Clear Sky Science · en
Phage-meropenem synergy against OXA-48-producing Klebsiella pneumoniae clinical isolates
Why tiny viruses could rescue failing antibiotics
Hospitals around the world are struggling with infections that no longer respond to our strongest antibiotics. One of the worst culprits is a bacterium called Klebsiella pneumoniae, which can cause pneumonia, bloodstream infections, and urinary tract infections. This study explores whether carefully chosen viruses that attack bacteria—called phages—can team up with a powerful antibiotic, meropenem, to wipe out highly resistant strains that currently threaten patients’ lives.
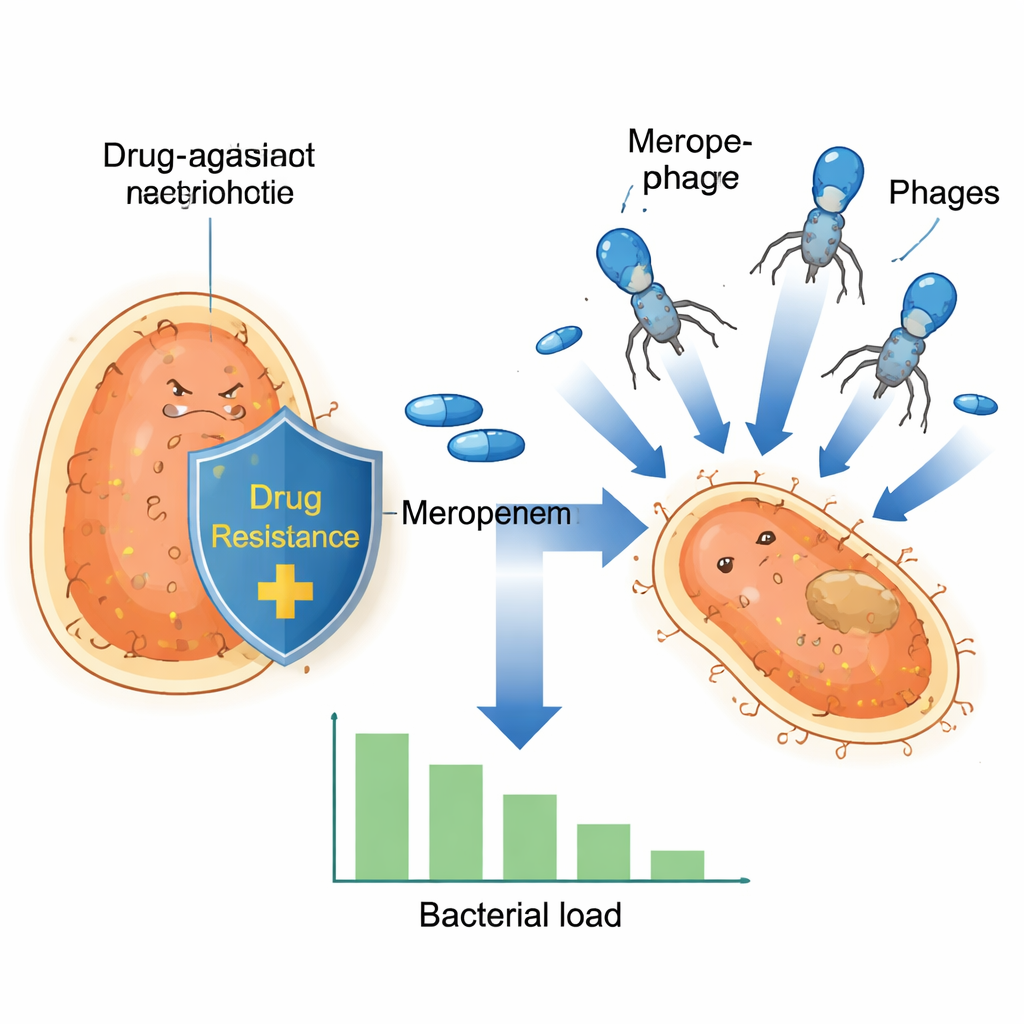
A stealthy hospital germ on the rise
Klebsiella pneumoniae has become a major hospital superbug because many strains have learned to resist carbapenems, a family of antibiotics reserved as a last line of defense. These bacteria often carry resistance genes on tiny DNA circles called plasmids, which they can swap with each other like trading cards. One such plasmid, known as pOXA-48, encodes an enzyme that breaks down carbapenems, leaving drugs like meropenem largely useless. As these plasmids spread through high‑risk bacterial clones across Europe and beyond, standard treatments become less reliable, making even routine medical procedures more dangerous.
Recruiting phages as precision attackers
Because new antibiotics are slow to arrive, researchers are turning to phage therapy—using viruses that specifically infect and kill bacteria. In this study, the team focused on a lytic phage called vB_Kpn_2-P4, which can infect a broad set of clinical Klebsiella isolates collected from Spanish hospitals. In lab tests, this phage was able to attack bacteria carrying several different carbapenem-resistance mechanisms. However, as often happens with both antibiotics and phages, some bacterial cells eventually survived and grew back, highlighting the need for combination strategies rather than relying on a single weapon.
When drug and virus work better together
The researchers then tested how meropenem and the phage behaved when used together. They grew twelve resistant bacterial isolates and exposed them to meropenem alone, phage alone, or both at once, tracking growth over many hours. While all of the isolates could tolerate meropenem under the test conditions, a striking pattern emerged: in strains carrying the OXA‑48 plasmid, the combination of phage plus high-dose meropenem caused a dramatic crash in bacterial numbers, followed by no detectable regrowth. At lower meropenem levels, these OXA‑48 strains still showed a strong drop in bacterial load. By contrast, strains with other resistance enzymes did not show this powerful joint effect, suggesting that some special feature of the OXA‑48 plasmid makes the bacteria uniquely vulnerable when facing both threats at once.
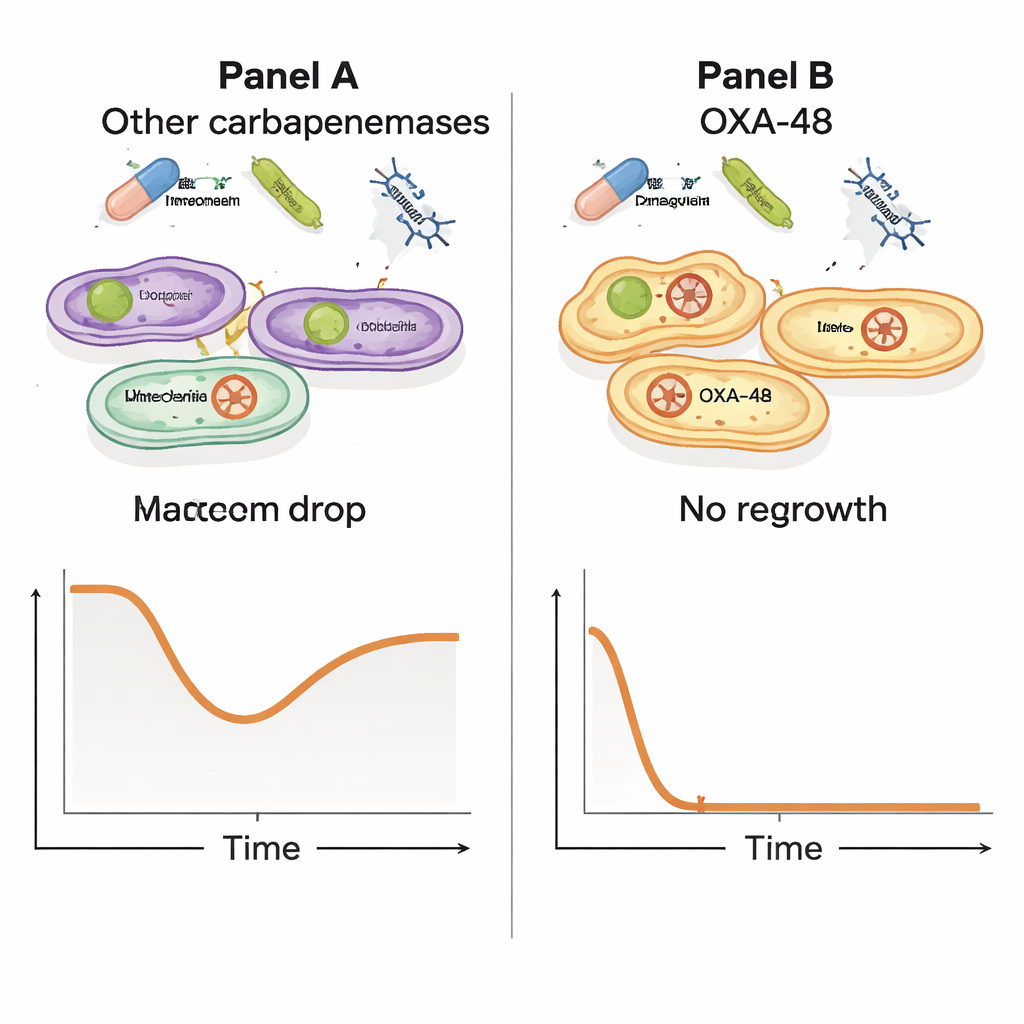
Tracking the hidden genetic helper
To understand why only the OXA‑48 producers showed this synergy, the team compared the genomes of the different bacterial isolates. They identified dozens of genes associated with the synergistic response, almost all clustered on the pOXA‑48 plasmid. Importantly, when they isolated bacteria that had evolved resistance to the phage, these survivors still carried an intact OXA‑48 gene; they had not simply thrown away the plasmid to escape. This points to a subtler picture in which the plasmid that grants drug resistance also imposes hidden costs on the bacteria’s biology—costs that become lethal when the phage and antibiotic strike together, tipping the balance against the pathogen.
What this could mean for future treatments
For non‑specialists, the key message is that the very genetic tricks bacteria use to dodge our drugs can sometimes be turned against them. In this case, a specific resistance plasmid appears to make certain Klebsiella strains exquisitely sensitive to a combined attack by a tailored phage and meropenem. While this work was done in laboratory conditions and more research is needed in animal models and patients, it suggests that matching the right phage to the right resistance profile could revive antibiotics that seem to have lost their power. Rather than replacing antibiotics, phages may become smart allies that help us outmaneuver superbugs that once seemed unbeatable.
Citation: Cantallops, I., Ferriol-González, C., Barcos-Rodríguez, T. et al. Phage-meropenem synergy against OXA-48-producing Klebsiella pneumoniae clinical isolates. npj Antimicrob Resist 4, 12 (2026). https://doi.org/10.1038/s44259-026-00186-8
Keywords: phage therapy, antibiotic resistance, Klebsiella pneumoniae, OXA-48 plasmid, meropenem synergy