Clear Sky Science · en
Microbial interaction networks as climate thermometers: redefining temperature sensitivity of aerobic methanotrophy in freshwater ecosystems
Why hidden lake microbes matter for climate
Most of us think of climate change in terms of smokestacks, cars, or melting ice. But a powerful, invisible player is at work beneath the surfaces of lakes and rivers: methane, a potent greenhouse gas, and the microbes that eat it. This study reveals how special methane-eating bacteria in freshwaters respond to warming across the globe, and shows that their relationships with other microbes—not just who is there or how many—can act like a biological thermometer for our warming planet.
Tiny methane eaters as a safety valve
Freshwater ecosystems, from tropical reservoirs to Arctic lakes, are now Earth’s largest natural source of methane. As temperatures rise, methane production in sediments speeds up, threatening to accelerate climate change. Standing in the way are methane-oxidizing bacteria (MOB), specialists that “burn” methane into carbon dioxide before it can escape to the air. These bacteria sit at the boundary between oxygen-rich and oxygen-poor waters and can remove between 10 and 90 percent of methane produced below. Yet until now, scientists had only a patchy view of where these microbes live, how diverse they are, and how strongly their methane-eating activity reacts to temperature around the world.
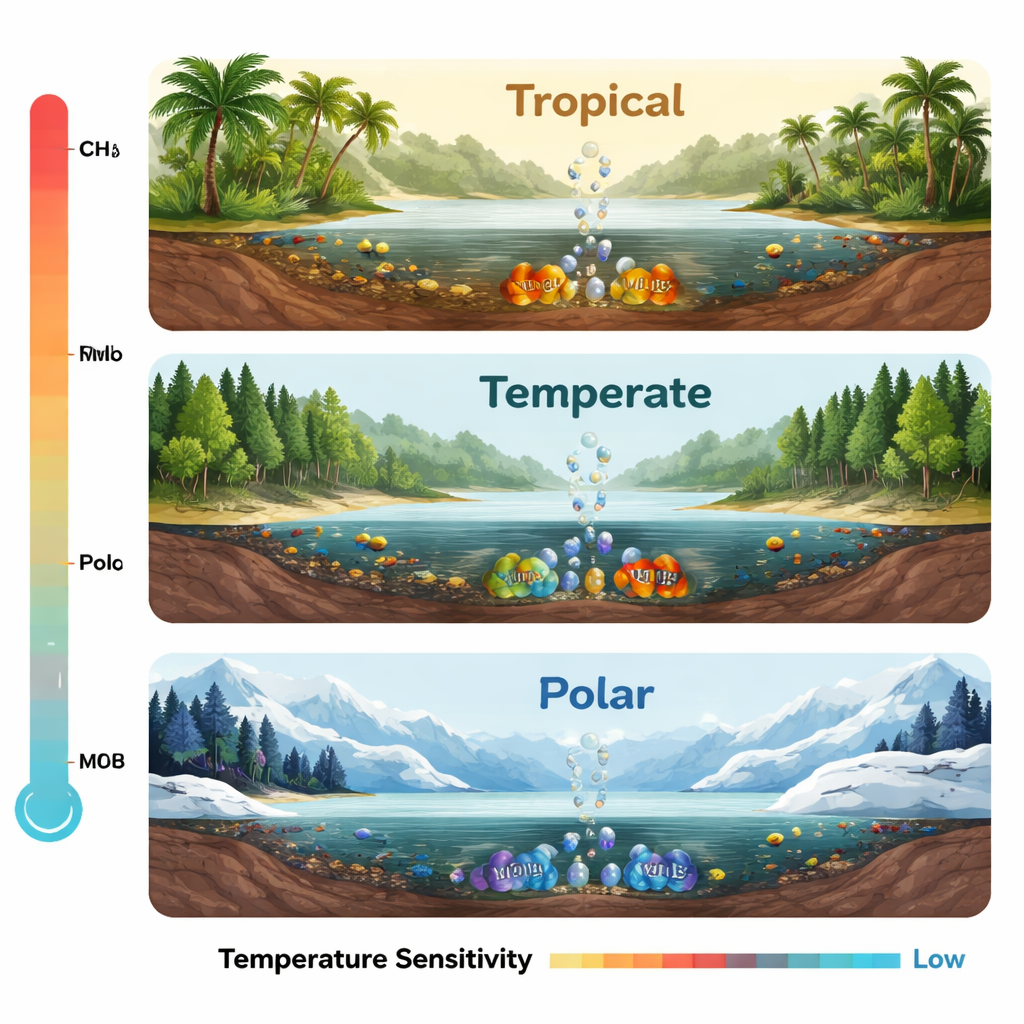
Who lives where: a global map of methane eaters
The authors assembled data from thousands of DNA samples taken from rivers, lakes, reservoirs, and estuaries worldwide, along with a huge genome catalog, to chart the global “biogeography” of MOB. They found clear patterns with latitude. In warm tropical and mid-latitude temperate waters, one major group, called type I MOB, dominates; these microbes are fast-growing “competitors” well-suited to abundant methane. Temperate regions, despite moderate abundances, host the richest and most diverse MOB communities. Near the poles, the balance flips: hardy type II MOB families, especially Beijerinckiaceae, take over. These cold-adapted “stress-tolerators” are better at making a living when energy is scarce and temperatures are low, and together they outnumber type I MOB in polar freshwaters.
How strongly warming revs up methane oxidation
To understand how sensitive this methane filter is to temperature, the team compiled measurements of methane oxidation rates from dozens of freshwater studies and compared them across tropical, temperate, and polar zones. They defined temperature sensitivity as how much the oxidation rate increases with each degree of warming. Surprisingly, the tropical region showed the strongest response: methane oxidation there rose sharply with temperature, followed by a moderate response in polar waters and the weakest in temperate systems. In other words, the microbial methane safety valve is most “thermally reactive” in the warmest regions, less so where seasons are pronounced, and somewhat reactive again in the cold.
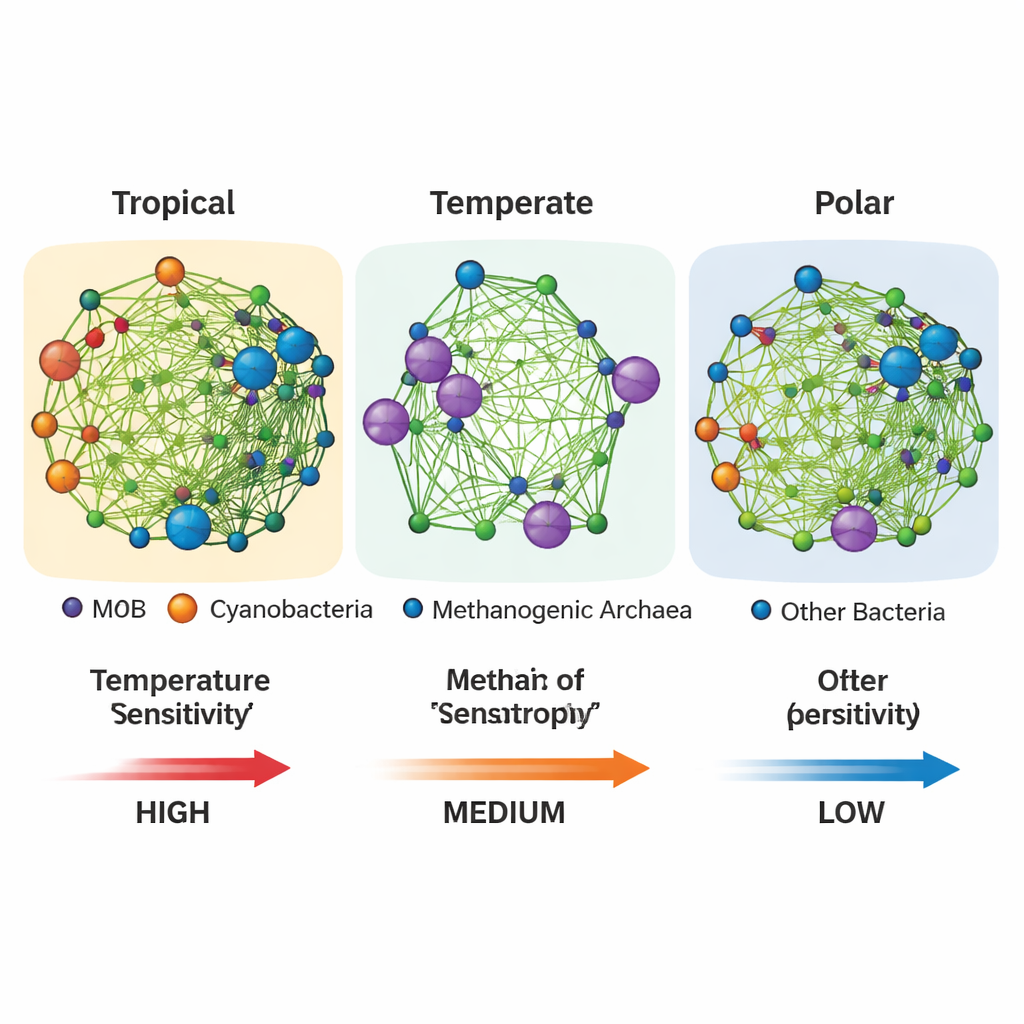
Networks, not headcounts, control climate response
The most striking result came when the authors treated microbes not as isolated species but as members of interaction networks. Using statistical tools, they reconstructed who tends to co-occur with whom and inferred webs of cooperation, signaling, and shared resources around methane-oxidizing bacteria. Across all bacteria, temperate waters showed the most tightly knit overall networks. But when the authors zoomed in on the subnetworks directly linked to methane oxidizers, a different picture emerged: in tropical and polar regions, these methane-centered subnetworks were denser, more tightly connected, and dominated by positive relationships, such as cross-feeding of nutrients and oxygen exchange with photosynthetic cyanobacteria. These positive ties amplify how quickly methane oxidation ramps up with warming. In temperate regions, by contrast, the methane-focused subnetworks were more fragmented and isolated from the rest of the community, and the temperature response was weaker.
Lessons from Earth’s deep past
To place today’s patterns in context, the study looks back over billions of years. Methane-producing microbes arose early in Earth’s history, and methane-eating bacteria and oxygen-producing cyanobacteria later reshaped the atmosphere. The authors argue that shifts in who partners with whom—methane oxidizers teaming first with cyanobacteria, then with methane producers, and now forming new alliances in modern lakes—have repeatedly influenced global temperatures. As warming continues, strengthened ties between methane oxidizers and cyanobacteria, especially in surface waters, could create new local methane cycles that either curb or enhance emissions, depending on how these networks reorganize.
What it means for future climate
For nonspecialists, the key takeaway is that the climate impact of lakes and rivers cannot be predicted from methane-producing microbes alone, nor simply by counting methane-eating bacteria. Instead, the strength and structure of the relationships among these microbes—who cooperates with whom, how tightly they are connected, and how quickly they respond together—act as a “climate thermometer” that controls how much methane reaches the air as the planet warms. By incorporating these interaction networks into climate models, scientists can better forecast future methane emissions and identify where protecting or restoring freshwater ecosystems might most effectively slow climate change.
Citation: Tang, Q., Lu, L., Xiao, Y. et al. Microbial interaction networks as climate thermometers: redefining temperature sensitivity of aerobic methanotrophy in freshwater ecosystems. npj biodivers 5, 8 (2026). https://doi.org/10.1038/s44185-026-00120-1
Keywords: methane-oxidizing bacteria, freshwater methane emissions, microbial interaction networks, climate feedbacks, aerobic methanotrophy