Clear Sky Science · en
Environmental performance of bioplastics: degradation pathways, chemical leaching, and life-cycle implications
Why “green” plastics matter to everyday life
Shopping bags, coffee cups, and food wrappers made from bioplastics are sold as eco‑friendly answers to our plastic waste crisis. But are they truly better for people and the planet, or do they simply change the kind of pollution we face? This review pulls together the latest science to show how bioplastics break down, what chemicals they release, and how they compare with conventional plastics over their entire life—from crop or waste feedstock to disposal. The findings reveal a more nuanced picture than the simple “good plastic, bad plastic” story we often hear.
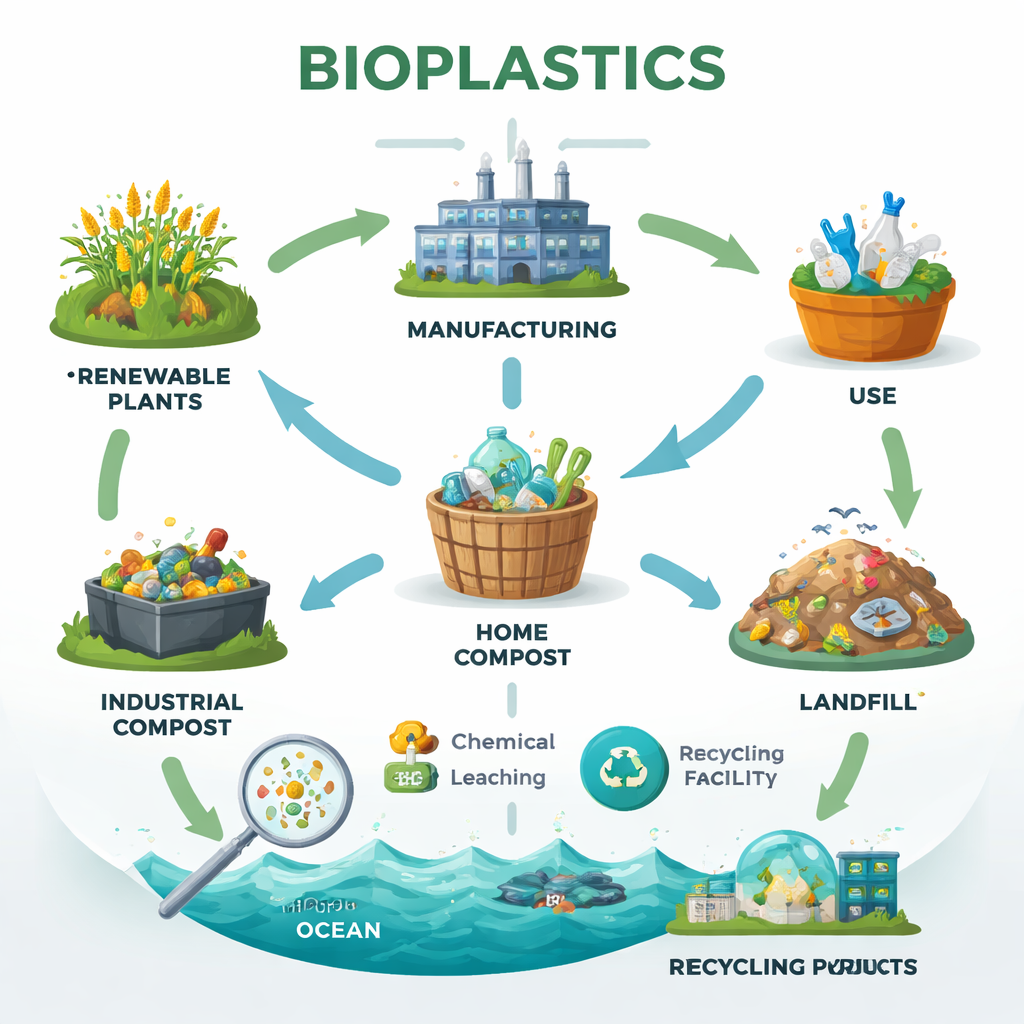
What makes a plastic “bio” or “compostable”?
Bioplastics are not all the same, and the terms used to market them can be confusing. “Bio‑based” simply means the material comes partly or fully from renewable sources such as corn, sugarcane, or plant oils; it says nothing about how it behaves in nature. Some bio‑based plastics, like bio‑polyethylene, act much like ordinary plastics and do not readily break down. “Biodegradable” plastics can, in principle, be eaten away by microbes into carbon dioxide, water, and biomass—but usually only under specific conditions of heat, moisture, and oxygen. “Compostable” plastics are a subset designed to disintegrate and biodegrade in controlled composting facilities, often at temperatures around 58 °C. Many products marketed as compostable will degrade quickly only in industrial plants, not in a backyard pile or the open environment, so matching the material to the right waste system is crucial.
How bioplastics break apart into tiny pieces
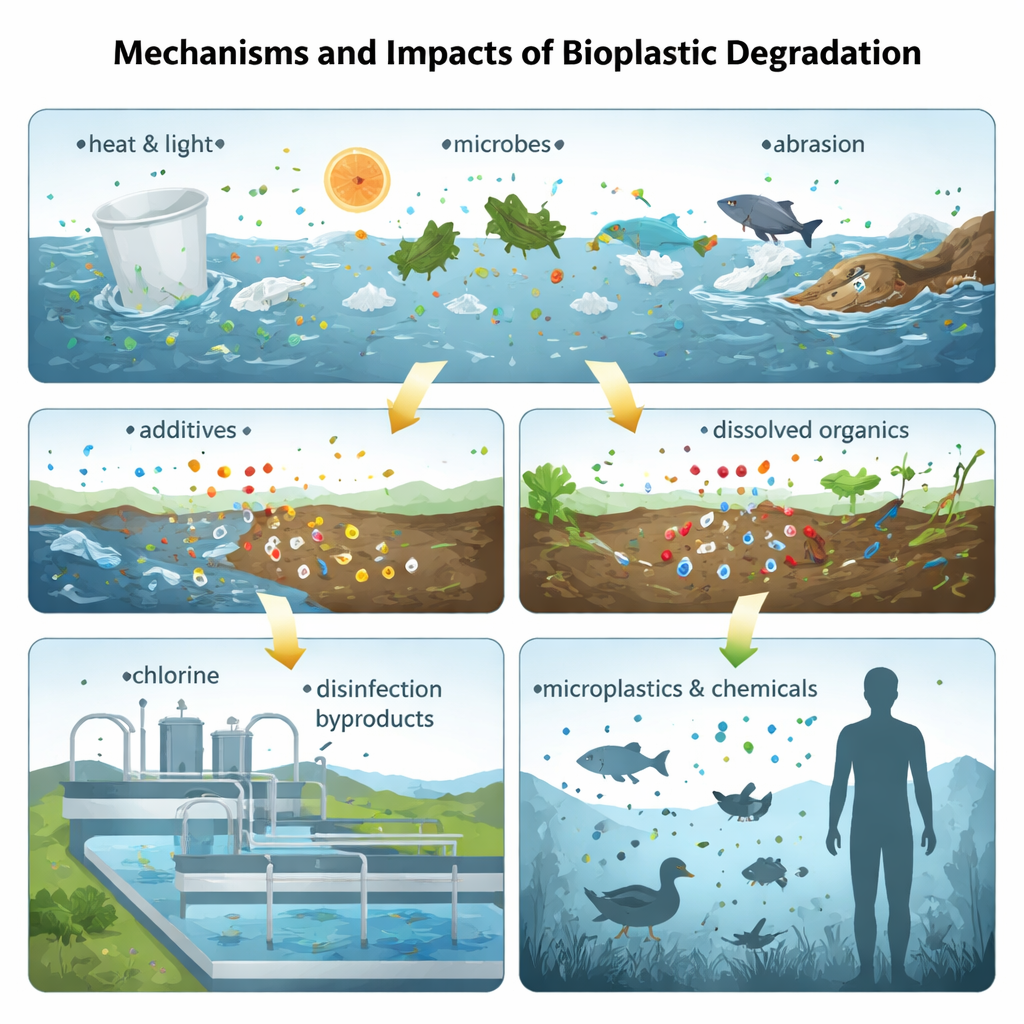
Once discarded, bioplastics face sun, heat, water, and physical wear, just like conventional plastics. These forces, along with microbes, gradually crack and weaken the material, generating ever smaller fragments known as microplastics and nanoplastics. Studies show that popular bioplastics such as polylactic acid (PLA) and certain polyester blends can shed large numbers of particles under ultraviolet light and mechanical stress, sometimes more rapidly than traditional plastics. In soils and sediments, however, even “biodegradable” plastics can linger for months to years, with degradation slowed by limited nutrients or low temperatures. As they fragment, their surface chemistry changes, which can make them more reactive and more likely to carry other pollutants or microorganisms.
Chemicals that leak out as bioplastics age
Bioplastics are not pure, natural substances; they contain plasticizers, stabilizers, fillers, and colorants much like fossil‑based plastics. When exposed to water, heat, or abrasion, these additives and small pieces of the polymer itself can leach into surrounding air, soil, and water. Analyses of everyday items made from PLA, polyhydroxyalkanoates (PHA), starch blends, and plant‑fiber composites have detected thousands of distinct chemical features, including phthalates, bisphenol A, metal compounds, and other molecules whose health effects are poorly understood. Laboratory tests find that leachates from some bioplastics can shorten the lifespan and impair movement of tiny worms, harm sea urchin embryos and mussel larvae, stress photosynthetic microbes, and disturb the lungs and livers of test animals exposed to airborne particles. In short, switching to a bio‑label does not automatically remove toxicity concerns; it changes the mixture of chemicals we must evaluate.
Hidden risks in drinking water and climate impacts
Another emerging issue is what happens when bioplastic‑derived organic matter meets disinfectants in water treatment plants. When compounds released from materials like PLA react with chlorine, they can form regulated disinfection byproducts—such as trichloromethane and various haloacetic acids—at levels comparable to or higher than those formed from aged conventional plastic particles under similar conditions. These byproducts are linked to cancer and reproductive problems, yet regulations typically focus on natural organic matter, not microplastics. At the same time, life‑cycle assessments show that bioplastics can significantly cut climate‑warming emissions when they are made from agricultural residues, food waste, or industrial by‑products, and when end‑of‑life pathways like efficient recycling or well‑managed composting are available. If made from dedicated crops with heavy fertilizer use, or sent to landfills or incinerators without energy recovery, their climate advantage can shrink or even vanish.
Building a truly sustainable future for plastics
The overall message of this review is that bioplastics can help reduce dependence on fossil fuels, but they are not a silver bullet. In the wrong conditions, they can still generate persistent microplastics, leak complex chemical cocktails, and contribute to harmful byproducts in drinking water. To make them genuinely safer, manufacturers must design materials for recycling and composting, use fewer hazardous additives, and rely more on waste‑derived feedstocks. Researchers need standardized tests that link degradation, leaching, and toxicity to real‑world scenarios, while policymakers must align labels and collection systems so that “compostable” or “biodegradable” claims reflect what actually happens after use. Only when design, science, and infrastructure move together can bioplastics live up to their promise as cleaner, lower‑impact materials rather than simply a new form of plastic pollution.
Citation: Shanmugam, V., Kaynak, E., Das, O. et al. Environmental performance of bioplastics: degradation pathways, chemical leaching, and life-cycle implications. npj Mater. Sustain. 4, 9 (2026). https://doi.org/10.1038/s44296-026-00096-w
Keywords: bioplastics, microplastics, chemical leaching, disinfection byproducts, life cycle assessment