Clear Sky Science · en
Divergent roles of Hsp70 chaperones in orthoflavivirus protein secretion and virion formation
Why tiny heat‑shock helpers matter for brain‑infecting viruses
Viruses carried by ticks and mosquitoes, such as tick‑borne encephalitis virus and West Nile virus, can invade the brain and cause serious illness, yet there are still no specific medicines to stop them. This study looks at how these viruses hijack a set of our own protein “helpers,” called Hsp70 chaperones, to build and release new virus particles. Because Hsp70 proteins are already being explored as drug targets in cancer, understanding this relationship could open new ways to treat dangerous viral infections without attacking the viruses directly.
Viruses that lean on the cell’s folding machinery
Orthoflaviviruses are small RNA viruses whose genetic material encodes a long polyprotein that must be cut and folded into several functional pieces. Two key viral components are the envelope protein E, which coats the virus particle and allows it to enter new cells, and the non‑structural protein NS1, which helps the virus copy its genome and can be secreted to manipulate the immune system. All of this takes place in the endoplasmic reticulum, the part of the cell where many proteins are folded and prepared for export. There, the cell relies on Hsp70 chaperones, including a resident form called BiP, to keep newly made proteins from misfolding under stress. The authors asked how strongly neurotropic tick‑ and mosquito‑borne orthoflaviviruses depend on these chaperones to assemble infectious particles.
Direct handshakes between viral proteins and Hsp70
Using cells engineered to overproduce specific viral proteins, as well as cells infected with authentic viruses, the researchers pulled down BiP or Hsp70 and checked which viral partners came along. They found that BiP and Hsp70 consistently formed complexes with the envelope E protein from several orthoflaviviruses, whether carried by ticks or mosquitoes. This suggests that folding of E inside the endoplasmic reticulum is guided by these chaperones. The picture was more selective for NS1: BiP and Hsp70 bound strongly to NS1 from the tick‑borne viruses tick‑borne encephalitis virus and Langat virus, but not to NS1 from the mosquito‑borne West Nile and Usutu viruses. These differences likely reflect variations in how NS1 is decorated with sugar chains and how it folds, hinting that not all related viruses use the same host pathways.
Blocking chaperones sharply lowers virus output
Because Hsp70 proteins are druggable, the team tested two small molecules that interfere with different parts of the chaperone machinery. YM‑1 targets the nucleotide‑binding domain, freezing Hsp70 proteins in an inactive state, while PES‑Cl blocks the substrate‑binding pocket that normally grips client proteins. At doses that left most cells alive, YM‑1 drastically reduced the amount of infectious virus released for all four tested orthoflaviviruses and lowered the amount of envelope protein found in culture fluids, pointing to a general block in particle formation or release. PES‑Cl, in contrast, had little effect on infectious titers or envelope protein levels, even though it weakened the physical interaction between Hsp70/BiP and the envelope protein in binding assays. This suggests that briefly interfering with the gripping site is not enough to cripple virus assembly, whereas shutting down the chaperone’s energy‑driven cycle is.
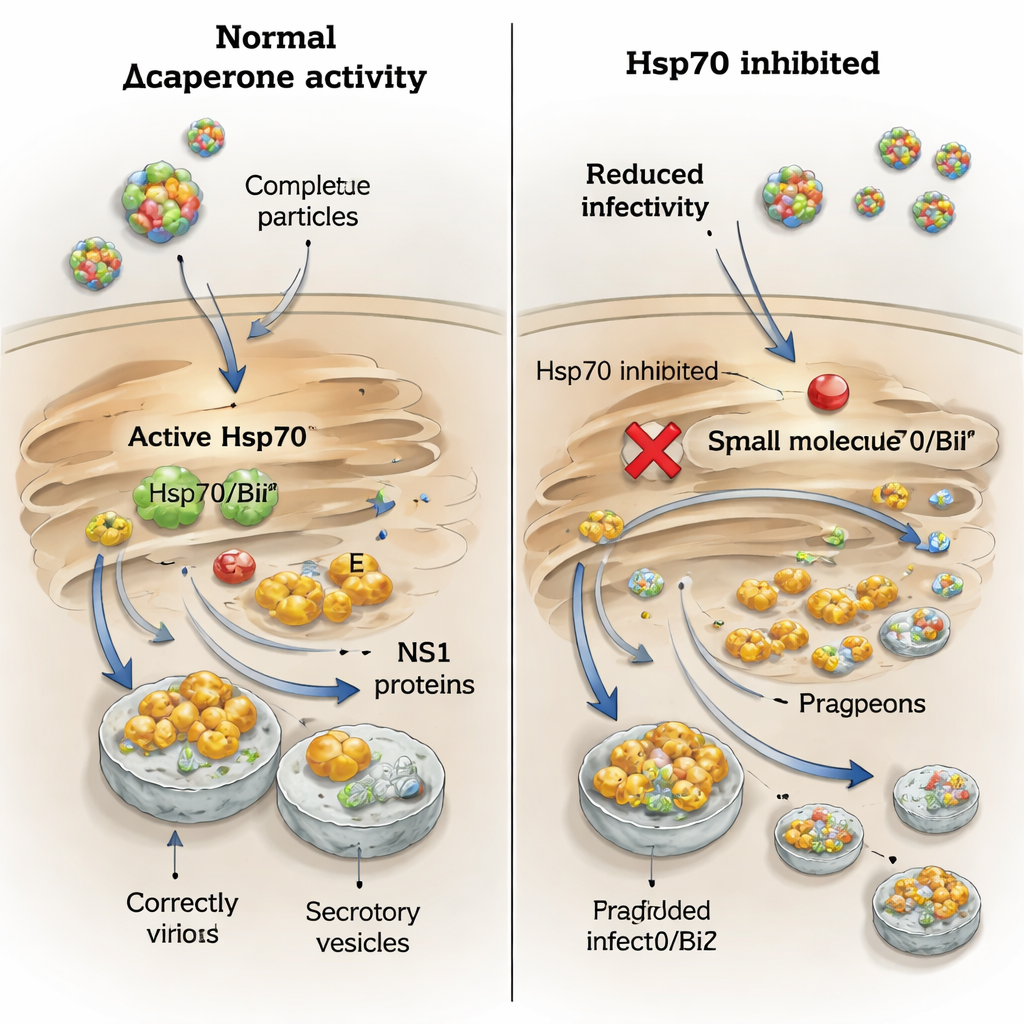
A special dependence of tick‑borne viruses on BiP for NS1
The story for NS1 was more nuanced. When infected cells were treated with PES‑Cl, secretion of NS1 from tick‑borne encephalitis and Langat viruses dropped sharply, but NS1 from West Nile and Usutu viruses was unaffected. This mirrors the earlier finding that only the tick‑borne NS1 proteins bind strongly to BiP and Hsp70. To zoom in on BiP’s role, the authors used small interfering RNAs to lower BiP levels specifically. This reduced the amounts of both envelope and NS1 proteins inside cells and in the surrounding fluid for tick‑borne encephalitis and West Nile viruses, yet infectious virus titers fell only modestly. When BiP‑depleted cells were treated with drugs that block protein degradation, NS1 levels inside the cell rebounded but its secretion did not, showing that BiP is needed not just to protect NS1 from breakdown but also to guide it along the secretory route.
What this means for future antiviral strategies
For a non‑specialist, the main message is that these brain‑infecting viruses strongly rely on the cell’s own protein‑folding helpers to build their outer coat and, for some species, to send out NS1 as a secreted weapon. A broad‑acting chaperone inhibitor like YM‑1 can therefore slash the production of infectious particles from multiple related viruses at once, although its powerful effects likely go beyond a single chaperone and could stress healthy cells too. More targeted approaches, possibly focusing on how BiP and Hsp70 handle specific viral clients such as NS1, might one day help design treatments that are harder for viruses to resist while sparing most normal protein folding in the host.
Citation: Blank, L., Lorenz, C. & Steffen, I. Divergent roles of Hsp70 chaperones in orthoflavivirus protein secretion and virion formation. npj Viruses 4, 8 (2026). https://doi.org/10.1038/s44298-026-00175-8
Keywords: orthoflavivirus, Hsp70 chaperone, tick-borne encephalitis virus, West Nile virus, antiviral drug targets