Clear Sky Science · en
STAT1 signaling controls cholesterol metabolism in epithelial cells and RSV-induced syncytia formation
Why this virus and your cholesterol are connected
Respiratory syncytial virus, or RSV, is best known as a winter bug that fills pediatric wards and threatens frail older adults. This study reveals that our cells do not just fight RSV with immune molecules—they also reshuffle their fats, especially cholesterol, to hold the virus in check. Understanding how a key immune switch called STAT1 keeps cholesterol under control may open new ways to limit lung damage in people at highest risk from RSV.
An airways virus with outsized impact
RSV is a leading cause of serious lung infections such as bronchiolitis and pneumonia in infants, older adults, and people with weakened immune systems. Vaccines and long-acting antibodies now help prevent infection, but once someone is sick, there are still few treatment options. One hallmark of severe RSV disease is the formation of “syncytia” – large, fused blobs of airway cells created when the virus forces neighboring cells to merge. These fused cells can damage the delicate lining of the airways and may speed local spread of the virus.
A master immune switch that also guards lipids
The researchers focused on STAT1, a protein switched on by interferons, the body’s early alarm signals during viral infection. STAT1 is well known for turning on dozens of antiviral genes, but mounting evidence suggests it also rewires how cells handle cholesterol. Cholesterol is a core building block of cell membranes and a favored tool that many viruses use to enter, replicate, and exit cells. By engineering human airway-like cells that completely lacked STAT1, and comparing them with normal cells, the team asked how this immune switch shapes cholesterol balance and, in turn, RSV-driven cell fusion.
When STAT1 is missing, cholesterol piles up and cells over-fuse
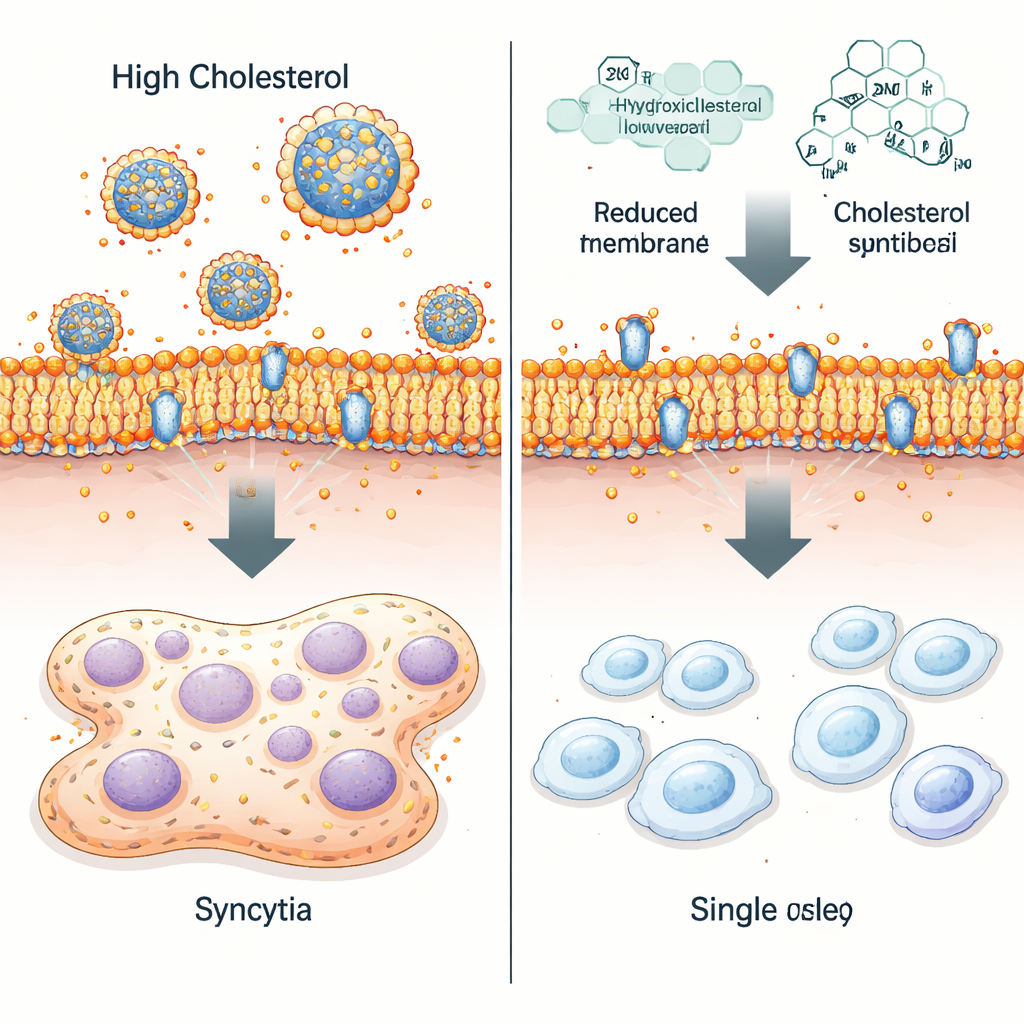
Using genome editing, the scientists knocked out both copies of the STAT1 gene in HEp-2 epithelial cells. The modified cells grew at a normal rate but adopted a more elongated shape, hinting that their membranes were altered. Detailed gene activity profiles showed broad changes in pathways that control cholesterol production, transport, and storage. Biochemical tests confirmed that, even without infection, STAT1-deficient cells accumulated about twice as much free cholesterol as normal cells. When both cell types were infected with RSV, viral titers—the amount of infectious virus produced—were surprisingly similar. What changed dramatically was how the cells looked: STAT1-lacking cultures developed far larger and more numerous syncytia than normal cells.
Dialing cholesterol up and down changes fusion, not virus count
The team then directly manipulated cholesterol levels. Chemicals that temporarily stripped cholesterol from the cell surface, or drugs that reduce cholesterol synthesis and trafficking, all shrank the size of RSV-induced syncytia—especially in STAT1-deficient cells. A naturally occurring cholesterol derivative, 25-hydroxycholesterol, was particularly effective: it lowered membrane cholesterol, disrupted the even coating of RSV fusion (F) protein on the cell surface, and largely restricted infection to individual cells instead of sprawling fused clusters. Yet overall virus growth in the culture remained close to that seen in untreated cells, indicating that cholesterol control mainly influenced how destructively the virus spread from cell to cell.
How this may matter for patients at risk
These findings outline a STAT1-dependent “immune–metabolic” pathway in which interferon signals help restrain RSV by tightening control over cholesterol and preventing excessive cell fusion. When STAT1 signaling is weakened—by genetic defects, immature infant immunity, or age-related changes in older adults—cholesterol handling becomes sloppy, cell membranes become richer in free cholesterol, and RSV can drive more aggressive syncytia formation. The study suggests that drugs which lower cellular cholesterol or mimic 25-hydroxycholesterol’s effects might complement direct antivirals, especially in people prone to severe RSV disease.
The big picture: cholesterol as a lever on RSV severity
For non-specialists, the message is that cholesterol in lung cells is not just a heart health issue; it also helps decide how badly RSV can damage the airways. STAT1 acts as a traffic cop, keeping cholesterol in balance during infection so that RSV has fewer opportunities to fuse cells together. When this control fails, cholesterol builds up and syncytia flourish, potentially worsening disease. Because many older adults already take cholesterol-lowering medications, the work raises the intriguing possibility that managing cholesterol might one day be part of protecting vulnerable people from the most dangerous forms of RSV infection.
Citation: Agac, A., Ludlow, M., Knittler, MC. et al. STAT1 signaling controls cholesterol metabolism in epithelial cells and RSV-induced syncytia formation. npj Viruses 4, 10 (2026). https://doi.org/10.1038/s44298-026-00173-w
Keywords: respiratory syncytial virus, STAT1, cholesterol metabolism, syncytia formation, antiviral immunity