Clear Sky Science · en
The soluble G protein of respiratory syncytial virus promotes viral dissemination via TLR2-mediated NLRP3 priming and pyroptosis
Why this lung virus matters to everyone
Respiratory syncytial virus, or RSV, is best known as a wintertime menace for babies, but it also sends many older adults and people with weak immune systems to the hospital. Even with new vaccines and protective antibodies, RSV still reinfects people again and again and can cause severe lung damage. This study looks at a little-known viral protein, a soluble form of the RSV G protein, and uncovers how it may quietly prepare lung cells for explosive inflammation and help the virus spread more efficiently through the airways.
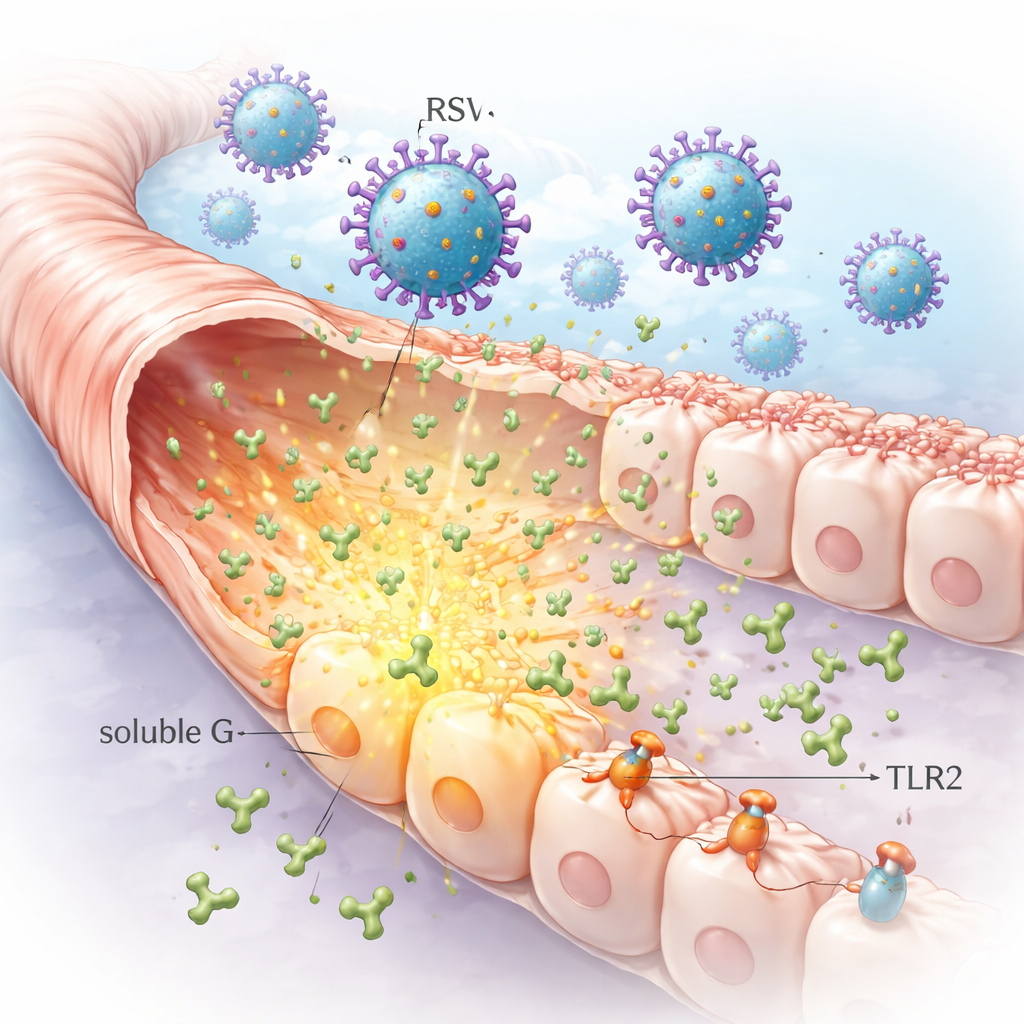
A secreted viral protein that travels ahead of infection
RSV carries an attachment protein called G on its surface that helps it latch onto airway cells. Unusually, infected cells also release large amounts of a free-floating version of this protein, known as soluble G. The researchers showed that lung cells infected in the lab can shed very high levels of soluble G into their surroundings. Because this form can diffuse away from the original infection site, it can reach neighboring cells that are not yet infected, setting the stage for broader changes in the lung lining than the virus alone would cause.
Soluble G as a double-duty key to cell surfaces
By combining microscopy, biochemical pull-down tests, and enzyme treatments, the team mapped how soluble G sticks to cells. They found that it can attach to sugary chains called glycosaminoglycans, as well as to a known RSV receptor called CX3CR1. Crucially, soluble G also binds to a pattern-sensing molecule on immune and airway cells called TLR2. A tiny motif in the G protein, known as CX3C, proved important for strong binding to CX3CR1 and for robust recruitment of TLR2. This means soluble G can first be captured loosely on the cell surface and then engage more specific receptors that control how cells respond to threats.
Priming lung cells for an inflammatory kind of cell death
Engagement of TLR2 by soluble G turned out to be far from harmless. In reporter immune cells and human lung cell lines, soluble G activated the MyD88–NF-κB signaling route downstream of TLR2, leading to the release of inflammatory messengers such as IL-6 and IL-8. At the same time, it boosted production of components of a molecular "alarm system" called the NLRP3 inflammasome and enzymes that generate reactive molecules like nitric oxide and oxygen radicals. On their own, these changes caused only mild damage. But when primed cells were later infected with RSV, the second hit triggered full inflammasome assembly, activation of the enzyme caspase‑1, formation of pores in the cell membrane, and a fiery form of cell death known as pyroptosis. This process punched holes in the cells, spilled inflammatory contents, and coincided with higher amounts of infectious virus being released into the culture fluid.
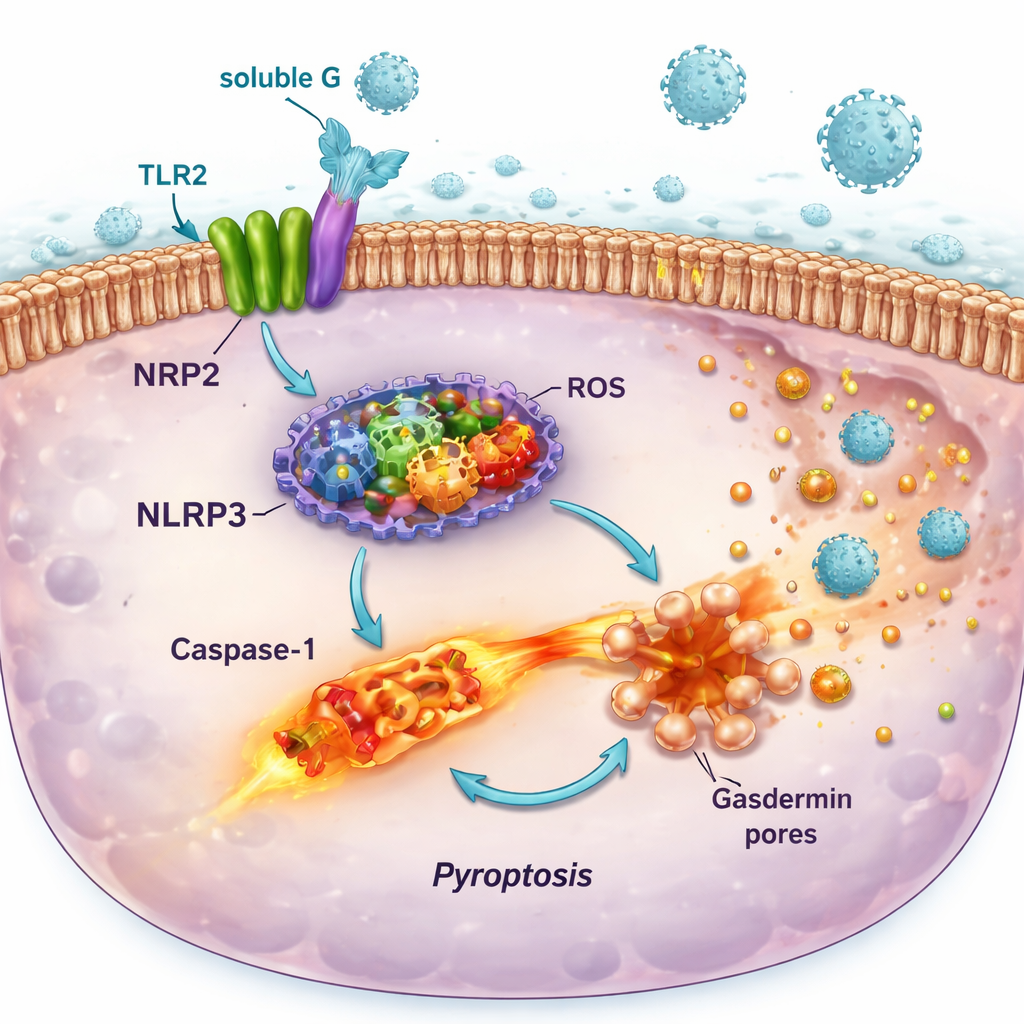
How this chain reaction may worsen lung disease
The pro-death effect of soluble G was strongest in airway epithelial cells—the very cells RSV prefers to infect—and depended on the NLRP3 inflammasome, because a specific NLRP3 inhibitor largely rescued cell viability. In contrast, certain immune cells rich in TLR2 but poor in the CX3CR1 receptor did not undergo strong lytic death under the same conditions. This pattern suggests that RSV uses soluble G to selectively weaken and destroy the protective lining of the airways while preserving some immune cells that can continue to produce signals. The result is a lung environment with more inflammation, more cell debris, and more opportunities for new virus particles to escape and infect neighboring cells.
What this means for future treatments
To a non-specialist, the key message is that RSV does not rely only on direct damage from viral invasion. By releasing large amounts of soluble G protein, it can "soften up" nearby cells through TLR2, making them more likely to die in an inflammatory way and to spill virus. This work identifies several steps in that chain—soluble G itself, its CX3C motif, TLR2, and the NLRP3 inflammasome—as potential drug targets. In principle, therapies that block the interaction between soluble G and TLR2, or that dampen NLRP3 activity, could reduce both lung injury and viral spread. Such approaches might one day complement existing RSV vaccines and antibodies, offering better protection for the youngest and most vulnerable patients.
Citation: Meineke, R., Agac, A., Knittler, MC. et al. The soluble G protein of respiratory syncytial virus promotes viral dissemination via TLR2-mediated NLRP3 priming and pyroptosis. npj Viruses 4, 6 (2026). https://doi.org/10.1038/s44298-026-00172-x
Keywords: respiratory syncytial virus, soluble G protein, TLR2 inflammasome, pyroptosis, lung inflammation