Clear Sky Science · en
Menopausal hormone therapy and risk of neuropsychiatric disease: a drug target Mendelian randomisation study
Why this research matters
Many women wonder whether taking hormones around menopause will protect their brains or put them at risk for problems like memory loss, depression, or anxiety. Doctors, too, are divided, because past studies have produced conflicting answers. This article uses a genetics-based approach, similar in spirit to a long‑term randomized trial, to ask a simple question: when the brain’s estrogen receptors are nudged in the way menopausal hormone therapy might nudge them, does that change the risk of Alzheimer’s disease or common mental health problems?
The puzzle around hormones and the brain
Women are more likely than men to experience depression, anxiety, and Alzheimer’s disease. One leading idea is that estrogen, a key female sex hormone, helps shape the brain across life and may protect it—until levels fluctuate or fall at midlife. During the menopausal transition, many women use menopausal hormone therapy (MHT) to ease symptoms such as hot flashes and sleep problems. Yet studies on whether MHT helps or harms brain health have disagreed: some found higher dementia risk, others suggested protection, and many showed no clear effect at all. These inconsistencies may stem from short trial durations, different drug formulations, and the fact that women with worse symptoms are more likely to be prescribed hormones in the first place.
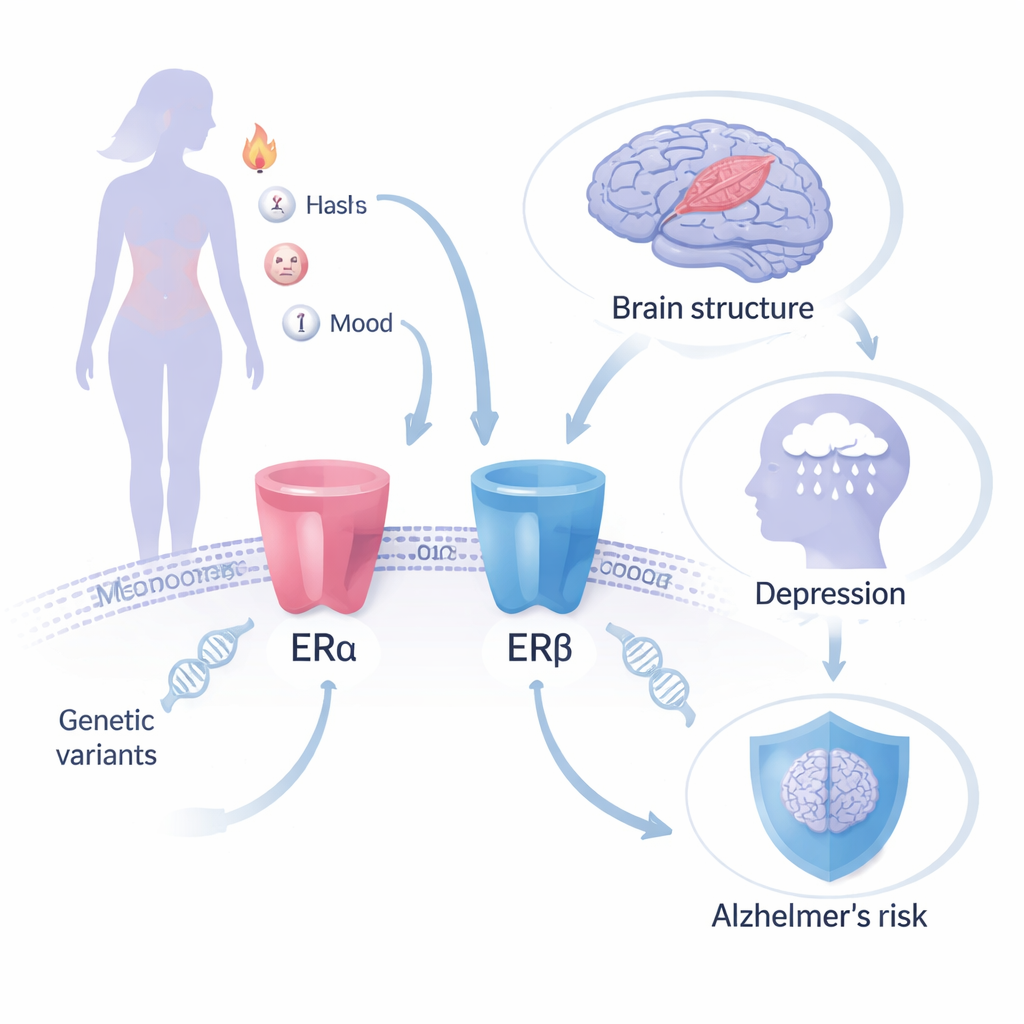
Using genes as a natural experiment
Instead of following women taking hormone pills, the researchers turned to Mendelian randomisation, a method that uses naturally occurring genetic differences as a stand‑in for a lifelong, randomly assigned “treatment.” They focused on two proteins in the body called estrogen receptors—ERα and ERβ—that MHT is designed to activate. Specific genetic variants in the genes encoding these receptors (ESR1 and ESR2) have known effects on “downstream” traits that change when estrogen acts, such as bone mineral density, a blood protein that carries sex hormones, and hemoglobin levels. By tracking how these receptor‑linked variants relate to large genetic datasets on Alzheimer’s disease, brain structure, depression, and anxiety, the team could estimate how tweaking each receptor might influence long‑term brain and mental health risks.
What the genetic evidence showed
The authors built three genetic instruments: two capturing ERα activity (via variants linked to bone density and to sex hormone‑binding globulin) and one capturing ERβ activity (via a variant linked to hemoglobin). They then combined these with some of the largest available genome‑wide association studies of Alzheimer’s disease, brain MRI measures (overall grey matter, hippocampal volume, and white matter “spots” linked to aging), depression, and anxiety. Overall, they found no convincing evidence that genetically simulated changes in either estrogen receptor meaningfully altered the risk of Alzheimer’s disease or affected these brain structure measures. A few weak signals for changes in grey matter or hippocampal volume did not survive statistical correction, suggesting they may be due to chance rather than a true biological effect.
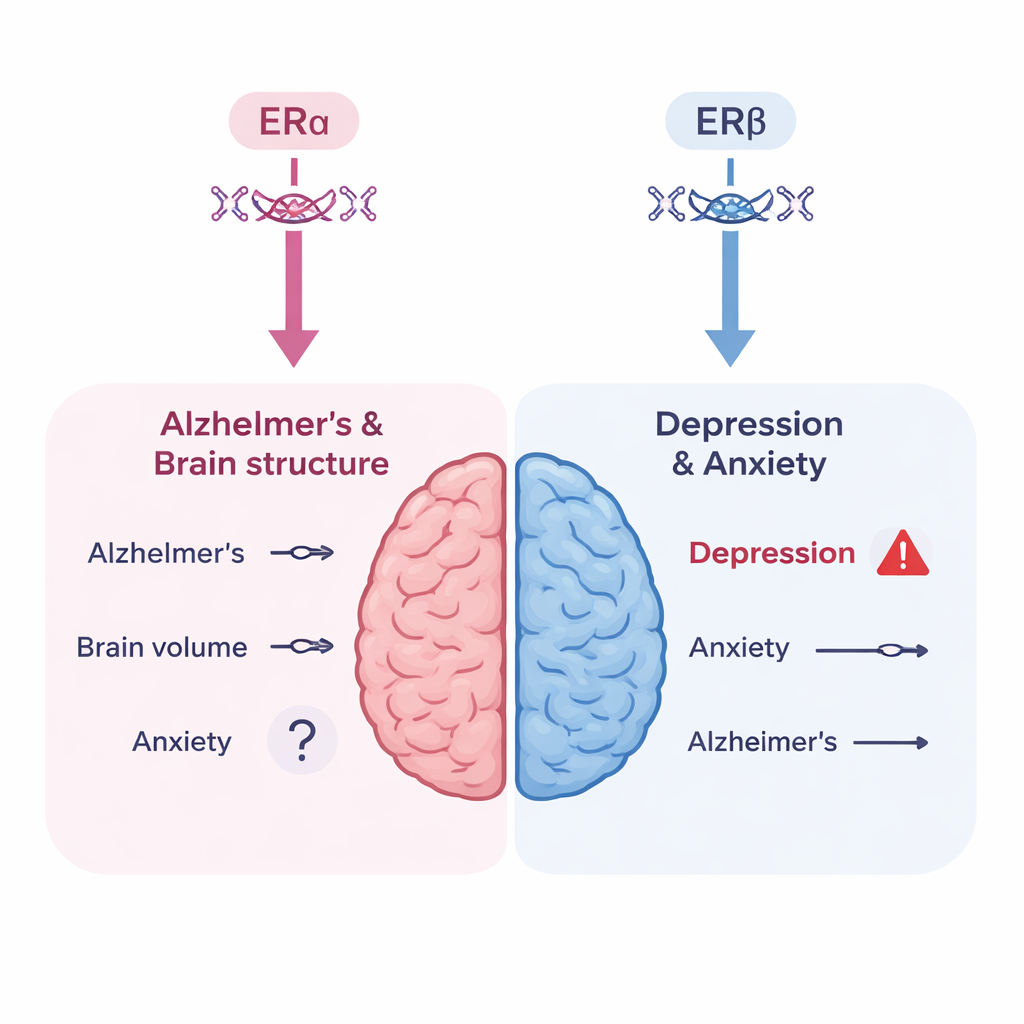
A warning sign for depression risk
The story was different for mood. When the researchers looked at ERβ activity, as captured by the hemoglobin‑based genetic proxy, they observed a statistically robust link with a higher risk of depression. This finding fits with what is known about where ERβ is most active in the brain—regions such as the hippocampus and thalamus that are heavily involved in mood regulation. However, the authors caution that the hemoglobin proxy is imperfect: low hemoglobin itself may contribute to fatigue and low mood, which could blur the picture. They found no strong genetic evidence that ERα activity on its own raises depression risk, and only a suggestive, non‑definitive hint that ERα might be related to anxiety. Importantly, genetic effects that operate steadily from birth are not identical to starting hormone therapy at midlife, so these results should not be read as a direct prediction of any specific MHT regimen.
What this means for women and their doctors
Translated for everyday decisions, this study suggests that targeting estrogen receptors in ways similar to MHT does not clearly increase or decrease lifetime risk of Alzheimer’s disease or cause major changes in brain structure, at least in people of European ancestry. At the same time, the result pointing to ERβ and depression underscores that hormone‑sensitive brain circuits are tied to mood, and that different estrogen receptors may carry different mental health implications. The work does not replace clinical trials, but it offers a powerful genetic “reality check” that can help refine future hormone therapies and guide research on safer, more tailored treatments for women navigating menopause.
Citation: Schindler, L.S., Gill, D., Oppenheimer, H. et al. Menopausal hormone therapy and risk of neuropsychiatric disease: a drug target Mendelian randomisation study. npj Womens Health 4, 10 (2026). https://doi.org/10.1038/s44294-026-00130-1
Keywords: menopausal hormone therapy, estrogen receptors, Alzheimer’s disease, depression risk, Mendelian randomisation