Clear Sky Science · en
STAR (stroma-tumor AI risk) assessment: association of AI-derived tumor-stroma proportion with patient survival provides added prognostic value beyond KELIM in epithelial ovarian cancer
Why the "neighborhood" around a tumor matters
When we think about cancer, we usually picture a mass of rogue cells. But tumors live in a busy neighborhood of support tissue, blood vessels, and immune cells called the stroma. This study asks a simple but powerful question: can we use the balance between tumor cells and this surrounding tissue, measured by artificial intelligence (AI) on routine microscope slides, to predict which ovarian cancer patients are likely to have better or worse outcomes—before treatment even begins?
A new way to read standard biopsy slides
Women with epithelial ovarian cancer typically have tissue removed at surgery or biopsy. Those samples are stained and examined under a microscope in every hospital. The researchers focused on a feature called tumor–stroma proportion (TSP): how much of the image is made up of supporting tissue compared with cancer cells. In earlier work, pathologists estimated this proportion by eye and found that tumors rich in stroma often behaved more aggressively and resisted standard platinum-based chemotherapy. In the current study, the team used a deep-learning AI system to automatically measure TSP on digitized slides, aiming to turn a manual, somewhat subjective task into a fast and reproducible readout available right at diagnosis.
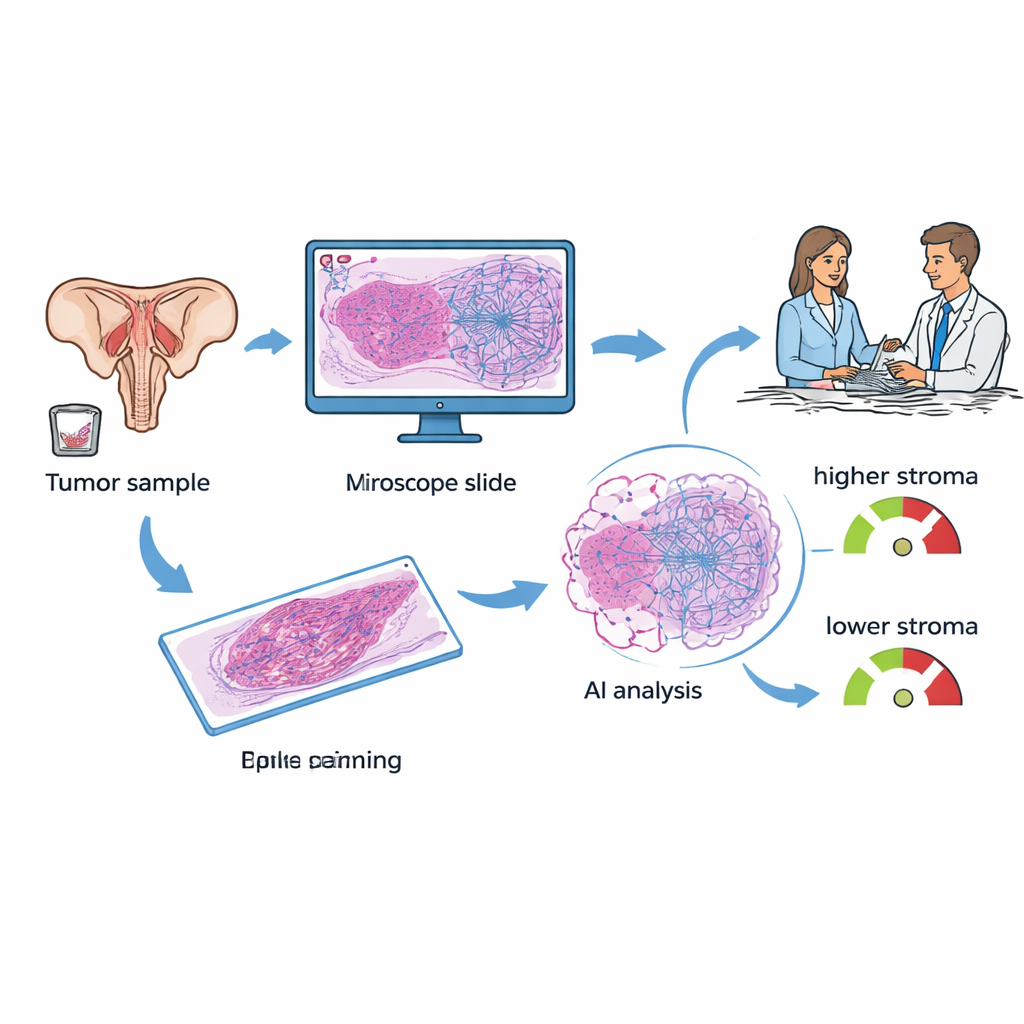
Comparing AI tissue analysis with a blood-based score
Today, one widely discussed tool for ovarian cancer is KELIM, a score based on how quickly a blood marker called CA-125 falls during the first 100 days of chemotherapy. KELIM is inexpensive and helpful, but it can only be calculated after several weeks of treatment, and not all patients have enough early blood tests to generate the score. The investigators assembled data from 89 women treated at a single cancer center who had both tissue slides and at least three CA‑125 measurements. They calculated KELIM for each patient, had a pathologist label tumor regions on the slides, and then let the AI model estimate TSP, classifying each case as "stroma-low" (less than 50% stroma) or "stroma-high" (50% or more).
AI matches experts and flags higher-risk tumors
The AI’s judgment closely matched that of experienced pathologists: in 94% of cases, the automated TSP classification agreed with the manual one, a level of concordance considered excellent. When the team looked at patient outcomes, they found that women whose tumors were stroma-high by AI had significantly worse overall survival than those with stroma-low tumors. In statistical terms, high TSP nearly doubled the risk of death during follow-up, and this effect persisted even after accounting for the KELIM score. By contrast, in this real-world group of patients, KELIM was linked to whether tumors were resistant to platinum drugs but was not clearly tied to how long patients lived.
What this could mean for treatment decisions
Because TSP can be measured from routine slides taken at the time of diagnosis, AI-based TSP offers a way to assess risk before chemotherapy starts, potentially months earlier than KELIM. A high TSP result might alert clinicians that a tumor is more likely to be aggressive and harder to treat, prompting closer monitoring, earlier consideration of additional therapies, or different surgical strategies. The method is also relatively low-cost: it relies on existing pathology images and a software model rather than new laboratory tests. The authors argue that automated TSP could work alongside other markers, such as genetic tests and CA‑125 trends, to refine prognosis without adding substantial burden to patients or pathology teams.
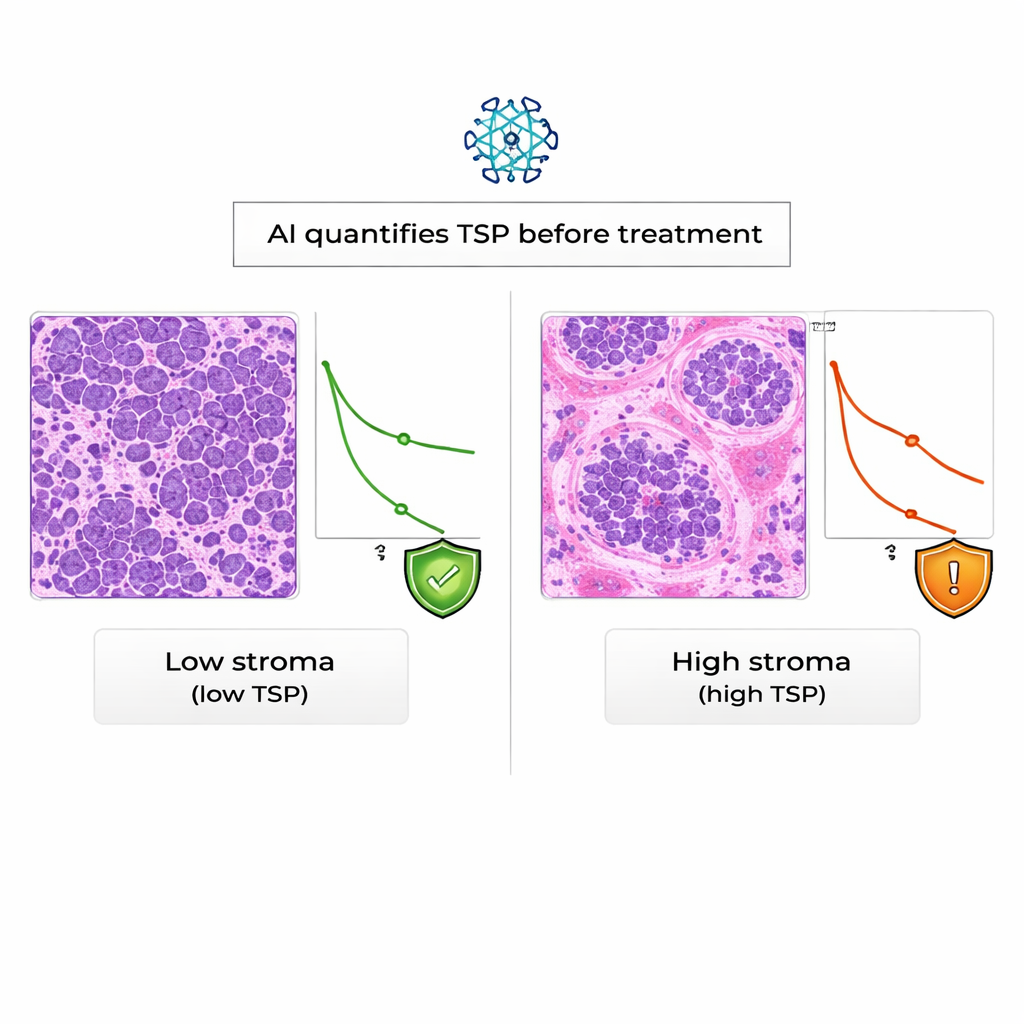
Looking ahead: smarter, earlier guidance for patients
For a layperson, the main message is that not all ovarian cancers are created equal, and the supporting tissue around the tumor carries important clues about how the disease will behave. This study shows that AI can read those clues reliably on standard biopsy slides and that a stroma-heavy tumor tends to be linked with poorer survival, independent of existing blood-based scores. While larger and more diverse studies are needed before this approach becomes part of routine care, AI-derived TSP holds promise as an early-warning sign that could help doctors tailor treatment plans and give patients clearer expectations right from the start.
Citation: Aggarwal, A., Madill, M., Jana, M. et al. STAR (stroma-tumor AI risk) assessment: association of AI-derived tumor-stroma proportion with patient survival provides added prognostic value beyond KELIM in epithelial ovarian cancer. BJC Rep 4, 4 (2026). https://doi.org/10.1038/s44276-026-00205-1
Keywords: ovarian cancer, artificial intelligence, tumor microenvironment, prognostic biomarker, digital pathology