Clear Sky Science · en
Genomic landscape of antimicrobial resistance in India: findings from a multi-species surveillance study
Why superbugs in India matter to all of us
Antibiotic-resistant “superbugs” are a growing global worry, but we still know surprisingly little about how these germs evolve and spread in many parts of the world. This study looks closely at dangerous bacteria from hospitals in India and reads their entire DNA to understand how they outsmart our strongest drugs. The results not only reveal what makes these microbes so hard to kill, but also test whether fast DNA-based methods can reliably replace slower lab tests—an issue that could affect how infections are treated everywhere.
Taking a closer look at hospital infections
Researchers collected 266 bacterial samples from seriously ill patients in large hospitals in northern and western India between 2022 and 2024. Most samples came from blood, but also from urine and lung infections, and the majority were from intensive care units, where patients are most vulnerable. The team focused on well-known problem germs—such as Escherichia coli, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, methicillin‑resistant Staphylococcus aureus (MRSA) and vancomycin‑resistant Enterococcus (VRE). For each sample, doctors had already done standard antibiotic susceptibility testing, which exposes bacteria to drugs in the lab to see which medicines still work. The scientists then sequenced the bacteria’s genomes to map the full set of resistance genes and compared what the DNA “predicted” with what actually happened in the test tubes.
When genes and test tubes disagree
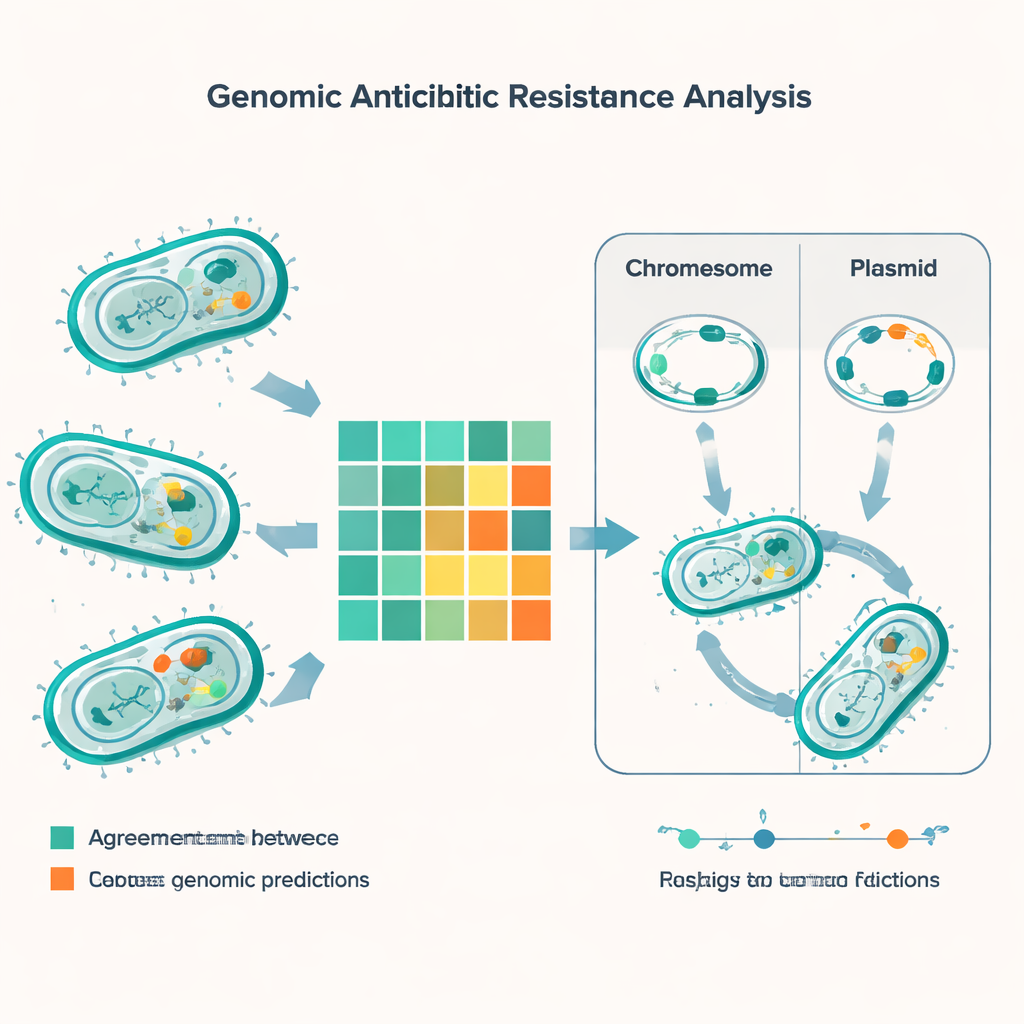
By matching genetic predictions against lab results for 56 different antibiotics, the study ran more than 5,000 comparisons. In most cases, the DNA‑based method and the traditional tests agreed, but nearly 600 mismatches stood out. The most common kind of error was that the genomic tool predicted a bacterium would be resistant when the lab test said it was still treatable. This happened often for drugs such as minocycline, colistin and gentamicin, especially in E. coli. The opposite type of error—where the lab saw resistance but the genes did not clearly explain it—was less frequent but more worrying, because it risks missed resistance. These “very major” discrepancies showed up notably in gut‑related bacteria called enterococci, particularly for widely used penicillin‑like drugs and for the combination trimethoprim‑sulfamethoxazole.
Hidden arsenals inside bacterial DNA
The genomic survey uncovered a dense arsenal of resistance genes across the major species. Every one of the common Gram‑negative pathogens carried at least one beta‑lactamase gene, which can break down penicillin and related drugs, and many strains had several such genes at once. Notorious culprits like NDM‑type carbapenemases—which neutralize last‑resort antibiotics—were widespread in E. coli, Klebsiella, Acinetobacter and Pseudomonas. The study also found genes that help bacteria survive powerful “peptide” antibiotics such as colistin, along with the classic mecA gene that makes MRSA resistant to methicillin and vancomycin resistance clusters in enterococci. By comparing DNA fingerprints called sequence types, the team linked some of these resistance genes to known high‑risk bacterial lineages that are already spreading in India and around the world.
Gene‑sharing via mobile DNA
A crucial part of the story lies not just in which genes the bacteria carry, but where those genes live. Many resistance genes sit on small DNA circles called plasmids, which bacteria can swap like trading cards. Using specialized software, the researchers predicted about 1,400 plasmids across the samples, with especially high diversity in E. coli and Klebsiella. In these species, a large share of resistance genes—including several critical beta‑lactamases—were plasmid‑borne, making them easier to pass between strains and even between species. Other resistance traits were hard‑wired into bacterial chromosomes, meaning they can persist even if plasmids are lost. The team also catalogued mobile genetic elements—short DNA segments that can hop around and carry resistance genes with them—highlighting yet another route for rapid spread.
What this means for future treatments
For non‑specialists, the central message is that reading bacterial DNA can greatly aid the fight against superbugs, but the technology is not yet perfect. The genomic tools tended to “overcall” resistance, which is safer than missing a dangerous strain but could push doctors to use stronger drugs than necessary. At the same time, a smaller number of cases showed that lab tests detected resistance that current gene catalogues could not fully explain, underscoring important blind spots in our knowledge. By building a detailed map of resistance genes, plasmids and mobile elements in Indian hospitals, this study creates a foundation for better, faster DNA‑based diagnostics and more informed antibiotic use—not just in India, but in any setting where superbugs threaten modern medicine.
Citation: Gheewalla, N., Karthikeyan, V., Jadhav, Y. et al. Genomic landscape of antimicrobial resistance in India: findings from a multi-species surveillance study. npj Antimicrob Resist 4, 13 (2026). https://doi.org/10.1038/s44259-026-00185-9
Keywords: antimicrobial resistance, genome sequencing, hospital infections, drug resistant bacteria, plasmid mediated resistance