Clear Sky Science · en
Steering charge transfer in CuInS2/BiOCl composites to enable sunlight-driven C–F bond cleavage of PFAS in water
Why Breaking “Forever Chemicals” Matters
For decades, a family of man‑made compounds known as “forever chemicals” has seeped into drinking water, food packaging, firefighting foams and our bodies. These chemicals, technically called PFAS, are prized for their resistance to heat and stains—but the same toughness makes them extremely hard to remove from the environment. This paper reports a sunlight-driven technology that can actually crack the strongest bonds in a key PFAS substitute in water, pointing toward practical ways to clean up contaminated rivers, tap water and industrial effluent.
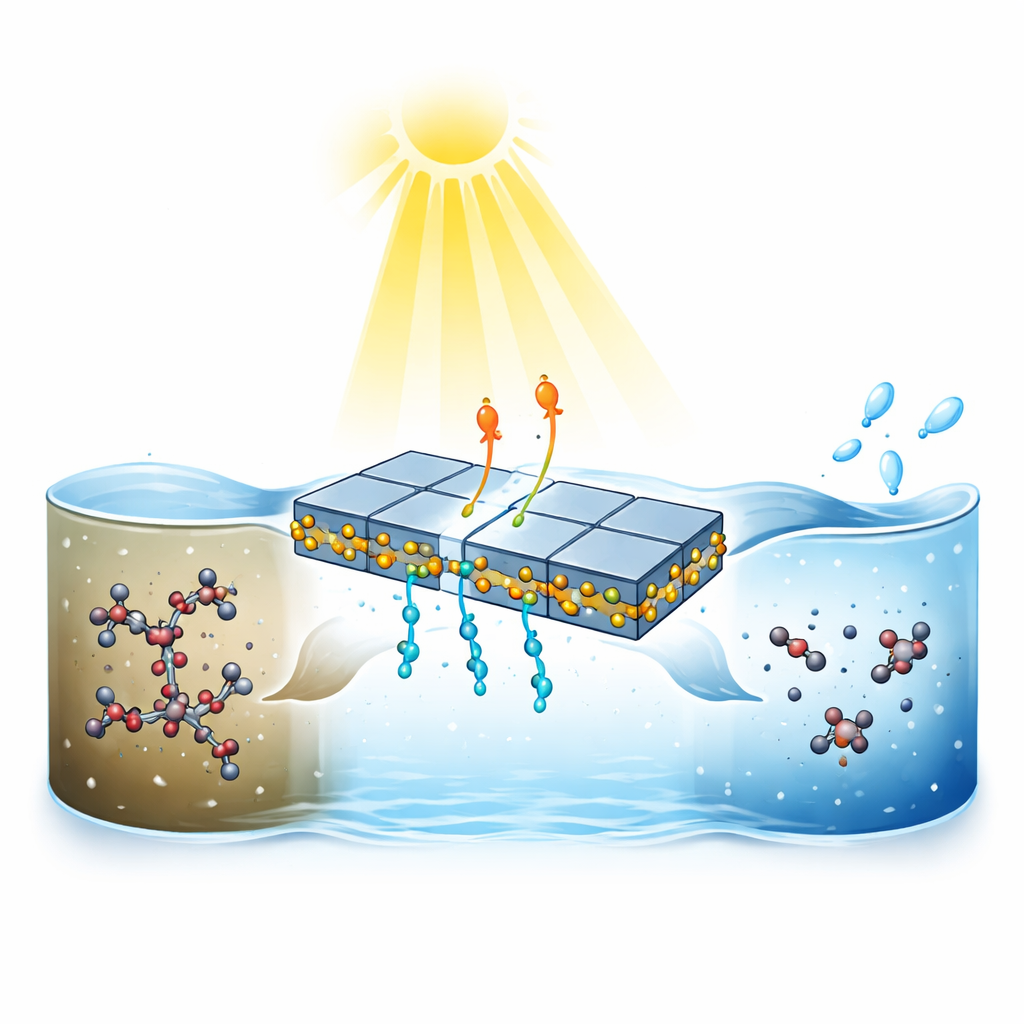
A New Way to Harness Sunlight
The researchers focused on sodium p‑perfluorous nonenoxybenzenesulfonate (OBS), a replacement for older PFAS that is now turning up in surface waters and raising health concerns. Conventional treatments struggle with PFAS because their carbon–fluorine bonds are among the strongest in chemistry, often requiring high temperatures, pressures or harsh chemicals to break. Inspired by how plants separate electric charges during photosynthesis, the team designed a layered light‑activated material that can use ordinary sunlight to attack OBS in water under mild conditions.
Building a Two‑Part Catalyst
The heart of the system is a carefully engineered partnership between two semiconductors: thin, plate‑like crystals of bismuth oxychloride (BiOCl) and tiny copper–indium sulfide (CuInS₂) quantum dots. When combined, these materials form what is known as a Z‑scheme heterojunction, a structure that channels light‑generated negative charges (electrons) into the CuInS₂ particles and positive charges (holes) into the BiOCl plates. Microscopy and advanced X‑ray measurements show that the quantum dots cling tightly to the edges of the plates through sulfur–bismuth bonds, creating intimate contact that speeds charge flow and prevents electrons and holes from simply recombining and wasting the absorbed light.
Snapping the Toughest Bonds
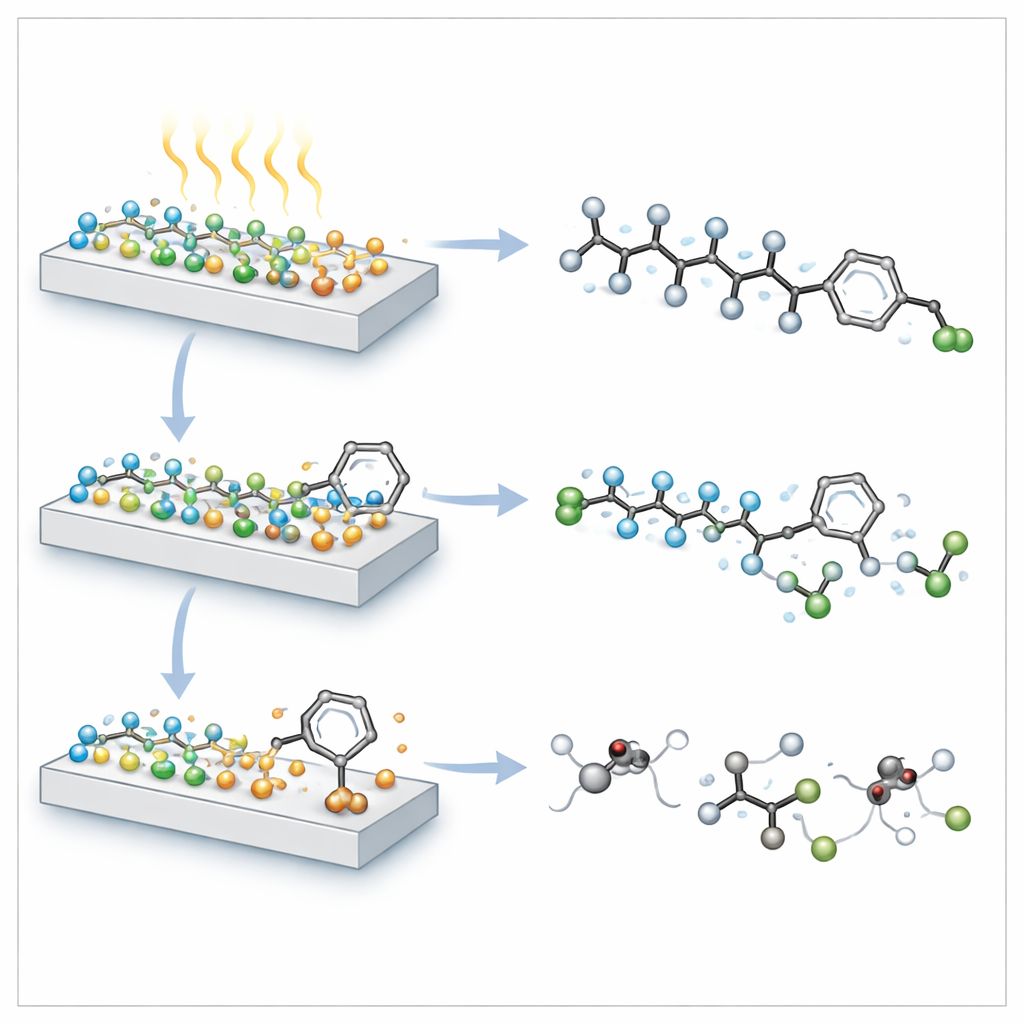
Once light hits the composite, the separated charges become powerful chemical tools. Calculations and spectroscopy reveal that electrons congregating on the CuInS₂ quantum dots are strongly reducing: they home in on the fluorine‑rich branch of the OBS molecule, weakening and then breaking carbon–fluorine bonds so that fluoride ions are released. At the same time, the positively charged holes on the BiOCl plates attack the sulfonic acid headgroup and the attached benzene ring, chopping up the carbon framework. Together, these twin processes shorten the carbon chain and strip off fluorine atoms far more efficiently than either material alone. Under ultraviolet light, the optimized composite removes about three‑quarters of both total fluorine and total organic carbon from OBS in just eight hours—among the highest performances reported so far.
From Lab Beakers to Flowing Water
To see whether this approach could work outside the lab, the team coated flexible polyester sheets with the catalyst and built a simple panel reactor through which contaminated water could flow while exposed to natural sunlight. In outdoor tests, the system removed more than 96% of OBS from water over ten hours, with almost no loss of catalyst. The composite also degraded mixtures of 17 different PFAS, including both long‑ and short‑chain versions, and did so in real river water containing minerals and natural organic matter. Toxicity tests using tiny worms and zebrafish embryos showed that the treated water had dramatically reduced biological effects compared with the untreated solutions.
What This Means for Cleaner Water
In plain terms, this study demonstrates a solar‑powered filter that does more than merely trap PFAS—it helps destroy them. By steering light‑generated charges to the right places within a two‑part material, the researchers were able to break some of the strongest bonds in modern chemistry and dismantle complex PFAS molecules into far less harmful pieces. While further work is needed before large‑scale deployment, the results suggest a realistic path toward continuous‑flow, energy‑efficient treatment systems that could tackle “forever chemicals” in drinking water and polluted waterways alike.
Citation: Liu, F., Li, H., Gao, Z. et al. Steering charge transfer in CuInS2/BiOCl composites to enable sunlight-driven C–F bond cleavage of PFAS in water. Nat Water 4, 334–347 (2026). https://doi.org/10.1038/s44221-026-00590-4
Keywords: PFAS, water treatment, photocatalysis, sunlight remediation, environmental chemistry