Clear Sky Science · en
Machine learning-accelerated CALPHAD analysis of impurity-driven intermetallic formation in secondary AlSi7Mg0.3
Why Scrap Aluminum Still Matters
Aluminum cans, car parts, and window frames can be melted down again and again with only a fraction of the energy needed to make new metal. But recycled aluminum comes with hitchhikers: trace amounts of other metals like iron, manganese, and copper. These tiny impurities can rearrange themselves into hard, brittle particles inside the solid metal, quietly undermining strength and corrosion resistance. This study shows how pairing advanced thermodynamic modeling with machine learning can turn that messy reality into a practical map for making safer, stronger recycled aluminum alloys.
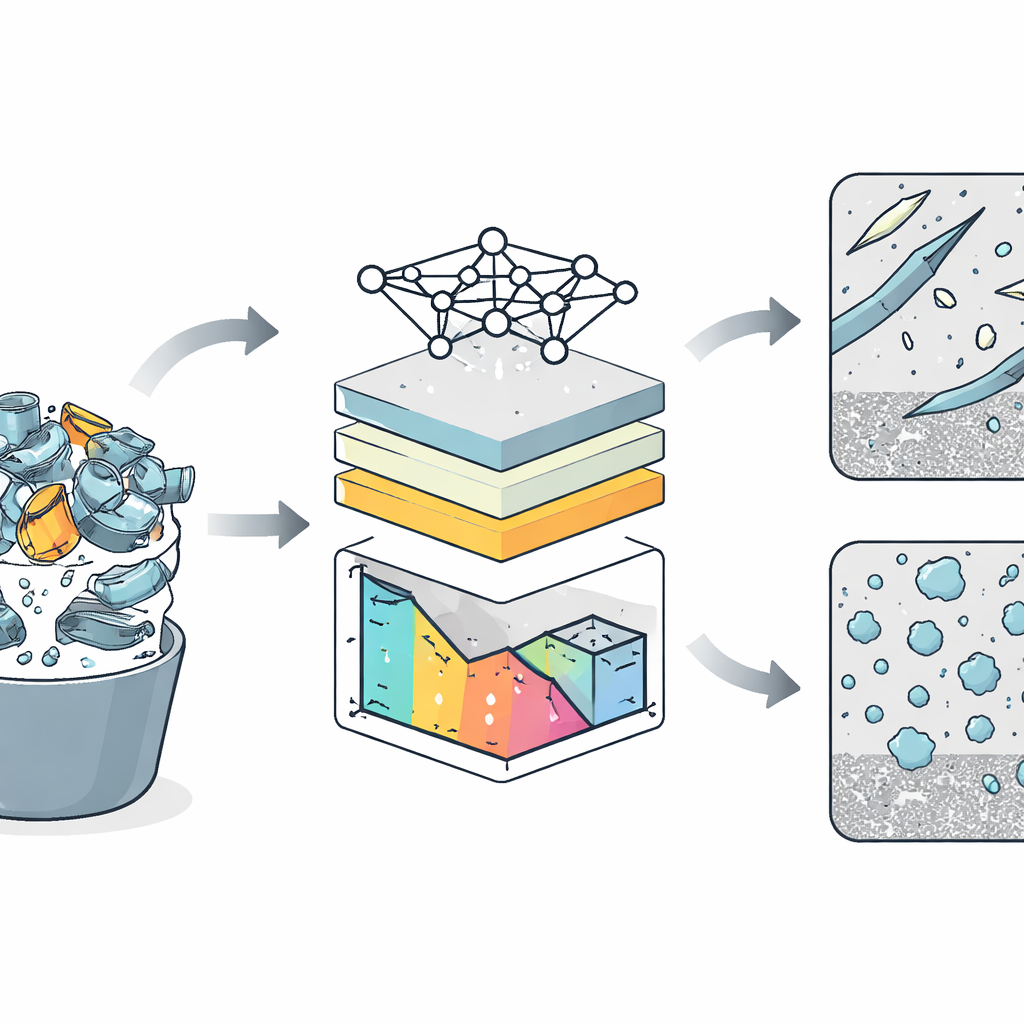
From Dirty Scrap to Clean Design
Recycling aluminum saves enormous amounts of energy and carbon emissions compared with smelting fresh metal from ore. The catch is that scrap streams are chemically messy. Elements such as iron and copper are hard to remove once they are in the melt, and even tenths of a percent can promote the formation of intermetallic particles—microscopic compounds that are much harder and more brittle than the surrounding aluminum. In common casting alloys used for engine blocks and structural parts, one such compound, a plate-like iron–silicon phase often seen as long needles in microscope images, is notorious for triggering cracks, porosity, and corrosion. Industry has learned that adding manganese can partly tame this problem by steering iron into a less harmful “Chinese-script” phase with a more rounded shape, but the safe window of impurity levels remained poorly charted.
Simulating Thousands of Alloys on a Computer
To tackle this challenge without casting and testing an impossible number of samples, the authors relied on a well-established thermodynamic framework called CALPHAD, which predicts what phases will form as an alloy solidifies. They focused on a widely used casting alloy, AlSi7Mg0.3, and systematically varied the levels of three common impurities—iron, manganese, and copper—over realistic ranges. Using specialized software, they simulated the solidification of 4,999 different compositions, recording how much of each important phase formed in each virtual alloy. These computer-generated data became the training ground for a machine learning model, specifically a Random Forest, which learned to predict phase amounts directly from impurity contents.
Teaching a Model to Read the Metal
Once trained and carefully validated, the model reproduced the thermodynamic calculations with high accuracy, but at a tiny fraction of the computational cost. That speed-up allowed the researchers to scan more than 20 million hypothetical alloys within the same impurity ranges. To understand not just what the model predicted but why, they used a method known as SHAP analysis, which attributes changes in the model’s output to individual inputs. This revealed clear patterns: iron strongly stabilized the harmful needle-like phase and weakened the manganese-rich phase, while manganese did the opposite. Copper, in contrast, mainly influenced copper- and magnesium-containing phases and only subtly nudged the iron–manganese balance, often competing with magnesium to form its own compounds.
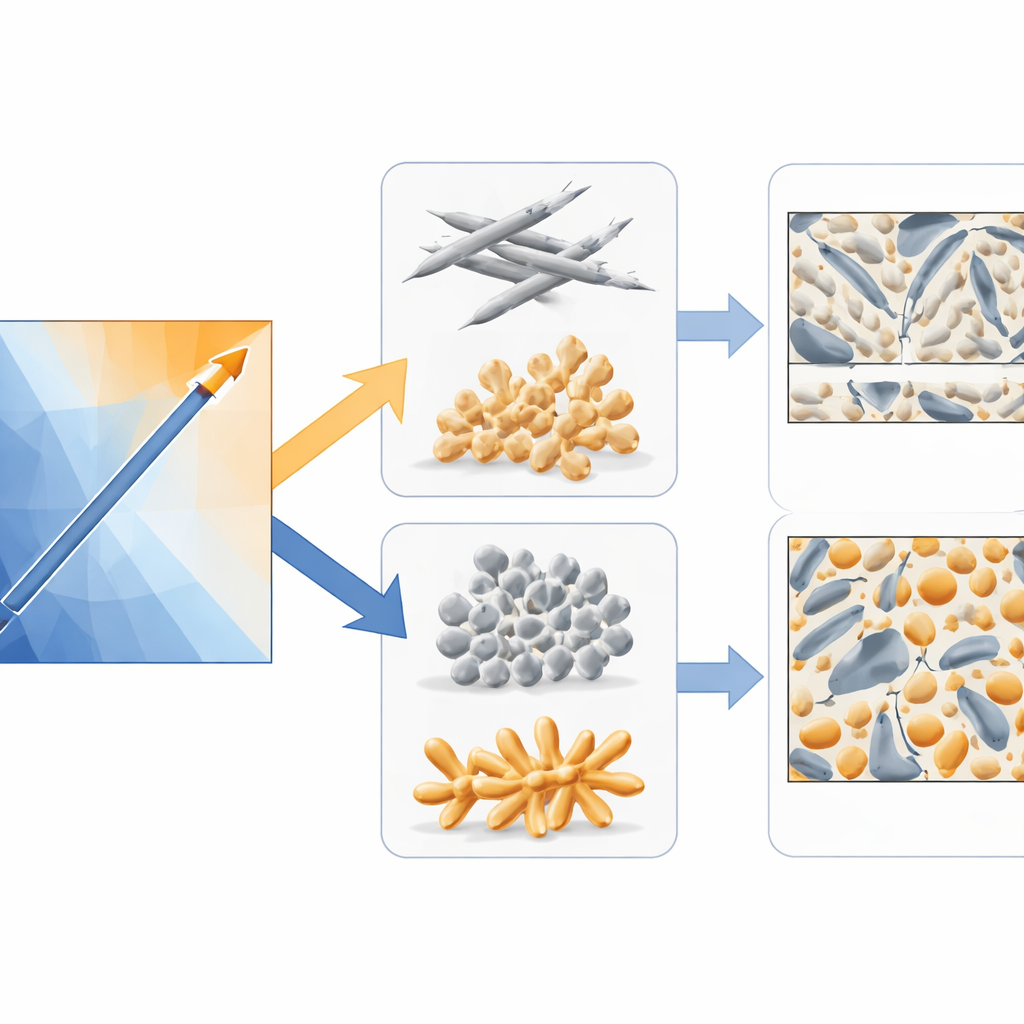
Drawing Maps for Alloy Makers
With millions of model evaluations in hand, the team could draw smooth “impurity maps” that showed, for any combination of iron and manganese, how much of each key phase is expected to form. These maps confirmed long-suspected trends and added new quantitative detail. Raising the manganese-to-iron ratio steadily shifted material away from the brittle needle-like phase toward the less damaging script-like phase. Notably, when this ratio exceeded about two—higher than typical industrial practice—the harmful phase was strongly suppressed for iron contents up to about one percent, without increasing the overall amount of iron-bearing particles. At the same time, the maps highlighted that simply adding more manganese is not a free lunch: at very high levels, it may hurt mechanical performance, a reminder that thermodynamic predictions must be balanced with processing and property data.
What This Means for Better Recycled Metal
In everyday terms, this work turns the vague notion of “dirty” recycled aluminum into a set of navigable roadmaps. By combining physics-based simulations with machine learning, the authors can quickly estimate how different mixes of scrap—and deliberate additions of manganese—will reshape the internal architecture of an Al–Si casting alloy. Their approach does not fix recycling challenges on its own, but it offers a powerful planning tool: foundries can use it to set impurity limits, choose scrap blends, and fine-tune alloy recipes that tolerate more recycled content while keeping dangerous needle-like particles in check. The same strategy can be adapted to other alloy families, helping push metal production toward a more energy-efficient, low-carbon future without sacrificing reliability.
Citation: Jarren, L.C., Viardin, A., Gazenbiller, E. et al. Machine learning-accelerated CALPHAD analysis of impurity-driven intermetallic formation in secondary AlSi7Mg0.3. npj Mater. Sustain. 4, 11 (2026). https://doi.org/10.1038/s44296-026-00097-9
Keywords: recycled aluminum alloys, metal impurities, machine learning in materials, thermodynamic simulations, intermetallic phases