Clear Sky Science · en
Changes in levels of endocannabinoidome mediators in mice with cancer cachexia: links with steatosis and gut microbial dysbiosis
Why this research matters
Cancer cachexia is a severe wasting condition in which people with cancer unintentionally lose weight, appetite, and strength. It greatly worsens quality of life and survival, yet doctors still have few effective treatments. This study explores an unexpected trio of players that may be driving some of the damage: natural cannabis-like molecules made by our own bodies, fat buildup in the liver, and the trillions of microbes living in the gut.
The body’s own cannabis-like system
Our bodies produce a range of fatty molecules that can activate the same receptors as cannabis; together with their related compounds, enzymes, and receptors, this network is called the “endocannabinoidome.” It helps regulate appetite, how much energy we burn, liver fat storage, and how leaky or tight the gut barrier is. Because all of these processes go awry in cancer cachexia, the authors asked whether changes in these cannabis-like mediators might be linked to the weight loss, fatty liver, and disturbed gut microbes seen in this condition.
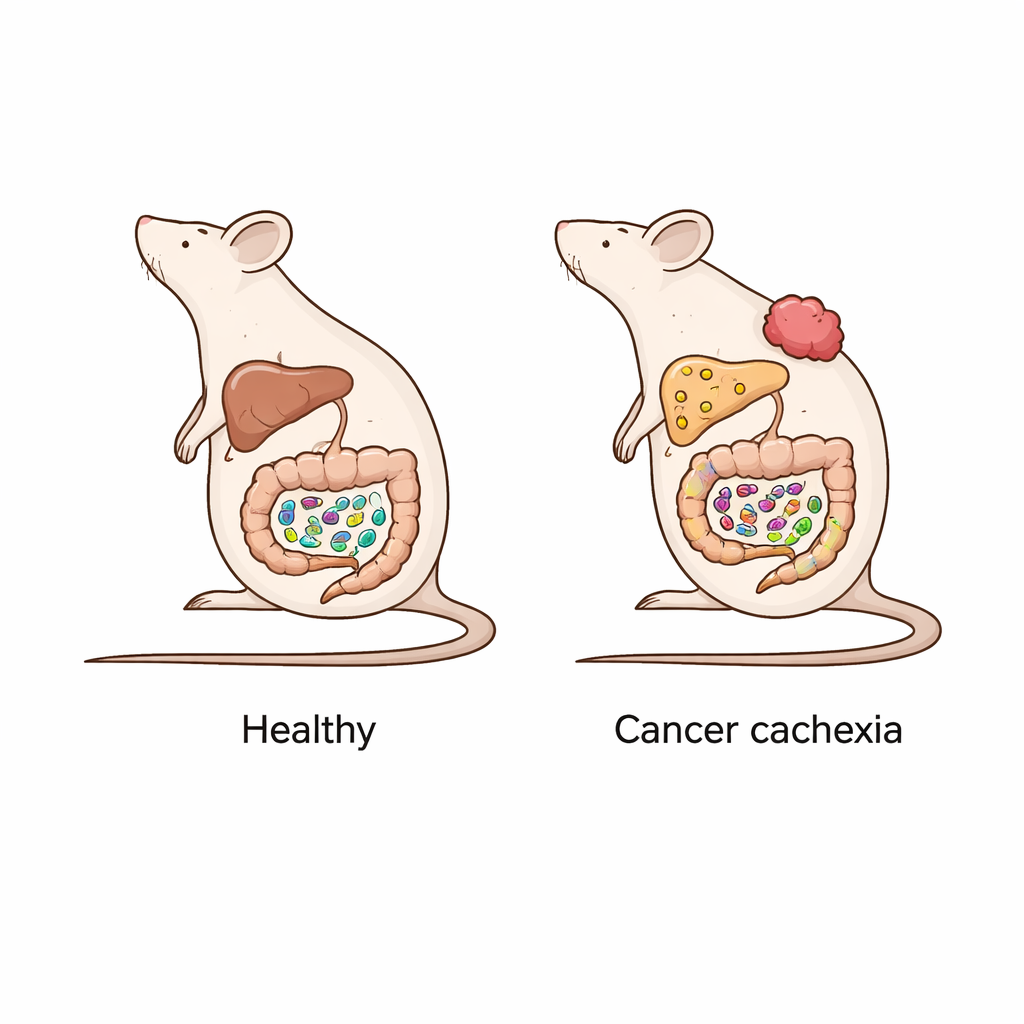
A mouse model of cancer-related wasting
The researchers used a common mouse model of cancer cachexia by injecting colon cancer cells under the skin. Within ten days, these mice lost more than 10% of their body weight, ate less, and developed clear signs of liver trouble: high amounts of fat, triglycerides, and cholesterol in the liver, along with altered levels of fats in the blood. The team carefully dissected different sections of the intestine and the liver, then used sensitive mass spectrometry techniques to measure dozens of endocannabinoid-related lipids. They also analyzed bacterial DNA from the gut to see how the microbial community had shifted compared with healthy control mice.
Shifts in cannabis-like molecules and liver fat
The study revealed striking organ-specific changes. In the small intestine, one key endocannabinoid, 2-AG, rose in the jejunum, while another, anandamide (AEA), fell sharply. In the liver, the pattern flipped: 2-AG dropped, but AEA and some of its chemical cousins, including OEA and several N-acyl-taurines, increased. These shifts were not simply a side effect of the mice eating less food; a separate “pair-feeding” experiment, where healthy mice were restricted to the same intake, did not reproduce most of the liver changes. Importantly, higher levels of several of these lipids in the liver went hand-in-hand with more liver fat, triglycerides, and cholesterol, suggesting they may help drive or respond to the development of fatty liver in cachexia.
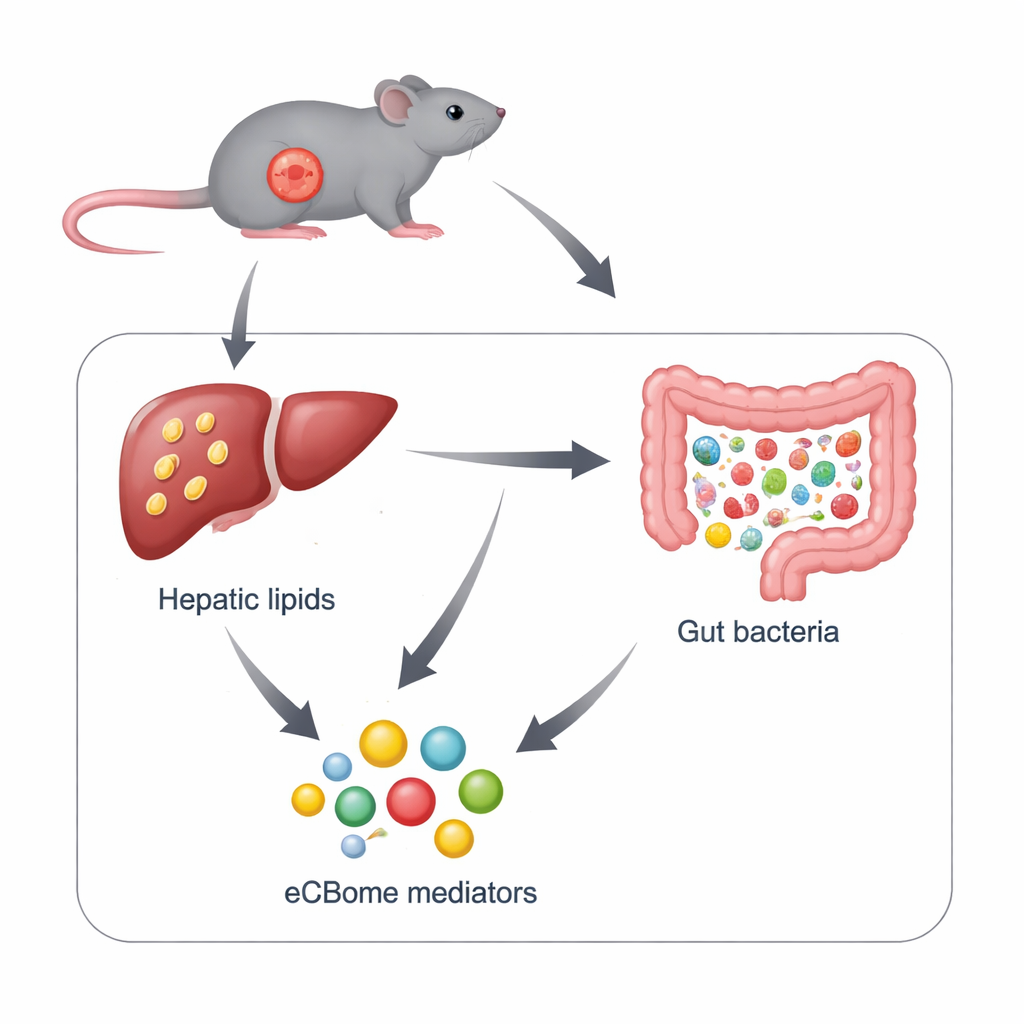
Gut microbes in disarray
The gut microbiome of cachectic mice was profoundly disrupted. While the total number of bacteria stayed similar, the balance between groups shifted dramatically: some potentially harmful families, such as Proteobacteria and Enterobacteriaceae, were greatly increased, whereas typically beneficial groups like Lachnospiraceae and certain fiber-fermenting genera declined. When the authors compared bacterial abundances with intestinal lipid levels, clear patterns emerged. Many endocannabinoid-related mediators tracked positively with genera that have been linked to metabolic problems, and negatively with genera that often support gut and liver health. This strengthens the idea of a two-way “microbiome–endocannabinoidome axis” in which gut microbes and these signaling lipids influence each other.
What this could mean for patients
Taken together, the findings suggest that in cancer cachexia, the body’s own cannabis-like molecules are not just innocent bystanders. Their levels change in the intestine and liver in ways that correlate with both fatty liver and a disturbed gut microbiome. Some of these shifts may be the body’s attempt to compensate; others may worsen liver fat buildup, gut leakiness, and inflammation. While this work was done in mice, it points to new avenues for research and eventually therapy, such as targeting specific endocannabinoid-related pathways or using microbiome-based strategies to restore a healthier balance and ease the burden of cancer-related wasting.
Citation: Degraeve, A.L., Cutignano, A., Piscitelli, F. et al. Changes in levels of endocannabinoidome mediators in mice with cancer cachexia: links with steatosis and gut microbial dysbiosis. BJC Rep 4, 7 (2026). https://doi.org/10.1038/s44276-026-00208-y
Keywords: cancer cachexia, endocannabinoidome, fatty liver, gut microbiome, mouse model