Clear Sky Science · en
Catalytic conversion of cellulose and its derived sugars to 5-Hydroxymethylfurfural, levulinate esters, and sorbitol: a comprehensive review
Turning Plant Waste into Useful Everyday Ingredients
Every year, farms and forests leave behind mountains of plant leftovers—stalks, sawdust, and other scraps rich in cellulose, a tough natural fiber. Instead of burning or discarding this material, scientists are learning how to turn it into valuable chemicals that can go into cleaner fuels, plastics, batteries, food, and medicines. This article reviews how researchers are transforming cellulose into three especially important products—5‑HMF, levulinate esters, and sorbitol—and what it will take to make these green processes work at industrial scale.
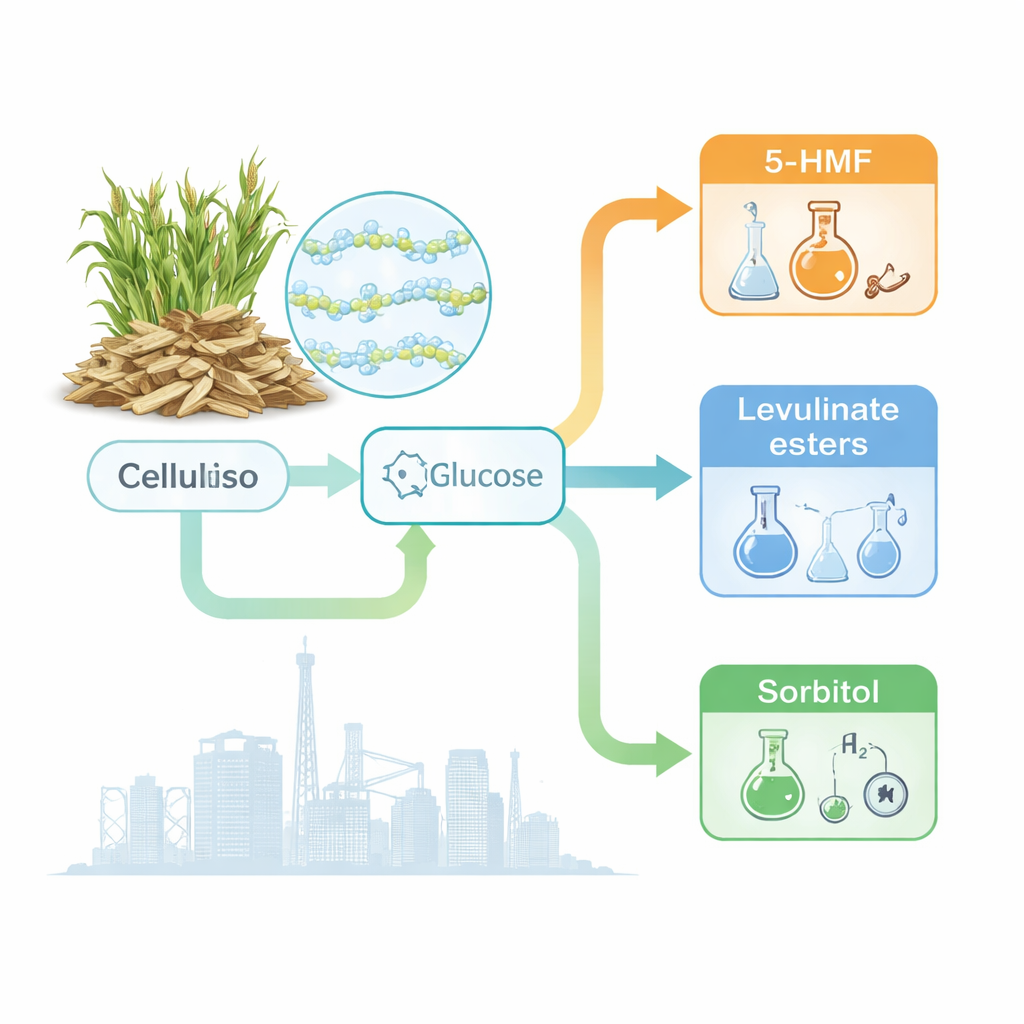
From Wood Fiber to Versatile Building Blocks
Cellulose is the main structural component of plants, made of long chains of sugar units tightly packed together. That tight packing, held by a dense web of hydrogen bonds, makes cellulose strong—and very hard to dissolve or react with. The review explains how scientists first break cellulose down into glucose, the familiar simple sugar, and then steer it into different products. One route dehydrates glucose into 5‑HMF, a highly adaptable building block for bio‑based plastics, fuels, and specialty chemicals. Another route reshapes related molecules into levulinate esters, promising fuel additives with high energy content and cleaner burning. A third path hydrogenates glucose into sorbitol, a sugar alcohol widely used as a sweetener and as a starting point for medicines and advanced materials.
Designing Liquids That Tame Stubborn Cellulose
Because cellulose resists ordinary solvents like water or alcohol, much of the innovation has focused on the reaction medium. The review compares four main solvent strategies for making 5‑HMF from cellulose. Single‑phase systems (one uniform liquid) are simple but often give modest yields and unwanted byproducts. Biphasic systems use two immiscible liquids: one where cellulose reacts, and another that continually pulls out fragile 5‑HMF before it can degrade, sharply improving yield and easing separation. Ionic liquids—salts that are liquid at room temperature—can dissolve cellulose directly by disrupting its hydrogen‑bond network and can even double as catalysts, but they are expensive, viscous, and hard to recycle. Deep eutectic solvents, made by combining cheap, often bio‑derived components, mimic many advantages of ionic liquids at lower cost and toxicity, but they are still new and not yet fully optimized. 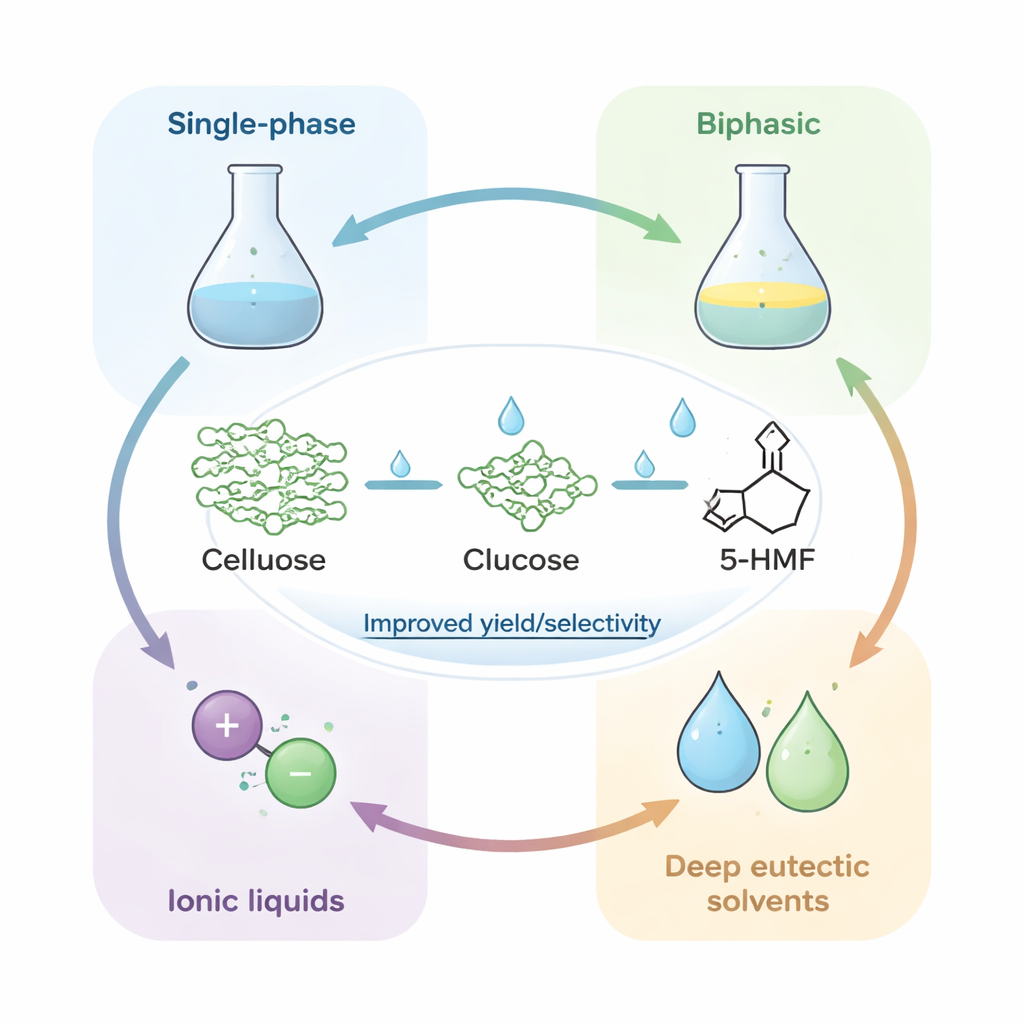
Crafting Solid Catalysts for Cleaner Fuels and Sweet Alcohols
Levulinate esters and sorbitol both rely heavily on carefully designed catalysts—the “traffic cops” that guide reactions along preferred routes. For levulinate esters, researchers have explored three approaches: reacting ready‑made levulinic acid with alcohols; converting furfuryl alcohol, itself derived from plant sugars; and direct “one‑pot” conversion of cellulose. Using solid acids such as zeolites, polyoxometalates, and functionalized carbons allows the catalyst to be filtered out and reused, avoiding the corrosion and waste associated with strong liquid acids. For sorbitol, metals like nickel and ruthenium supported on porous materials are central. These catalysts must strike a balance: enough acidity to open up cellulose and form glucose, and enough hydrogenation power to immediately turn that glucose into thermally stable sorbitol before it breaks down into other products. The review shows that carefully tuned non‑precious metal catalysts can approach the performance of noble metals, promising lower costs.
Simulating Reactions Atom by Atom
Beyond lab experiments, computer modeling has become a powerful ally in this field. Methods such as density functional theory, molecular dynamics, and thermodynamic screening help researchers see how cellulose chains, solvents, and catalysts interact at the atomic level. These tools have been used to sift through thousands of potential ionic liquids, revealing which ones dissolve cellulose best, and to map detailed step‑by‑step pathways from glucose to 5‑HMF. Simulations also expose how side reactions form tarlike “humins” that waste carbon and foul reactors. Looking ahead, the authors argue that combining such models with machine learning can accelerate the search for better solvents and catalysts, cutting down on trial‑and‑error work in the lab.
Bringing Green Chemistry to Real‑World Scale
The review concludes that turning cellulose into 5‑HMF, levulinate esters, and sorbitol could support cleaner fuels, lighter and greener plastics, and more sustainable food and drug ingredients. However, several hurdles remain: cellulose’s natural resistance, the cost and recyclability of advanced solvents and catalysts, and the need to limit energy use and waste. The authors call for “one‑pot” processes that combine steps, multifunctional catalysts that can both break down cellulose and upgrade the resulting sugars, and integrated biorefineries that co‑produce several products from the same feedstock. With help from modern computation and careful economic and environmental assessments, these technologies could turn low‑value plant leftovers into a cornerstone of a circular, bio‑based economy.
Citation: Huang, K., Song, J., Su, K. et al. Catalytic conversion of cellulose and its derived sugars to 5-Hydroxymethylfurfural, levulinate esters, and sorbitol: a comprehensive review. npj Mater. Sustain. 4, 7 (2026). https://doi.org/10.1038/s44296-025-00091-7
Keywords: cellulose valorization, 5-hydroxymethylfurfural, levulinate esters, sorbitol production, green solvents