Clear Sky Science · en
Immunogenicity and protective efficacy of MERS CoV, NL140422, and HKU4 spike protein nanoparticle vaccines
Why this research matters to you
After living through COVID-19, many people wonder what other coronaviruses might be waiting in the wings—and whether we can be ready before they spread. This study explores experimental vaccines aimed not just at the known Middle East respiratory syndrome virus (MERS-CoV), but also at closely related animal viruses that could someday jump into humans. The work offers a glimpse of how scientists are trying to build vaccines that protect against whole families of viruses, not just one at a time.
Hidden threats in animals
MERS first appeared in humans in 2012 and is deadlier, person for person, than COVID-19, with about a third of known patients dying from the infection. So far, most human cases have been linked to contact with infected camels in the Middle East, but related viruses are found in bats and other animals. Some of these animal viruses, including ones called NL140422 and HKU4, can already infect human cells in the lab by latching onto the same doorway on our cells that MERS uses. Because they belong to the same subgroup of coronaviruses, called merbecoviruses, they represent potential future spillover threats.
Building a nanoparticle vaccine
The researchers set out to design vaccines that display the “spike” protein— the knob-covered structure that coronaviruses use to enter cells—from three different merbecoviruses: MERS-CoV, NL140422, and HKU4. Instead of giving the spike protein alone, they attached many copies of each spike to a tiny hollow particle made from a bacterial virus shell. These virus-like particles act like a scaffold, presenting dozens of spikes in a tightly packed, spherical array. This multicolored display is meant to grab the attention of the immune system and train it to recognize the spikes more strongly than a single protein might.
Testing in mice
To see whether these vaccines worked, the team immunized ordinary laboratory mice with one of the three spike-decorated nanoparticles or with an empty particle as a control, all given with a standard booster substance. The mice produced high levels of antibodies that recognized the specific spike they had been vaccinated with, and the antibodies also showed some ability to bind the other two merbecovirus spikes. However, when the scientists looked for antibodies that could actually block live MERS virus from infecting cells, only the vaccine containing the true MERS spike produced measurable virus-blocking activity.
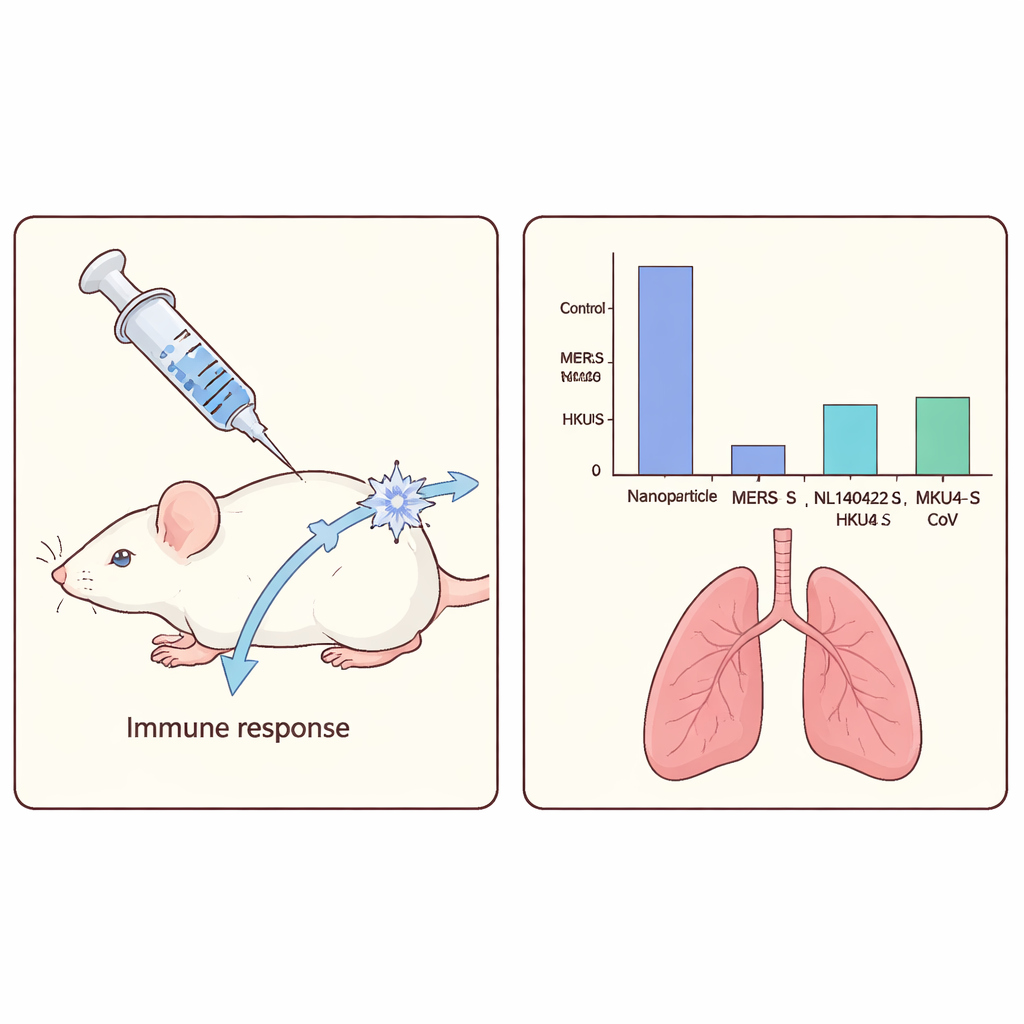
Protection against real infection
Next, the researchers tested how well the vaccines could protect against disease. For this, they used genetically engineered mice that make the human version of the cell-surface protein that MERS and related viruses use to enter cells, making these animals susceptible to MERS infection. After a single vaccination, the mice were exposed to a high dose of MERS virus through the nose. In unvaccinated animals, high levels of virus were found in the lungs and upper airways. Mice that had received the MERS-based nanoparticle vaccine, by contrast, had no detectable virus in either location, indicating complete protection. Mice vaccinated with the NL140422 or HKU4 spike vaccines still became infected, but the amount of virus in their lungs dropped by about 50- to 300-fold compared with controls, showing partial protection. These two vaccines did not meaningfully reduce virus levels in the nasal passages.
Steps toward broader coronavirus vaccines
The study shows that a nanoparticle vaccine carrying the MERS spike can fully protect susceptible mice from a strong viral challenge after just one dose, and that vaccines based on related animal viruses can still blunt infection even when they do not generate classic virus-blocking antibodies. This suggests that other arms of the immune system, such as non-blocking antibodies that flag infected cells for destruction or killer T cells, may also play important roles. Although the work is still at an early, animal-testing stage and was only able to measure protection against MERS itself, it outlines a strategy for building “family-wide” coronavirus vaccines. In everyday terms, the research moves us a step closer to shots that could soften or even prevent future coronavirus outbreaks before they begin.
Citation: Halfmann, P.J., Lee, J.S., Wang, T. et al. Immunogenicity and protective efficacy of MERS CoV, NL140422, and HKU4 spike protein nanoparticle vaccines. npj Viruses 4, 12 (2026). https://doi.org/10.1038/s44298-026-00179-4
Keywords: MERS vaccine, coronavirus, nanoparticle vaccine, spillover viruses, broad protection