Clear Sky Science · en
Priority effects inhibit the repeated evolution of phototrophy
Why sunlight is not a simple story
Sunlight powers nearly all life on Earth, yet the trick of turning light into usable biological energy—called phototrophy—has only evolved in two fundamentally different ways. This is puzzling: if nature managed to invent light-harvesting more than once, why did it stop there? This paper explores that mystery and argues that the earliest light-harvesting systems quickly filled up the available “space” for using light, leaving little room for any newcomers to take hold.
Two ways to live on light
Life uses two main strategies to capture light. One is the chlorophyll-based machinery familiar from plants and algae; the other relies on simpler pigments called retinal, used by many microbes in the oceans. Chlorophyll systems are elaborate, built from large protein complexes holding many pigment molecules and metal cofactors. They can power both energy production and the chemical reactions that pull carbon dioxide from the air or water to build biomass. Retinal systems, by contrast, are stripped-down: a single small protein holding one pigment molecule acts as a tiny light-driven pump that pushes protons across the cell membrane, providing a modest energy boost but not full-blown carbon fixation. Despite these contrasts, the total amount of sunlight captured by retinal users in the sea may rival that captured by classic chlorophyll photosynthesizers. 
Efficiency in the shade, power in the sun
The authors combine data from many modern organisms with a mathematical model to ask how these two systems perform under different light conditions. They measure two simple outcomes: how much energy each system gets per photon of light, and how much energy it can push through per unit of protein “hardware.” Chlorophyll machinery turns out to be excellent at squeezing a lot of energy from each photon, especially when light is scarce, such as in deeper water or shaded environments. But this comes at a cost: the complexes are bulky and expensive for the cell to build, so the maximum energy flow per unit protein is limited. Retinal machinery does the opposite. Each photon yields less energy, but the minimalist design allows very high energy throughput when light is intense, giving microbes a powerful but coarse tool for sunny conditions.
How early winners block late arrivals
Using their model, the researchers show that chlorophyll and retinal systems together span nearly the entire useful range of light-harvesting possibilities. For any given light level, there is a “best possible” combination of efficiency and power, forming what engineers call a Pareto front. Evolution should push any phototrophic lineage toward this front. The study finds that chlorophyll-based systems occupy the best territory under low light, while retinal-based systems dominate in bright light. Once both were established and refined early in Earth’s history, a would-be third phototrophic pathway would start off worse than both incumbents at all light levels. Such a newcomer would likely be outcompeted before it could evolve into something superior. In other words, the first successful light-harvesters created a priority effect: by getting there first and evolving to cover key niches, they shut the door on latecomers. 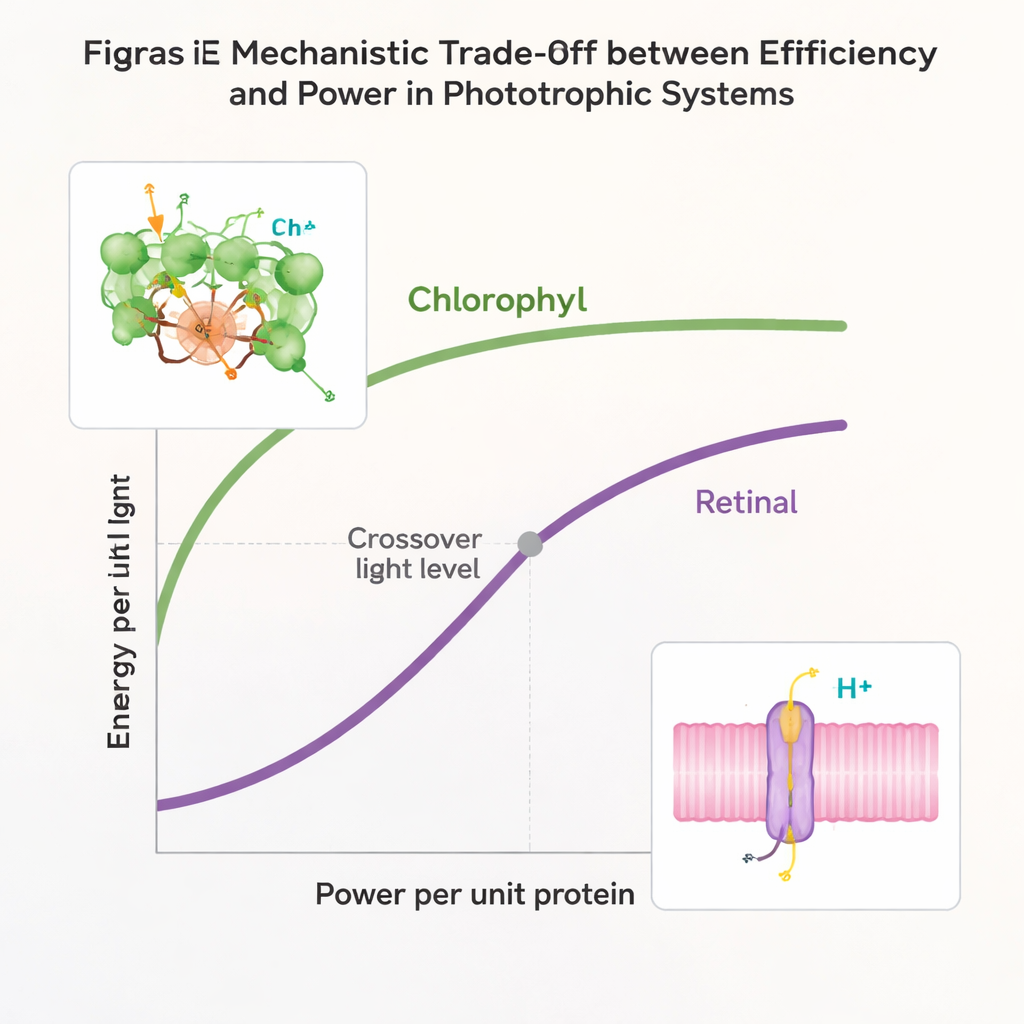
Who came first, and why both survived
The paper also asks why these two very different strategies coexist at all, rather than one ultimately displacing the other. A key difference is that chlorophyll systems can directly fuel carbon fixation, allowing organisms to build biomass from carbon dioxide alone, while retinal systems cannot. Retinal-based microbes must remain tied to existing organic matter; they can add energy to heterotrophic lifestyles but cannot independently support a large biosphere. This suggests a likely sequence: retinal-based phototrophy, being simpler, may have evolved first and tapped abundant mid-spectrum sunlight. Later, more complex chlorophyll systems arose, gained a foothold by enabling true autotrophy—living directly from light and inorganic carbon—and then expanded into wavelengths and environments not already dominated by retinal pigments. Once both systems had carved out their complementary roles, neither could easily replace the other across all conditions.
What this means for life on Earth and beyond
To a non-specialist, the main takeaway is that rarity does not always mean difficulty. Phototrophy feels like a once-in-a-billion-years innovation, but this work suggests it may actually be relatively easy to evolve under the right conditions. What makes it look rare is that once a successful version appears, it changes the environment and competitive landscape so thoroughly that parallel inventions cannot get started. The authors argue that this same “first come, first served” logic might apply to other big leaps in evolution, such as the origin of complex cells or even life itself. For astrobiology, the findings hint that other planets with life might also quickly settle into one or two dominant light-harvesting strategies, not because nature cannot invent more, but because early winners leave no room for rivals.
Citation: Burnetti, A.J., Stroud, J.T. & Ratcliff, W.C. Priority effects inhibit the repeated evolution of phototrophy. npj Complex 3, 9 (2026). https://doi.org/10.1038/s44260-026-00069-z
Keywords: phototrophy, photosynthesis, evolution, priority effects, astrobiology