Clear Sky Science · en
Mechanistic insights into the impact of prenatal viral infections on maternal and offspring immunity
Viruses and Pregnancy: Why It Matters for Families
Every year, millions of pregnancies occur in the shadow of viral outbreaks such as HIV, COVID-19, and influenza. Even when babies are not directly infected, scientists are learning that a mother’s immune response to these viruses can leave lasting marks on her child’s health. This review pulls together cutting-edge research to explain how common prenatal viral infections subtly reshape the womb environment, the placenta, and the developing immune system, with consequences that may echo from birth into adulthood.
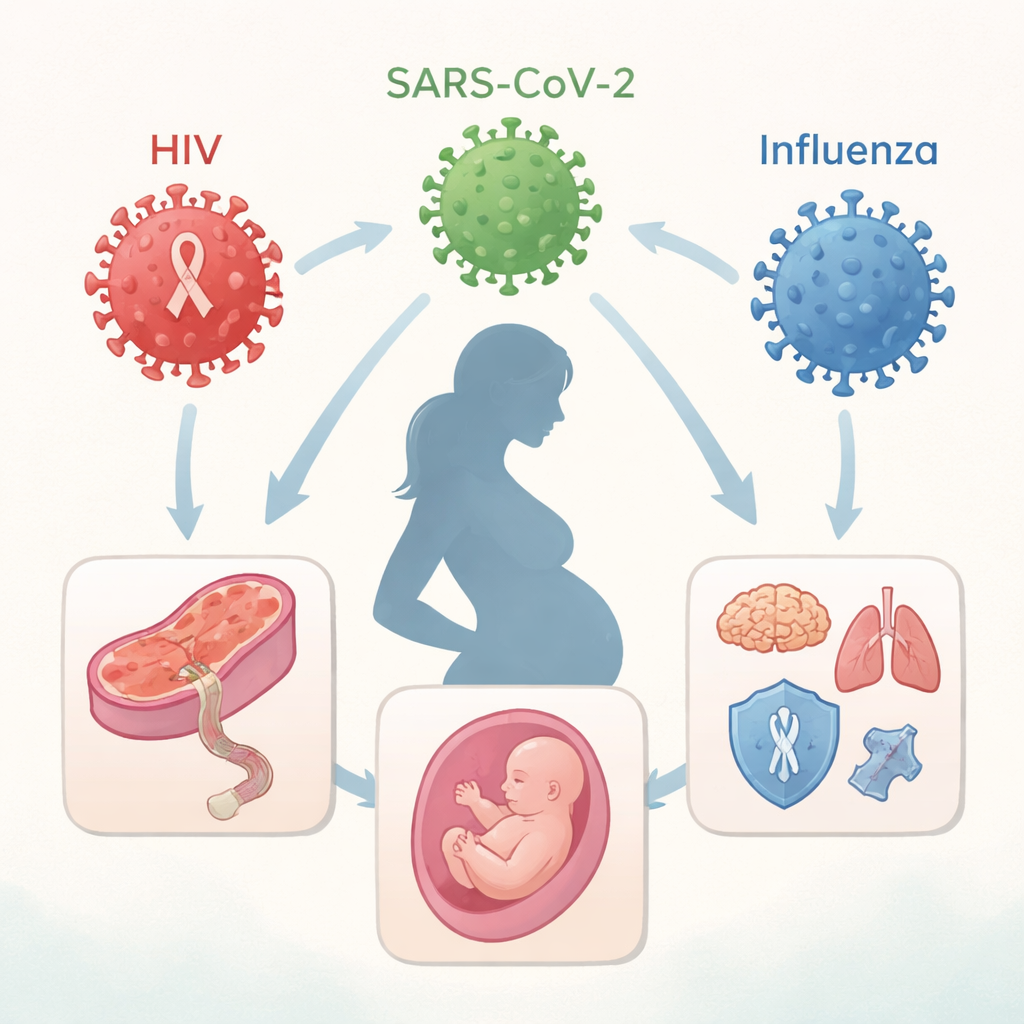
Three Different Viruses, One Shared Challenge
The article focuses on three globally important viruses: HIV, SARS-CoV-2 (the cause of COVID-19), and influenza. These infections differ in how they spread and how long they persist—HIV is chronic, while COVID-19 and influenza are usually short-lived. Yet all three can disrupt the delicate balance of the maternal immune system during pregnancy. Traditionally, doctors worried mainly about “vertical transmission,” where the virus itself crosses the placenta and infects the fetus. Now, evidence shows that even without direct fetal infection, the mother’s immune reaction and inflammation can change the placenta’s structure, alter blood flow and nutrient delivery, and influence how the baby’s immune and nervous systems develop.
How the Placenta Becomes a Battleground
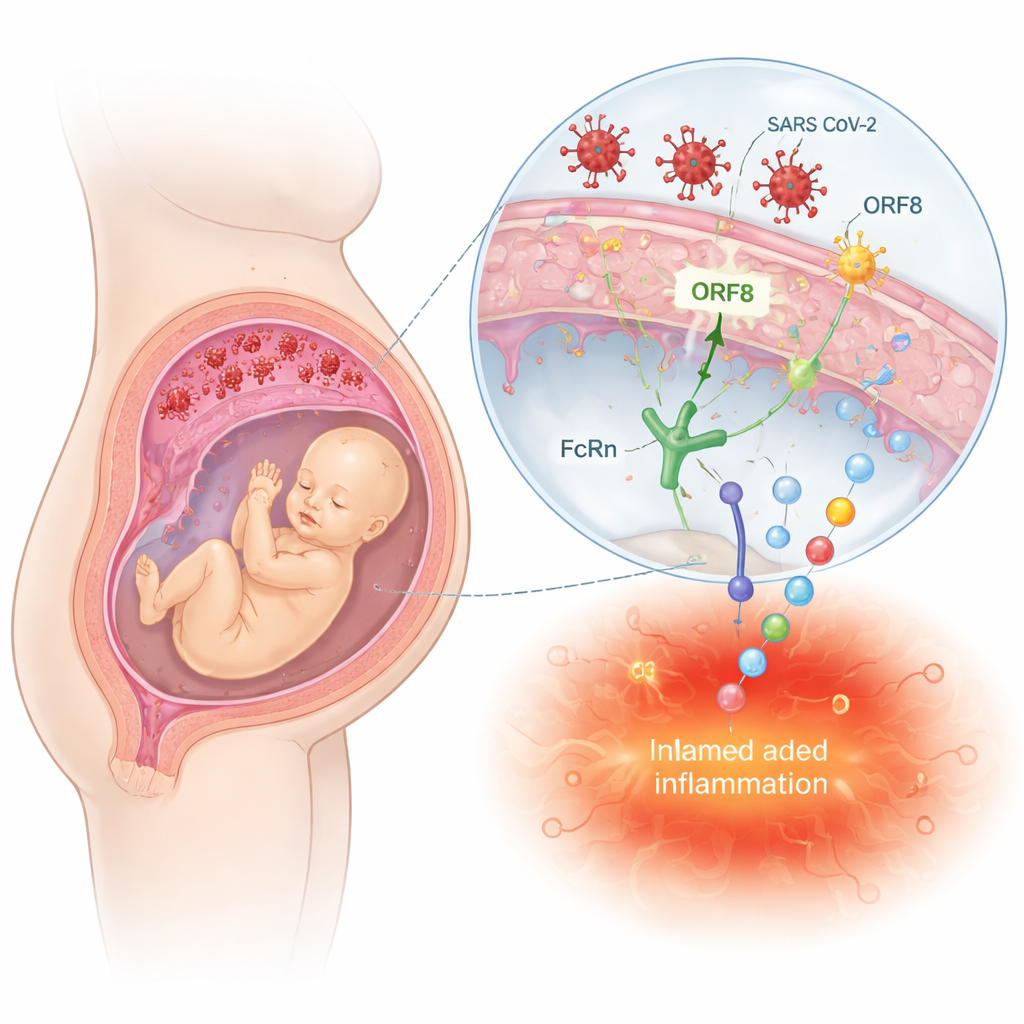
The placenta serves as both shield and bridge between mother and fetus. HIV can invade this barrier by infecting immune cells and placental cells, or by hitchhiking on antibodies that normally carry protective proteins to the fetus. In contrast, SARS-CoV-2 and influenza rarely reach fetal tissues, but they can still damage placental blood vessels and trigger inflammatory cascades. The review describes how viral proteins, such as the HIV Tat and Nef proteins or SARS-CoV-2’s ORF8 and spike protein, interact with receptors and signaling pathways in placental and immune cells. These interactions can set off strong inflammatory responses, interfere with antiviral defenses, and disturb hormone production—changes that may not cause obvious birth defects but can quietly reprogram fetal development.
Lasting Imprints on the Child’s Immune and Brain Health
One of the most important ideas in the paper is that prenatal infection can “train” the infant immune system in ways that persist long after birth. Babies exposed to HIV in the womb but born uninfected often show altered numbers and function of key immune cells, higher levels of inflammatory molecules, and differences in their gut bacteria. Similar patterns are emerging for children exposed to SARS-CoV-2 and, in animal studies, to influenza. These early shifts are linked to higher risks of serious infections in infancy, changes in vaccine responses, and possible increases in allergies and immune-mediated diseases. For the brain, maternal immune activation—especially surges of cytokines like IL-6 and IL-17—has been associated in human and animal studies with later neurodevelopmental issues, including learning difficulties, behavioral problems, and, in some cases, higher rates of conditions such as autism and schizophrenia.
Protecting Mothers and Babies: What Can Be Done?
The review also examines how clinical care is evolving to reduce these risks. For HIV, early and sustained antiretroviral therapy for the mother, coupled with tailored drug regimens for the newborn, has driven direct transmission rates below 1% in many settings, though inflammation-related effects still require careful follow-up. For COVID-19 and influenza, maternal vaccination stands out as a powerful tool: vaccinated mothers pass protective antibodies across the placenta, lowering the chances that their infants will be hospitalized in the first months of life. Emerging strategies include monoclonal antibodies, small-molecule antivirals, and therapies that carefully dampen excessive inflammation without suppressing essential immune defenses—approaches that must be tested specifically in pregnant populations.
What This Means for Future Parents and Children
Overall, the article concludes that prenatal exposure to viruses is not only an immediate threat but also a “long game” for health. Even when babies escape direct infection, the maternal immune response can subtly shape their immune system, brain development, and microbiome, influencing how they respond to infections and possibly their risk of chronic conditions later in life. Understanding these pathways opens the door to smarter vaccines, safer antiviral drugs in pregnancy, and better long-term monitoring of exposed children. For families and clinicians alike, the message is clear: protecting maternal health during pregnancy is an investment in the lifelong health of the next generation.
Citation: Salem, G.M., Azamor, T., Familiar-Macedo, D. et al. Mechanistic insights into the impact of prenatal viral infections on maternal and offspring immunity. npj Viruses 4, 7 (2026). https://doi.org/10.1038/s44298-026-00174-9
Keywords: prenatal viral infection, maternal immune activation, placental inflammation, offspring immunity, neurodevelopment