Clear Sky Science · en
Genomic survey uncovers the emergence of a multidrug-resistant dominant lineage in Proteus mirabilis populations
Why this hospital germ matters to you
For many people, urinary tract infections are an occasional, painful nuisance. But in hospitals and nursing homes, some infections are driven by hardy bacteria that cling to catheters, shrug off multiple antibiotics, and spread silently between people and animals. This study follows one such culprit, Proteus mirabilis, across the globe, using its DNA as a travel diary to reveal how a single, hard-to-treat family line has quietly risen to dominance over the last century.
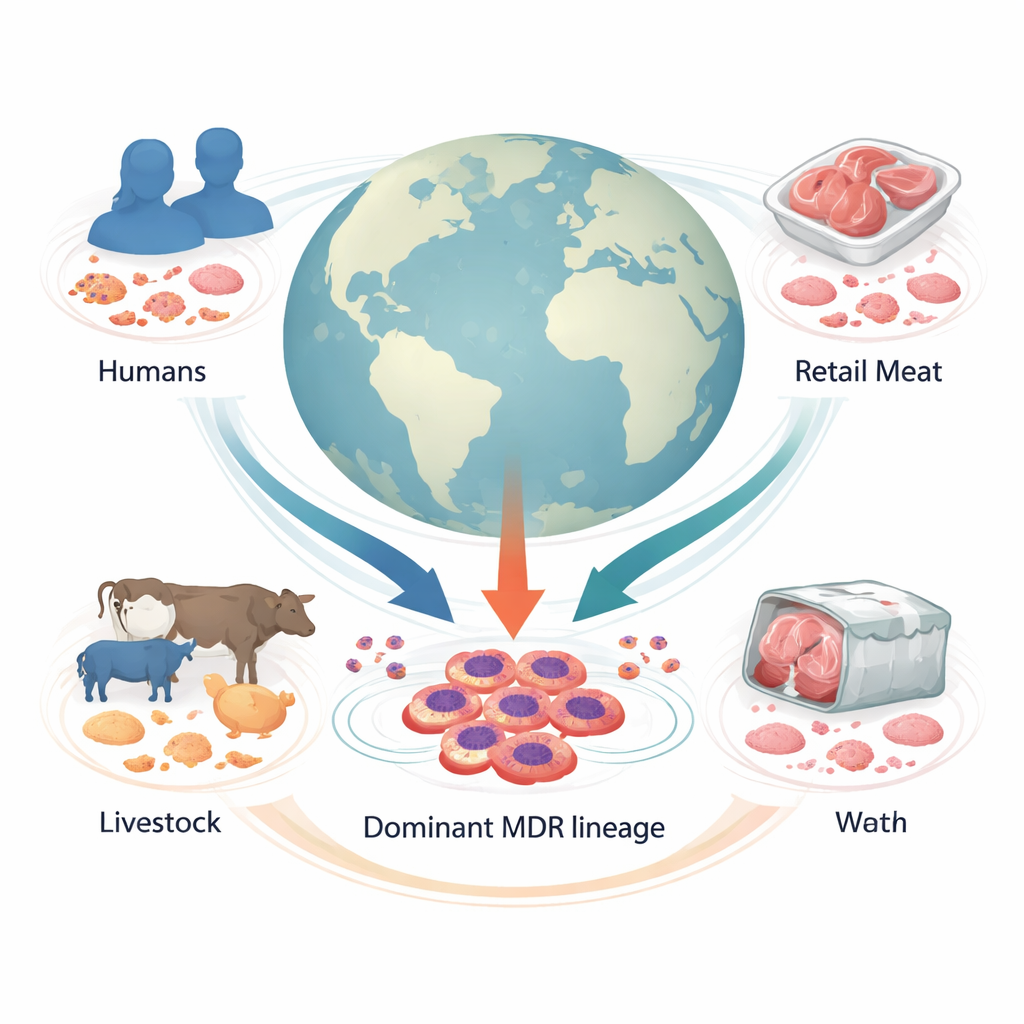
A globe-spanning look at a hidden threat
The researchers assembled genomes from 1,142 Proteus mirabilis samples collected in 34 countries from humans, livestock, retail meat, and other environments. By comparing whole genomes, they sorted the bacteria into 178 genetic families. One stood out: “Cluster-1,” a major lineage that accounted for one in five of all samples and included strains from hospitals on several continents. Most of these strains came from people, but a substantial share also came from farm animals and meat, indicating that this lineage moves across species and along the food chain.
A lineage armed with many drug shields
When the team scanned the genomes for antibiotic resistance genes, Cluster-1 carried a noticeably heavier load. On average, its members harbored almost 18 resistance genes each, far more than other lineages. These included genes that disable some of our most powerful antibiotics, such as carbapenems, often reserved as last-resort drugs. Many of these resistance genes sit inside a large DNA “island” called PmGRI1, which can hop between bacteria. Some versions of this island carry more than 20 resistance genes at once, effectively turning Proteus into a walking pharmacy of defenses and leaving doctors with few treatment options.
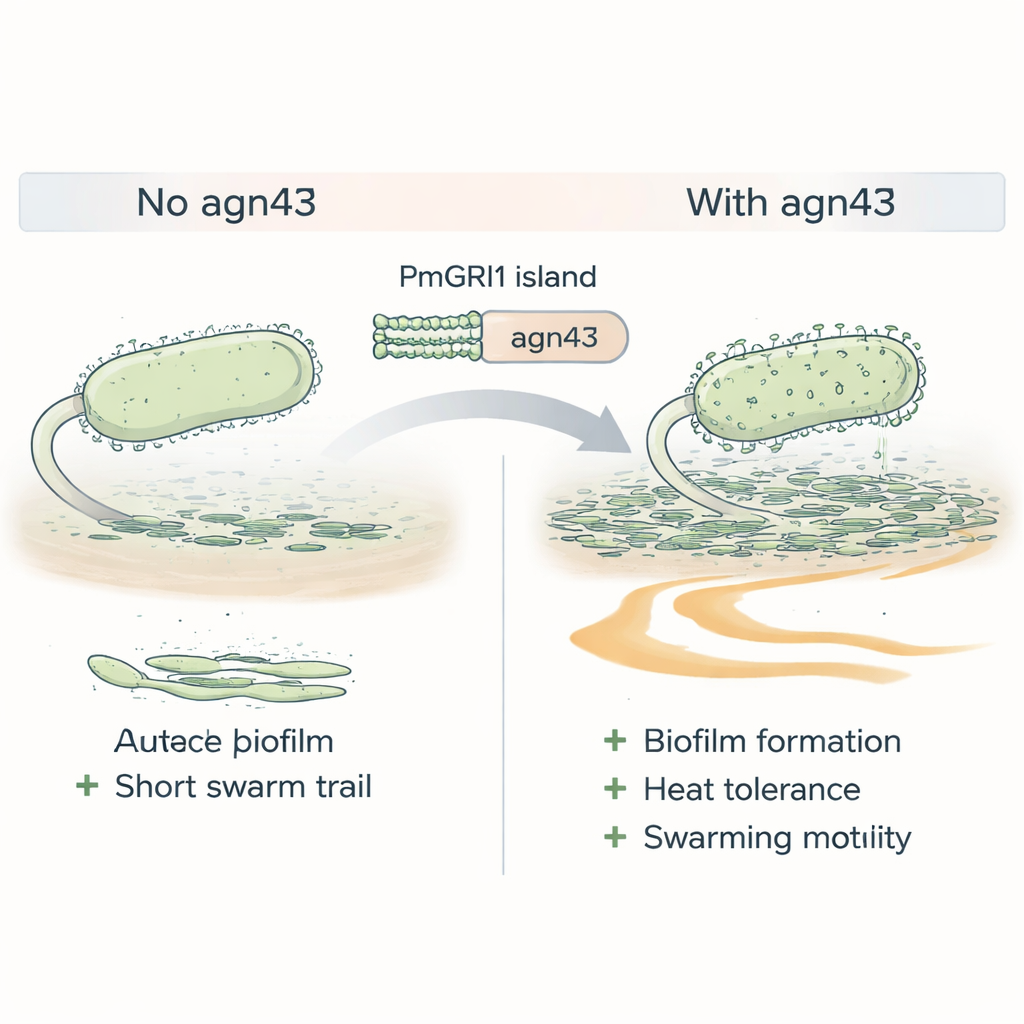
Sticky traits that help the bacteria hang on
Resistance alone does not guarantee success; bacteria must also be good at surviving in the body and hospital environment. Cluster-1 strains were enriched in genes linked to sticking to cells and forming biofilms—slimy communities that protect bacteria from drugs and the immune system. One standout gene on the PmGRI1 island, called agn43, makes a surface protein that acts like molecular Velcro, helping cells clump together. When the scientists deleted agn43 in a Cluster-1 strain, the mutant formed weaker biofilms, tolerated heat stress less well, and swarmed less efficiently across surfaces, all traits that normally help Proteus colonize urinary catheters and spread within the urinary tract.
How and where this super-lineage spread
By combining genetic differences with dates of isolation, the researchers reconstructed a timeline for Cluster-1. They estimated that its most recent common ancestor appeared around 1910, decades before modern antibiotics were widely used. The lineage later split into two medically important branches: one mainly seen in China that carries a carbapenem-breaking gene called blaKPC-2, and another mainly in the United States carrying a related gene, blaIMP-27. From the late 1980s onward, Cluster-1 expanded quickly and was repeatedly detected moving between countries and between humans, livestock, and meat, underlining its epidemic potential.
What this means for patients and public health
To a layperson, the message is that a particular family of Proteus mirabilis has evolved into a highly successful hospital germ by combining two advantages: a thick armor of antibiotic resistance genes and extra “stickiness” that helps it form protective films and persist on catheters and tissues. This combination makes infections harder to treat and easier to spread. The work highlights the need for close genetic surveillance of this lineage in both hospitals and agriculture, stricter infection control around catheters, and careful antibiotic use to slow the rise and global circulation of such multidrug-resistant strains.
Citation: Zhang, T., Wei, H., Ju, Z. et al. Genomic survey uncovers the emergence of a multidrug-resistant dominant lineage in Proteus mirabilis populations. npj Antimicrob Resist 4, 17 (2026). https://doi.org/10.1038/s44259-026-00189-5
Keywords: antibiotic resistance, urinary tract infection, hospital infections, bacterial genomics, biofilm