cells caught in a cycle of death and poor repair
The team then asked why the nose seemed stuck in a damaged state. They saw clear signs of ongoing programmed cell death, a controlled way in which cells dismantle themselves. Markers of this process were strongly elevated in the nasal tissues up to 120 days after infection, whereas similar markers in the lungs had largely returned to baseline. At the same time, genes linked to the Notch pathway—a key control system that decides whether cells grow, mature, or die—remained abnormally switched on in the nasal lining. This combination of continued cell death and altered repair signals suggests that the tissue’s self-healing machinery is misdirected, making it hard for the nose to fully rebuild its normal structure and function.
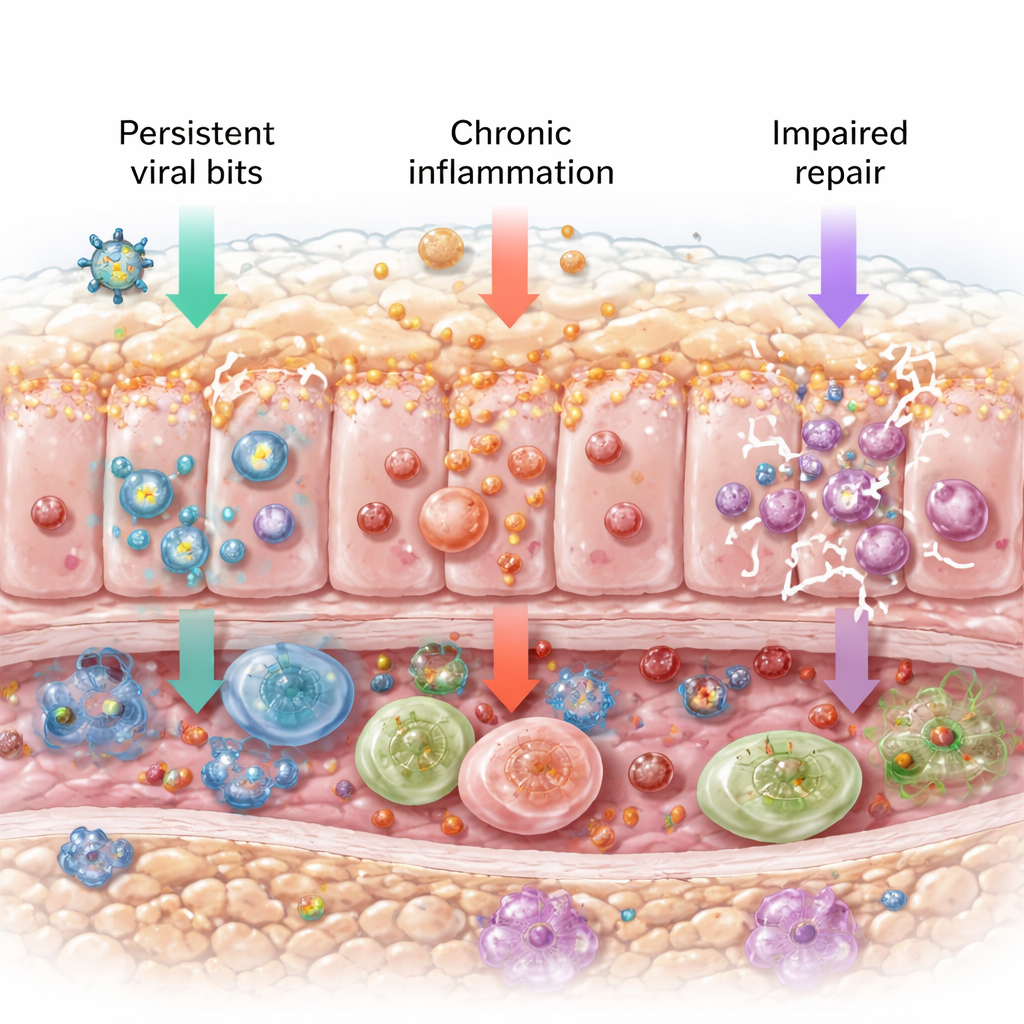
Greater openness to other germs
Another concern raised by the study is increased vulnerability to other respiratory viruses. The researchers measured the activity of several host molecules that different viruses use as door handles to enter cells. In previously infected hamsters, some of these entry factors, especially the receptor CX3CR1 and stress-related proteins like GRP78, stayed elevated long after the first infection. Normal aging already boosted certain entry molecules in uninfected older animals, but prior SARS-CoV-2 infection pushed some of these levels even higher. This pattern hints that a past encounter with SARS-CoV-2 could leave the nasal passages more welcoming to other cold and flu viruses.
What happens when infection strikes again
The team also explored what a second infection would do. When hamsters were re-exposed to SARS-CoV-2 several months after the first bout, their noses showed severe fresh tissue damage—heavy cell loss and immune cell buildup—even though the virus could barely replicate and inflammatory markers were not further increased. This mismatch between limited virus growth and strong tissue injury suggests that non-inflammatory forces, such as killer immune cells or cellular stress, can still inflict substantial harm on already fragile nasal tissue during reinfection.
What it means for people living with long-term symptoms
Taken together, the findings paint a picture of a nose that remains biologically disrupted long after SARS-CoV-2 stops multiplying. Residual viral bits appear to keep the immune system on a low boil, driving ongoing cell death, faulty repair programs, and structural damage in the upper airways. At the same time, the nasal lining may become more permissive to other viruses and more easily injured during repeat infections. While hamsters are not humans, this work offers a plausible biological explanation for persistent nasal and breathing problems in people with long COVID and highlights the need for treatments that calm chronic inflammation and support proper tissue repair, rather than focusing only on clearing active virus.
Citation: Liu, F., Xia, Y., Lee, A.CY. et al. Prolonged dysregulation and pathological changes in the upper respiratory tract of SARS-CoV-2 infected hamsters. npj Viruses 4, 15 (2026). https://doi.org/10.1038/s44298-026-00181-w
Keywords: long COVID, nasal inflammation, SARS-CoV-2 persistence, upper respiratory tract, hamster model