Clear Sky Science · en
Graphene oxide as smart sustainable nanomaterial: a versatile multifunctional material with transformative potential in advanced materials science research
Why a two-dimensional powder matters for everyday life
From clean drinking water to longer-lasting phone batteries and safer medicines, many of today’s biggest challenges boil down to how well we control materials at very small scales. This article reviews graphene oxide, a sheet-like form of carbon only one atom thick, and explains how it could become a key "smart material" for tackling pollution, improving energy storage, and advancing healthcare — all while aiming to be more sustainable than many current technologies.
A thin carbon sheet with a surprising personality
Graphene oxide (GO) starts from graphite, the same carbon found in pencil leads, but is chemically treated so that oxygen-containing groups attach to its surface. This turns an ultra-flat carbon sheet into a kind of molecular Velcro: one side remains largely carbon, good for conducting electricity and stacking, while scattered oxygen groups make it strongly interact with water, metals, and many organic molecules. The review explains how classic chemical methods, newer electrochemical routes, and “green” syntheses from waste biomass (such as pine needles, tea waste, or coconut shells) all produce GO with slightly different amounts and patterns of oxygen. Those subtle differences – in layer spacing, surface charge, and defect density – end up controlling how well GO adsorbs pollutants, conducts charge, or withstands repeated use.
Designing a smart surface: tunable chemistry and shape
Because its surface is dotted with reactive oxygen sites, GO can be “programmed” by adding or swapping chemical groups. Researchers attach amines, gels, dyes, polymers, and even drug-like molecules to make GO more selective for heavy metals, specific dyes, or biological targets. The paper shows that these modifications can strongly boost water purification, sensing, or antibacterial performance by combining several weak forces – such as electrostatic attraction, hydrogen bonding, and stacking between flat aromatic rings. GO is also not confined to a single shape: it can be cut into zero-dimensional quantum dots that glow under light, spun into one-dimensional fibers, stacked into two-dimensional membranes, or assembled into three-dimensional aerogels. Each form offers a different balance of strength, porosity, and transport pathways, expanding the range of technologies GO can plug into.
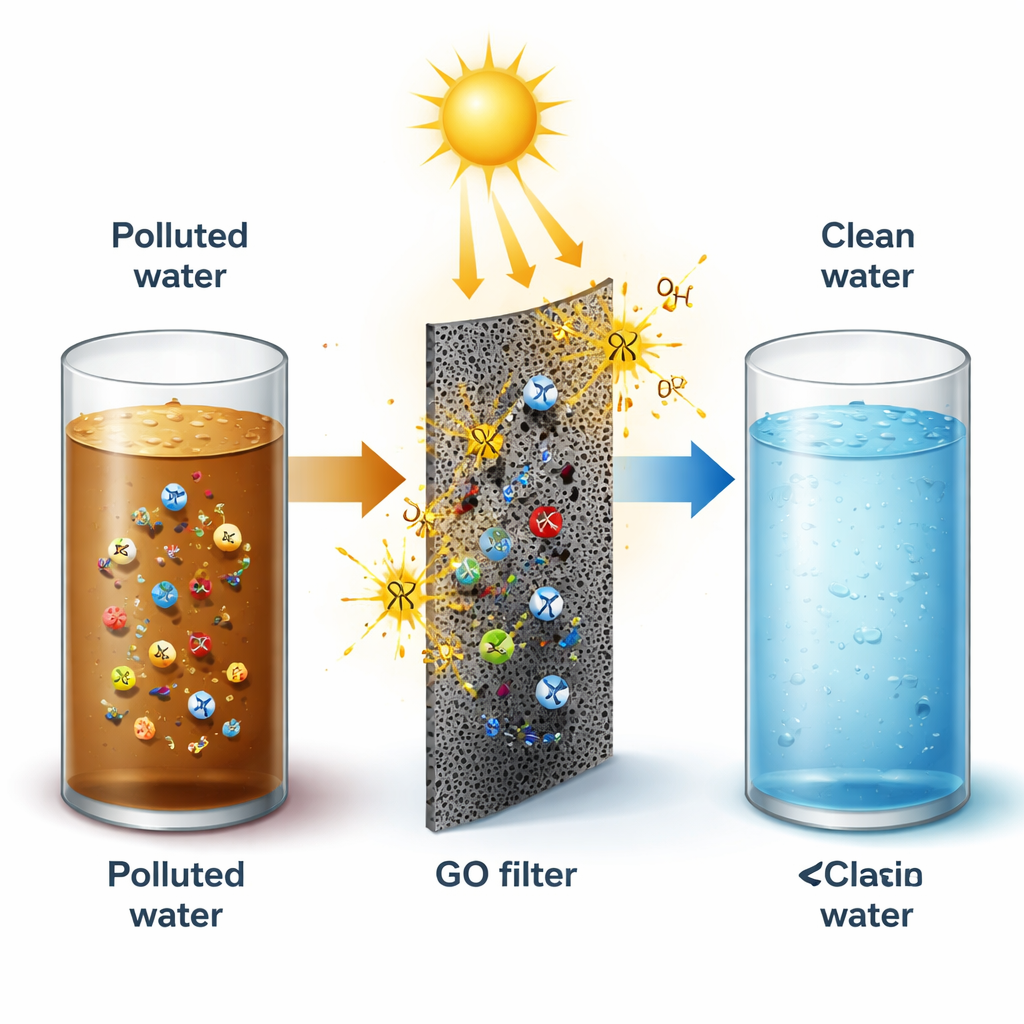
Cleaning water, converting sunlight, and storing energy
A major theme of the article is GO’s role in environmental cleanup. As an adsorbent, powdered GO can capture many pollutants from water — including bright industrial dyes, antibiotic molecules, heavy metals like lead and chromium, and even tiny plastic particles — often with very high capacities and the possibility of partial reuse. Under light, GO can also act as a photocatalyst: when it absorbs photons, electrons and “holes” form and trigger reactive species that break down stubborn chemicals such as pesticides, drug residues, and microplastics. Beyond cleanup, GO shows promise in solar-driven conversion of carbon dioxide into fuels like methanol, and in hydrogen production from water when paired with suitable dopants or co-catalysts. In batteries, GO and its reduced form help build more robust electrodes and sulfur hosts, stabilizing lithium- and sodium-based cells so they can charge faster and last longer.
From lab bench to clinic and hospital ward
The review also explores how GO is being adapted for health-related uses. Its flat, aromatic surface can hold large amounts of anticancer drugs, releasing them in response to pH or light, and its ability to quench or enhance fluorescence makes it useful in biosensors that detect DNA, pathogens, or trace chemicals. GO and reduced GO show notable antibacterial action, physically damaging bacterial membranes, binding key cell-wall components, and generating reactive oxygen species under light. At carefully chosen sizes and surface coatings, these same traits can support bioimaging and tissue engineering. However, the authors stress that biocompatibility is highly sensitive to sheet size, dose, and surface chemistry, and that long-term toxicity and environmental fate must be much better understood before medical and consumer applications are widely deployed.
Promise, pitfalls, and the road to real-world impact
Despite its versatility, graphene oxide is not a magic bullet. The paper highlights practical hurdles: dry GO tends to clump, cutting its effective surface area; photocatalytic efficiency can be modest unless GO is carefully engineered; and regeneration after multiple purification cycles often degrades performance. Large-scale production is still costly, energy intensive, and reliant on strong acids, although greener electrochemical and biomass-based methods are advancing. There are also unresolved questions about worker safety and what happens if GO escapes into the environment. Even so, by showing how one tunable material can link water treatment, clean energy, sensing, and healthcare, the review argues that graphene oxide is a powerful test case for building technologies that are both high-performing and more sustainable — provided that scale-up, safety, and life-cycle impacts are addressed head-on.
Citation: Thakur, S., Badoni, A., Sharma, R. et al. Graphene oxide as smart sustainable nanomaterial: a versatile multifunctional material with transformative potential in advanced materials science research. npj Mater. Sustain. 4, 8 (2026). https://doi.org/10.1038/s44296-026-00095-x
Keywords: graphene oxide, water purification, photocatalysis, energy storage, nanomaterials