Clear Sky Science · en
Prevalence and patterns of antimicrobial resistance among wildlife populations in Africa: a systematic review
Why wild animals matter for antibiotic resistance
Antibiotic resistance is often talked about in hospitals and farms, but wild animals live in the same landscapes, drink the same water, and move across the same borders as people and livestock. This study pulls together decades of scattered research to ask a simple but important question: how common are antibiotic‑resistant bacteria in African wildlife, and what does that tell us about the wider environment? The answers matter for anyone concerned with food safety, conservation, or the future effectiveness of lifesaving drugs.
Looking across the continent
The authors systematically searched major scientific databases and sifted through 4,802 papers to find studies that actually tested bacteria from free‑living wild animals in Africa. Only 61 studies, from 21 of Africa’s 54 countries, met their criteria. Most came from North and West Africa, with relatively few from central or southern regions, and almost none from truly untouched habitats. The research mainly relied on samples from animal droppings, collected opportunistically in protected areas or near villages, farms, and landfills. This patchy coverage means we know a lot more about wildlife living near people than animals in remote ecosystems. 
What scientists found in wild animals
Across the studies, scientists isolated 55 different bacterial species from birds, monkeys, herbivores, bats, rodents, and other wildlife. One common gut bacterium, Escherichia coli, was by far the most frequently examined. When the authors combined data from 27 studies that reported clear counts of resistant and non‑resistant isolates, they found that about 59% of bacterial samples from African wildlife were resistant to at least one antibiotic. When they looked only at more recent work (after 2010), the estimate rose to about 65%. For E. coli specifically, roughly six in ten isolates carried resistance. Even more concerning, nearly one quarter of tested isolates in a subset of studies were resistant to multiple classes of antibiotics at once, a pattern known as multidrug resistance.
Different animals, different risks
The level of resistance varied sharply between animal groups, reflecting how and where they live. Large plant‑eating mammals, which often roam in relatively undisturbed savannas, showed the lowest pooled prevalence of resistant bacteria, around 25%. Non‑human primates, which frequently share space and sometimes food with people, had a higher rate of about 35%. Wild birds topped the list, with an estimated 93% of tested bacterial isolates showing resistance. Many birds feed at landfills, sewage outflows, or farm fields and can travel long distances, making them plausible carriers of resistant microbes across regions and even continents. These patterns suggest that contact with human activity—through waste, water, and shared grazing areas—strongly shapes where resistance shows up in wildlife.
Gaps in what we know
Despite relatively strong laboratory procedures, the studies had notable blind spots. Most used convenience sampling—collecting whatever animals or droppings were easiest to reach—rather than methods designed to represent whole populations. Few studies recorded ecological details such as diet, movement patterns, or exact levels of human and livestock contact, making it hard to link resistance clearly to specific behaviours or environments. Research also focused heavily on human‑associated bacteria like E. coli and Klebsiella, with limited attention to pathogens that primarily affect wildlife health. As a result, it remains unclear whether wild animals are long‑term reservoirs that maintain resistant bacteria, short‑term “passengers” that pick them up from contaminated sites, or early warning sentinels pointing to pollution hotspots. 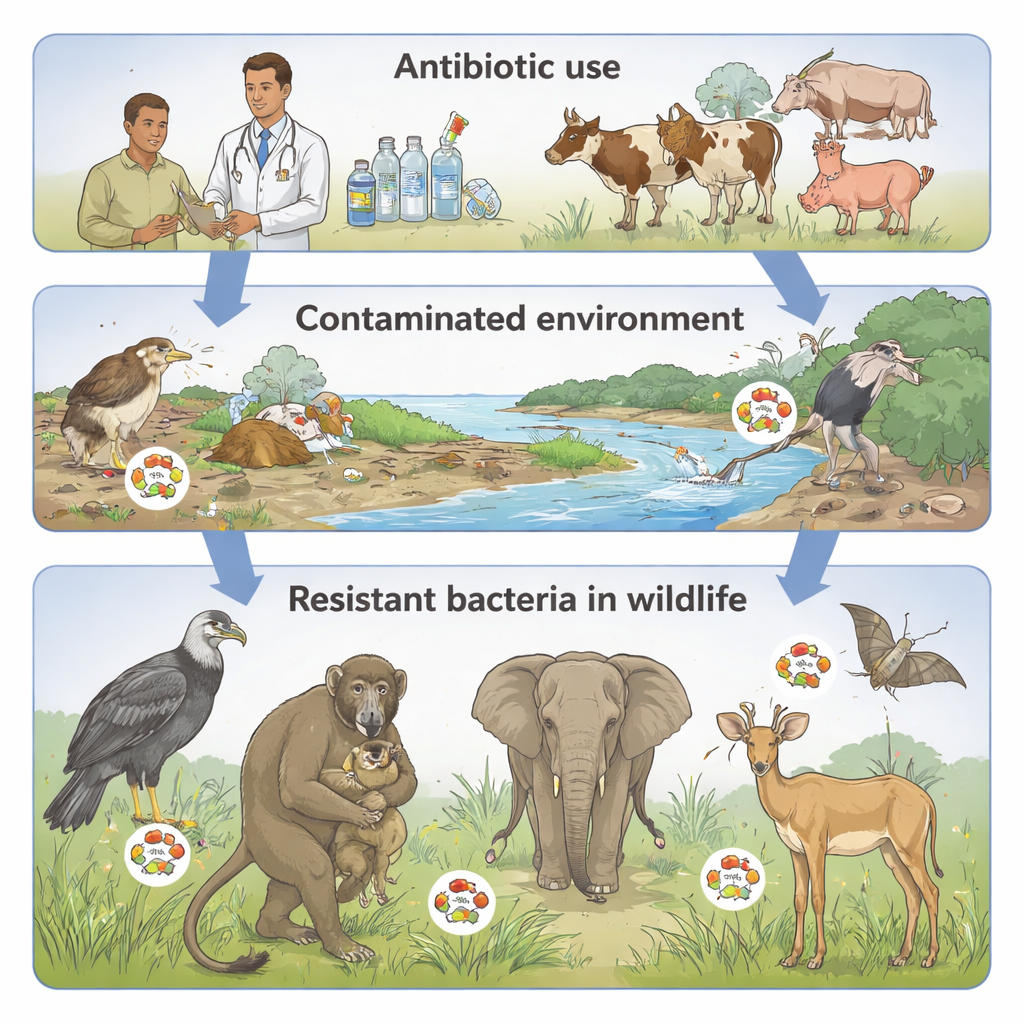
What this means for people and the planet
For a general reader, the key message is that antibiotic resistance is not confined to hospitals or farms; it is woven through entire landscapes and shows up in many wild species across Africa. The review indicates that resistant and multidrug‑resistant bacteria are widespread in wildlife, especially in places heavily shaped by human activity. However, because most data come from biased sampling near settlements and from a limited set of countries, we still lack a clear picture of what “normal” resistance looks like in untouched ecosystems, or how often bacteria move between wildlife, livestock, and humans. The authors argue that wildlife should be seen as context‑dependent participants in this problem—sometimes victims of pollution, sometimes potential carriers, sometimes useful sentinels. They call for better‑designed, continent‑wide monitoring that integrates wildlife into existing human and veterinary surveillance systems. Such efforts, they suggest, will be vital for understanding where resistance arises, how it spreads, and how we can protect both human health and biodiversity in a shared environment.
Citation: Mwangi, J.W., Kimeu, A., Moodley, A. et al. Prevalence and patterns of antimicrobial resistance among wildlife populations in Africa: a systematic review. npj Antimicrob Resist 4, 9 (2026). https://doi.org/10.1038/s44259-025-00179-z
Keywords: antimicrobial resistance, wildlife, Africa, One Health, bacteria