Clear Sky Science · en
Global response to antibiotic exposure reveals a critical role for nucleotide metabolism in high-level β-lactam tolerance
Why some bacteria shrug off our best antibiotics
Antibiotics are supposed to wipe out harmful bacteria, yet many infections stubbornly linger or come back after treatment. This paper explores one underappreciated reason why: some bacteria can hunker down and temporarily survive even very high doses of powerful drugs like penicillin. By uncovering how these microbes rewire their inner chemistry to ride out the attack, the researchers point to new ways we might restore the punch of existing antibiotics.
A stealthy survival trick inside infections
When exposed to beta-lactam antibiotics such as penicillin, many dangerous Gram-negative bacteria don’t simply die. Instead, they can shed their rigid cell wall and turn into fragile, round bodies called spheroplasts. In this form, they stop multiplying but remain very much alive and metabolically active. Once the drug is gone, they rebuild their wall, resume their normal rod shape, and can reignite infection. Because this “tolerance” is a stepping-stone toward full-blown antibiotic resistance and treatment failure, understanding how spheroplasts survive is crucial for future medicine.
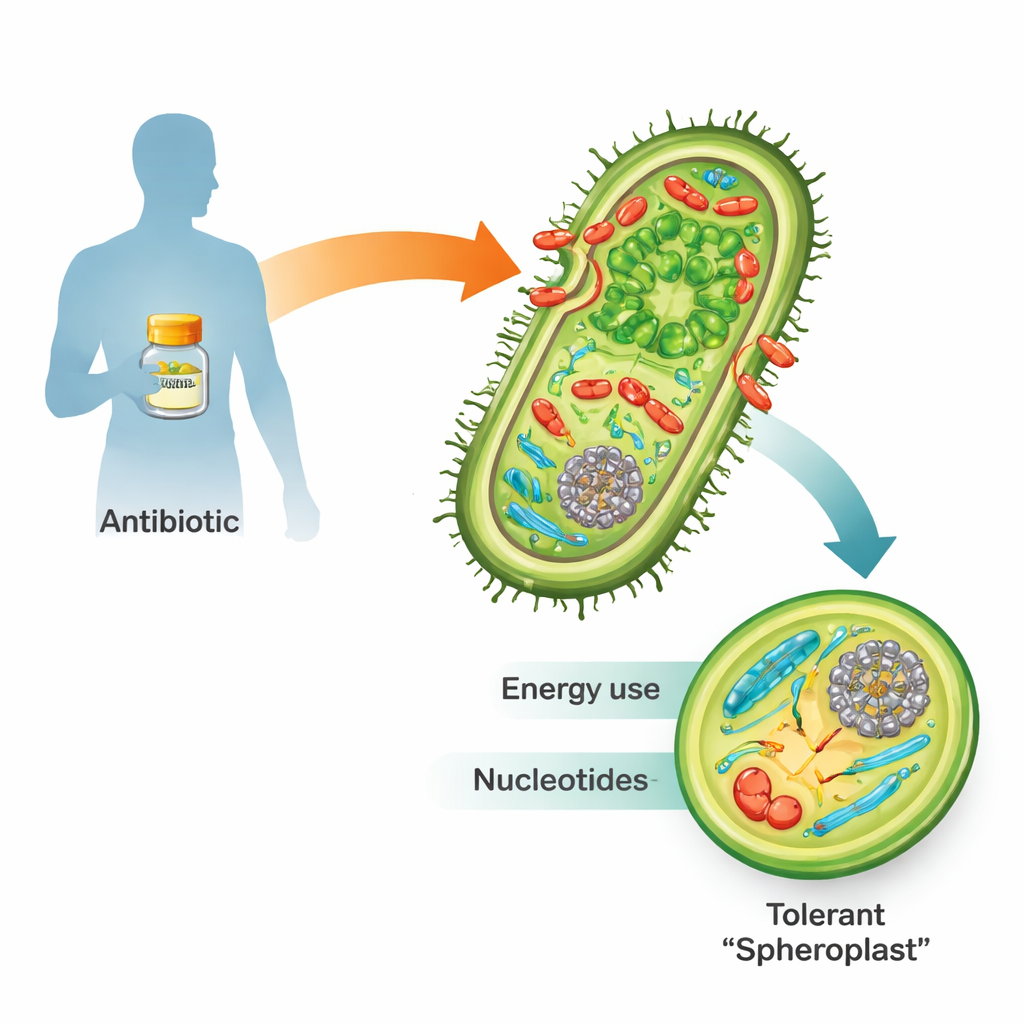
Listening in on the cell’s emergency broadcast
The team used Vibrio cholerae, the bacterium that causes cholera, as a model because it is exceptionally tolerant to beta-lactams and easy to manipulate genetically. They bathed the bacteria in ten times the minimum lethal dose of penicillin and followed the response over time with two powerful tools. Transcriptomics tracked which genes were turned on or off, while metabolomics measured hundreds of small molecules that fuel and build the cell. Together, these “multi-omics” techniques created a time-resolved map of how a tolerant cell remodels its inner workings during a drug assault.
Metabolic rerouting and a hidden weak spot
The data revealed sweeping changes in core pathways. Genes for building the cell wall switched on strongly, consistent with the cell’s attempt to repair damage and prepare for recovery. Heat-shock and stress-response systems also flared, likely dealing with misfolded, oxidized proteins created by antibiotic-induced stress. At the same time, central carbon metabolism shifted: certain steps of the TCA cycle (the cell’s main energy generator) were boosted, while key glycolysis intermediates such as glucose-6-phosphate and fructose-6-phosphate were dramatically depleted. These intermediates normally feed both energy production and cell-wall building, hinting that continuous, wasteful “futile cycling” of wall material was draining resources.
Nucleotides under pressure
The most striking change involved nucleotides, the building blocks of DNA, RNA and many energy-carrying molecules. Levels of many nucleotides and their precursors plummeted in penicillin-treated cells, even though the genes for making them from scratch were strongly activated. At the same time, genes involved in “recycling” nucleotides were dialed down, as if the cell were trying to conserve what little remained. These patterns suggested that spheroplasts are under severe nucleotide stress. When the researchers deliberately interfered with pathways that supply nucleotide precursors—such as the pentose phosphate pathway—or blocked nucleotide production with another drug, trimethoprim, the combination with beta-lactams killed far more bacteria than either drug alone. This strong synergy was seen not only in Vibrio cholerae but also in highly tolerant clinical strains of Klebsiella pneumoniae and Escherichia coli.
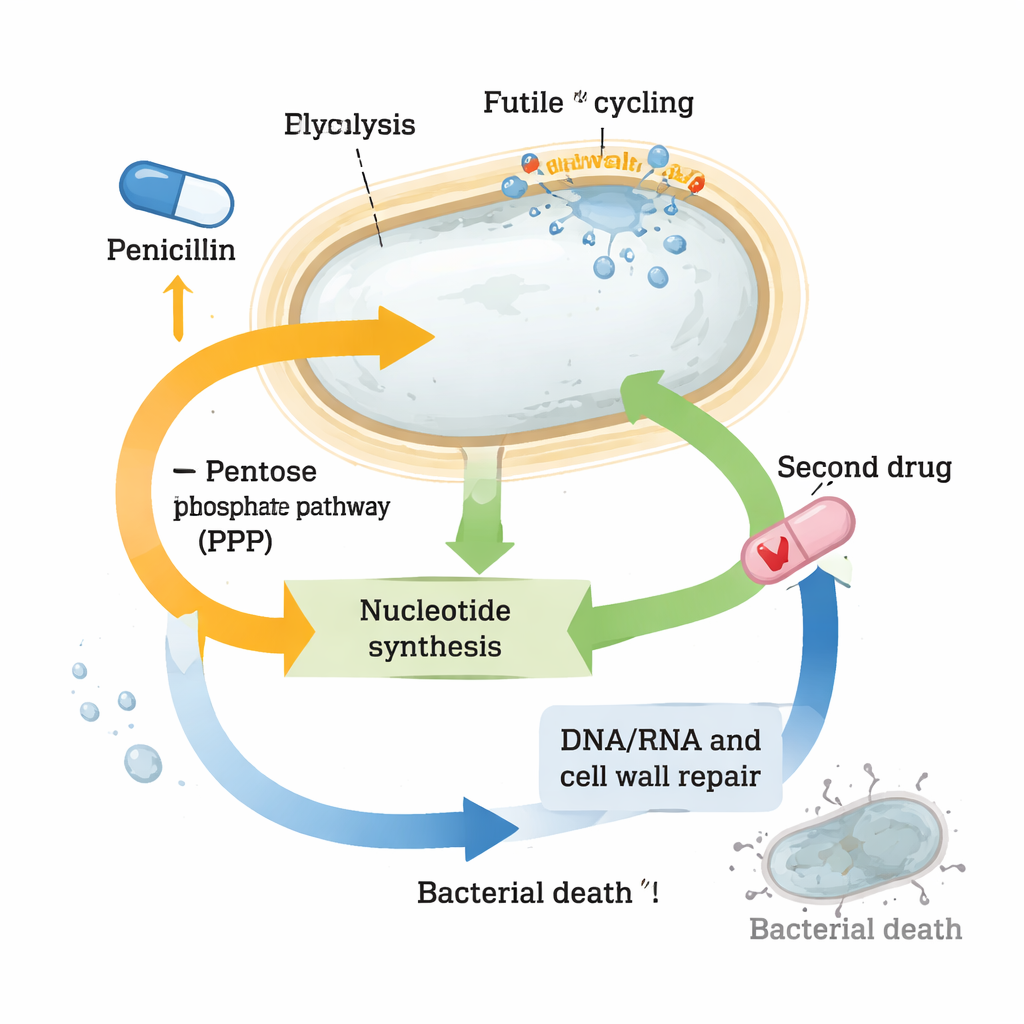
Turning survival chemistry into a therapeutic advantage
Despite major reshaping of their metabolism, tolerant spheroplasts manage to keep their energy currency, ATP, relatively stable and avoid catastrophic damage, which helps them survive long antibiotic exposures. But the work shows that, in doing so, they push their nucleotide pools to the brink. That precarious balance creates a vulnerability: nudging nucleotide metabolism just a bit further with a second drug collapses their defenses and restores potent killing. For lay readers, the takeaway is that some bacteria survive antibiotics not because the drugs fail to hit their targets, but because the cells rapidly rewire their chemistry to endure the blow. By finding and exploiting the weak links in this survival wiring—here, nucleotide metabolism—researchers may be able to turn old antibiotics into powerful combination therapies that outsmart even highly tolerant pathogens.
Citation: Keller, M.R., Kazi, M.I., Saleh, A. et al. Global response to antibiotic exposure reveals a critical role for nucleotide metabolism in high-level β-lactam tolerance. npj Antimicrob Resist 4, 11 (2026). https://doi.org/10.1038/s44259-026-00183-x
Keywords: antibiotic tolerance, beta-lactam antibiotics, nucleotide metabolism, bacterial persistence, drug combinations