Clear Sky Science · en
A comprehensive genomic framework for identifying genes predisposing to homologous recombination repair-deficient breast or ovarian cancer
Why some families face higher cancer risks
Many women with breast or ovarian cancer have a strong family history of the disease, yet genetic testing often finds no clear explanation. This gap, sometimes called “missing heritability,” leaves families without answers and can limit access to tailored screening or treatments like targeted drugs. This study set out to build a new, more powerful way to search our DNA for hidden cancer‑risk genes by combining several kinds of genomic and clinical information rather than looking at each in isolation. 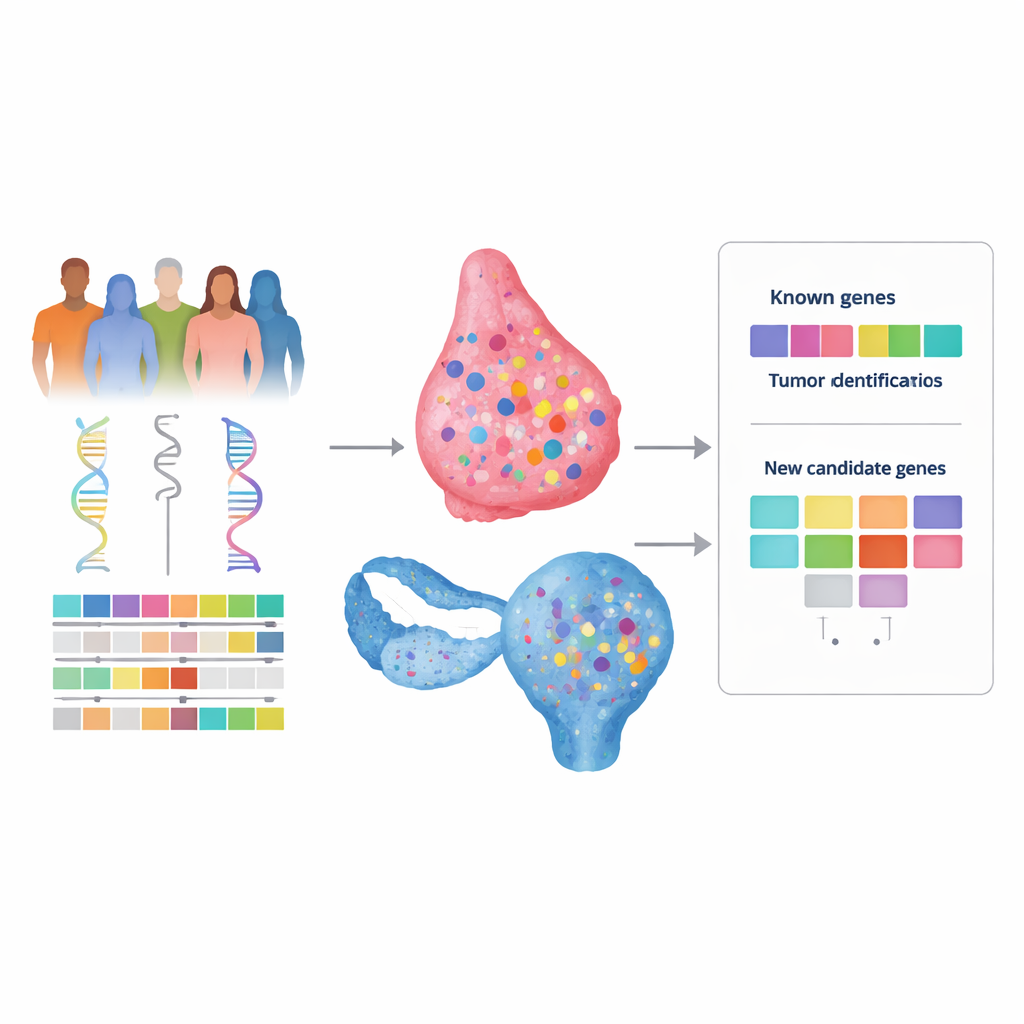
Looking for a fingerprint of faulty DNA repair
The researchers focused on a particular kind of DNA damage repair, known as homologous recombination repair, that normally fixes dangerous breaks in our genetic material. When this repair system fails, cells accumulate a telltale “mutational signature” – a characteristic pattern of changes in the tumor’s DNA that acts like a molecular fingerprint. This pattern is especially common in certain hard‑to‑treat breast and ovarian cancers. The team reasoned that if a tumor shows this fingerprint, it likely lost both working copies of some DNA repair gene, known or unknown, and that this could be traced back to inherited changes in that gene.
Building a combined genetic and clinical framework
To test this idea, the authors analyzed DNA from hundreds of breast and ovarian cancer patients in The Cancer Genome Atlas, which provides both blood (inherited) and tumor (acquired) genetic data, along with clinical information. They searched the entire exome – the protein‑coding part of the genome – for rare inherited variants that looked harmful and that also had a “second hit” in the tumor, such as loss of the remaining healthy copy. For each gene, they then asked whether such two‑hit events were more common in tumors with the repair‑defect signature than in those without it. Crucially, they did not restrict themselves to a preset panel of genes, allowing unexpected candidates to emerge.
Checking that the method works and finding new suspects
As a reality check, the framework correctly highlighted the well‑known breast and ovarian cancer genes BRCA1 and BRCA2 as strongly linked to the repair‑defect signature in both cancer types, confirming that the approach behaves as expected. In breast cancer, it also flagged an additional gene, THBS4, and suggested possible roles for genes called KIF13B and TESPA1. However, detailed case‑by‑case review showed that THBS4 changes often appeared alongside other, more established repair‑related events, making it a less convincing driver. 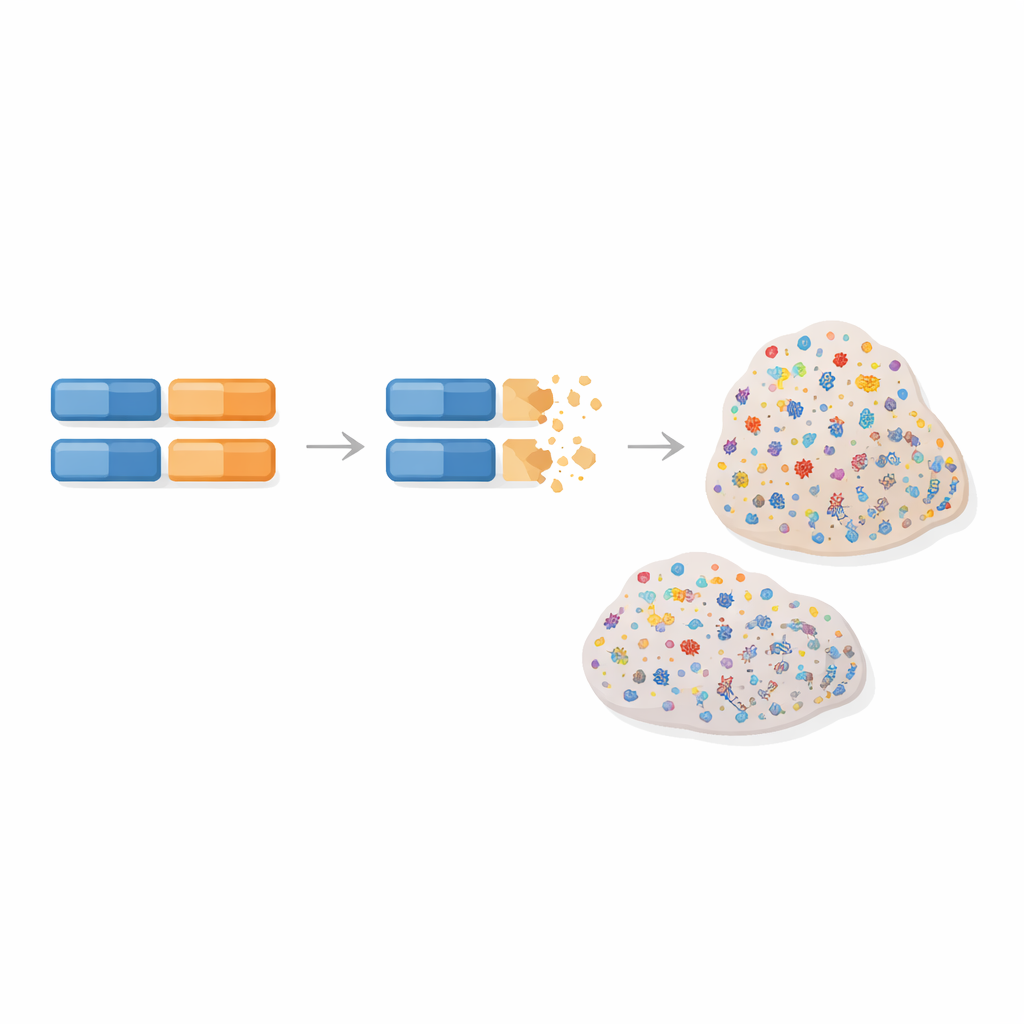
Zooming in on unexplained high‑risk tumors
To go beyond statistics, the researchers layered in clinical details such as tumor subtype, age at diagnosis, and ancestry. They focused on patients whose tumors clearly showed the repair‑defect signature and belonged to clinical groups already linked to this biology – basal‑like breast cancers and high‑grade serous ovarian cancers – yet lacked any known BRCA‑type events. In these patients, they looked again for inherited variants with second hits, this time within a broad, curated list of cancer and DNA repair genes. This “clinico‑genomics” view spotlighted several genes involved in DNA double‑strand break repair and the closely related Fanconi anemia pathway, including RAD51B, RAD54B, RAD54L, FANCD2, and others, as plausible new contributors to inherited risk.
What this means for patients and future research
The study does not claim to have definitively proven new cancer‑risk genes; the number of affected patients for each candidate gene is still small, and larger, more diverse cohorts will be needed to confirm their roles. Instead, the authors provide a reusable blueprint: a way to combine inherited and tumor DNA, characteristic mutational fingerprints, and clinical features to systematically prioritize genes that may underlie unexplained familial cancer. Over time, applying this framework to bigger datasets and other cancer types could shrink the “missing heritability” gap, refine genetic testing panels, and help more patients understand their personal risk and options for prevention and targeted treatment.
Citation: Camacho-Valenzuela, J., Matis, T., Roca, C. et al. A comprehensive genomic framework for identifying genes predisposing to homologous recombination repair-deficient breast or ovarian cancer. BJC Rep 4, 15 (2026). https://doi.org/10.1038/s44276-026-00218-w
Keywords: homologous recombination deficiency, breast cancer genetics, ovarian cancer, DNA repair genes, cancer susceptibility