Clear Sky Science · en
IL10RB expression in cancer cells is associated with evolutionary changes to solidify treatment resistance
Why Some Cancers Refuse to Stay Down
Cancer treatment has made huge strides, yet many tumors still come back, spread, or stop responding to drugs. This study explores why certain cancer cells become especially hard to kill and how a little-known surface molecule, called IL10RB, may act as a hidden switch that helps tumors resist both chemotherapy and modern immunotherapy—and how blocking it could tip the balance back in favor of the immune system.
A Shape-Shifting Step in Tumor Evolution
Many aggressive cancers go through a shape-shifting process known as an epithelial-to-mesenchymal transition. In simple terms, formerly well-behaved cells loosen their bonds, become more mobile, and take on traits linked to stem cells, which are hardier and more adaptable. The researchers focused on cancer cells driven into this state by a master control protein called Snail. They discovered that once Snail is active, it strongly boosts production of IL10RB on the surface of these cells, marking an evolutionary step toward a more stubborn, treatment-resistant tumor.
A Switch That Makes Cancer Tougher
To see what IL10RB actually does, the team altered its levels in mouse and human cancer cell lines. When they dialed IL10RB down, fast-growing, Snail-driven tumor cells lost some of their ability to multiply, cling to surfaces, and invade surrounding tissue. When they forced other cancer cells to make more IL10RB, the cells changed shape, sprouting tentacle-like protrusions used to dig through nearby tissue. These IL10RB-rich cells became more invasive, stuck more firmly to their environment, and were harder to kill with common chemotherapy drugs, even though they sometimes divided more slowly. At the genetic level, IL10RB overexpression switched on a broad program of changes tied to spread, survival, and drug resistance.
When Treatment Backfires on the Body
The story became even more troubling in live mouse experiments. Tumors loaded with IL10RB did not always grow the fastest, but they proved far more dangerous for the host. Standard chemotherapy that shrank ordinary tumors could actually worsen IL10RB-rich tumors, speeding their spread to distant organs. These tumors also distorted the animals’ immune systems. In and around the tumors, key killer immune cells became exhausted and overburdened, while in the spleen—the body’s immune reservoir—the numbers and function of helpful T cells and natural killer cells dropped sharply. The cancer was not just dodging drugs; it was quietly disarming the body’s defenses.
Turning the Tables with Targeted Antibodies
Because IL10RB is also part of normal immune signaling, there was a concern that targeting it might do more harm than good. To test this, the researchers developed antibodies that specifically block IL10RB. In multiple mouse cancer models, including bone and colon cancers with local tumors and widespread metastases, these antibodies shrank tumors and extended survival, often after only one or two doses. The benefit depended heavily on a particular type of immune cell: CD8-positive T cells, which specialize in killing abnormal cells. When these killer T cells were experimentally removed, the benefit vanished. Importantly, blocking IL10RB did not wipe out normal immune cells, but instead restored their ability to attack the cancer, and it worked across several different tumor types.
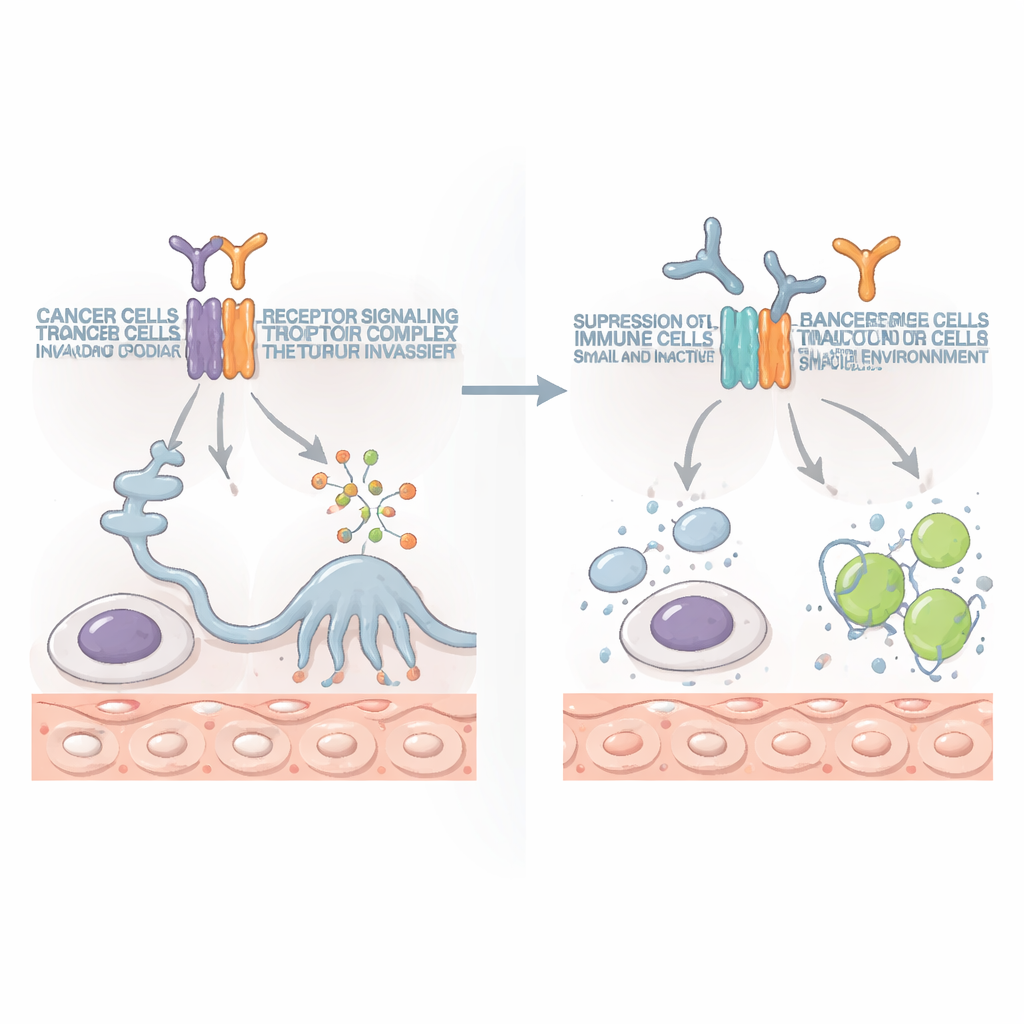
A New Partner for Modern Immunotherapy
Today, drugs that release immune “brakes,” such as PD-1 blockers, have transformed treatment for some cancers but help only a minority of patients. In this study, IL10RB-blocking antibodies on their own outperformed PD-1 blockade in certain models. When the two were combined, they reinforced each other: tumors shrank more, spread less, and in some mice disappeared entirely. The combination boosted the number and vigor of killer T cells and natural killer cells, which produced more cancer-fighting molecules and mounted stronger attacks on tumor cells.
What This Means for Future Cancer Care
Taken together, the findings suggest that IL10RB marks and supports a late, hardened stage of cancer evolution—one that is less about rapid growth and more about survival, spread, and immune escape. By blocking IL10RB, it may be possible to strip away these protections, prevent chemotherapy from backfiring, and make existing immunotherapies more effective. While additional work in human samples and careful safety studies are still needed, IL10RB emerges here as a promising new handle for tackling some of the most treatment-resistant cancers.
Citation: Kudo-Saito, C., Ozawa, H., Imazeki, H. et al. IL10RB expression in cancer cells is associated with evolutionary changes to solidify treatment resistance. BJC Rep 4, 11 (2026). https://doi.org/10.1038/s44276-026-00211-3
Keywords: treatment resistance, cancer evolution, immunotherapy, tumor microenvironment, IL10RB