Clear Sky Science · en
Photo-activated ultrasound localization imaging with laser-activated nanodroplets
Sharper Views of Tiny Blood Vessels
Doctors and researchers increasingly rely on ultrasound to look inside the body in real time. But when it comes to the tiniest blood vessels, today’s scanners can blur important details. This study introduces a new way to "switch on" ultrasound contrast inside the bloodstream using light-controlled nanodroplets, opening the door to clearer images of the brain and other organs, longer scanning sessions, and potentially safer, more precise image-guided treatments.
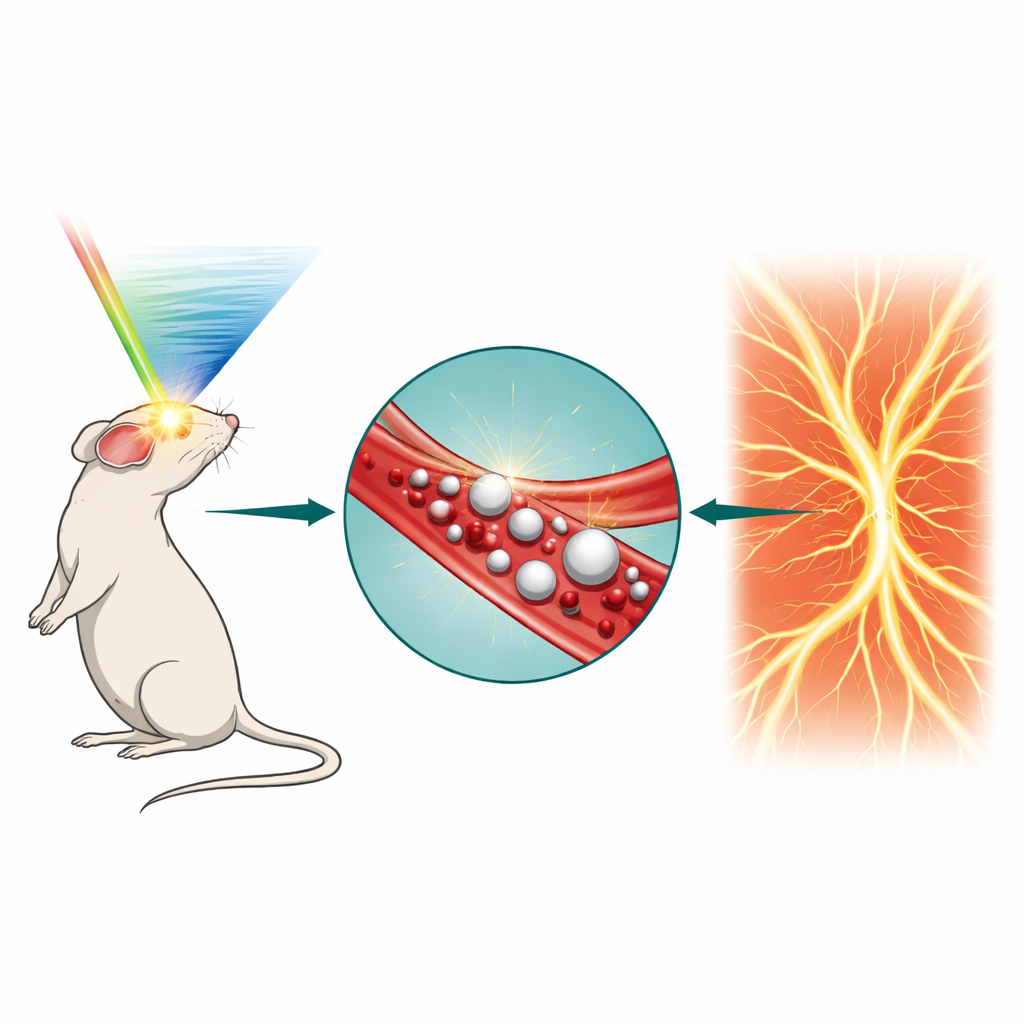
Why Seeing Small Vessels Is So Hard
Conventional ultrasound works well for larger structures, but its resolution is limited by the physics of sound waves: objects smaller than roughly half the ultrasound wavelength blur together. A recent advance, called ultrasound localization imaging, gets around this by tracking individual microbubbles injected into the bloodstream and building a super-sharp map of vessels from their paths, a bit like tracing city streets by following thousands of cars at night. However, these microbubbles are relatively large, circulate for only a few minutes, do not distribute evenly in small vessels, and their signal fades quickly, especially during long or repeated scans. These drawbacks limit how thoroughly and how long doctors can examine delicate microvascular networks in the brain, tumors, or kidneys.
Lighting Up Nanodroplets on Demand
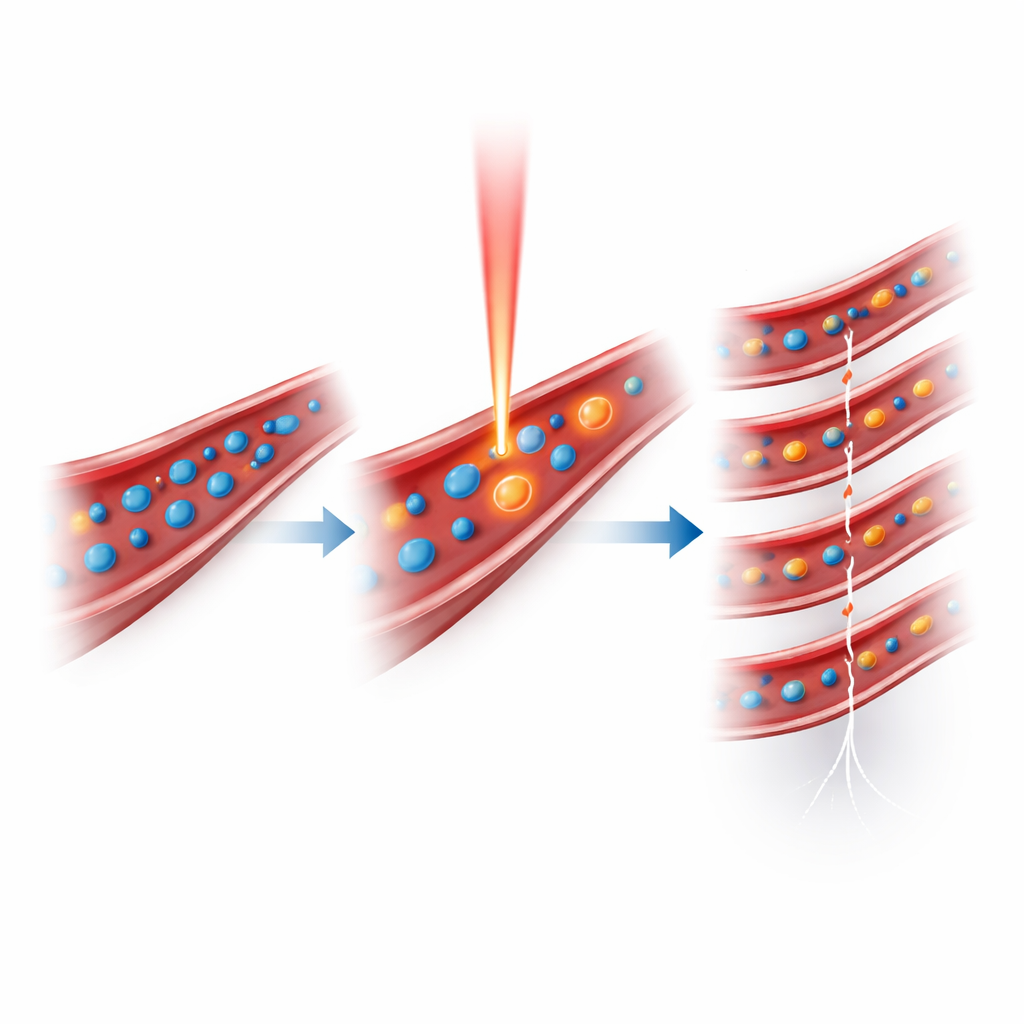
The authors tackled these limits by engineering tiny nanodroplets that can be triggered by short laser pulses to turn into microbubbles only when and where needed. Each droplet has a liquid core of perfluoropentane wrapped around a water-based solution of a light-absorbing dye (indocyanine green) and stabilized by a surfactant shell. At body temperature and normal ultrasound power, these droplets remain as harmless, stable nanoscale spheres that circulate for a long time. When a brief pulse of near-infrared laser light is aimed at the region of interest, the dye heats slightly, causing the core liquid to vaporize and inflate into a gas bubble that strongly reflects ultrasound. By adjusting the laser energy, the team could control how many droplets convert, achieving a sevenfold lower light dose than earlier light-activated droplet systems while still generating strong ultrasound and photoacoustic signals.
From Triggered Bubbles to Super-Resolved Maps
To turn this effect into a practical imaging method, the researchers built a setup that interleaves laser pulses with bursts of ultrafast ultrasound. After a single intravenous injection of nanodroplets in mice, the system repeatedly fired a low-rate laser pulse, then immediately captured hundreds of ultrasound frames as the newly formed microbubbles floated along the blood vessels. Using advanced filtering to remove background tissue signal, followed by localization algorithms, they pinpointed the position of individual bubbles frame by frame and stacked these positions over time into a fine-grained map of the microvasculature. In the mouse brain, this photo-activated ultrasound localization (PaUL) imaging revealed vessels down to about 21 micrometers—roughly a quarter the width of a human hair—through intact skin and skull, with sharper contrast than standard power Doppler ultrasound.
Faster Scans and Longer Imaging Windows
Because nanodroplets are much smaller than conventional microbubbles before activation, they can slip into finer capillaries and be triggered selectively in chosen regions. In head-to-head comparisons, PaUL imaging reconstructed detailed brain vessel networks about 2.4 times faster than regular microbubble-based localization imaging, thanks to the higher density of localized events in small vessels. The method also produced hemodynamic maps—showing blood flow speeds—comparable in accuracy to the standard technique, but with denser sampling and longer traceable paths for individual bubbles. Importantly, nanodroplets circulated for much longer: while microbubble signals fell off sharply within several minutes, nanodroplet-derived signals stayed strong beyond 20 minutes, enabling up to three times more localization events and allowing the researchers to scan multiple brain regions in sequence without reinjection.
Potential Uses and Future Improvements
These results suggest that light-activated nanodroplets can provide flexible, high-resolution imaging of tiny vessels over extended periods, which could be particularly valuable for studying brain function, monitoring stroke, or assessing tumor blood supply. The same droplets also generate photoacoustic contrast, allowing simultaneous mapping of oxygen levels and dye distribution alongside vessel structure and flow. The authors note that current performance is limited by how far light can penetrate tissue, confining the most efficient activation to depths of a few millimeters, but they outline several paths to go deeper: better light delivery geometries, dyes that absorb in more penetrating wavelength ranges, and minimally invasive fiber-based illumination. With future improvements and safety studies, PaUL imaging could complement existing ultrasound and photoacoustic tools, and may ultimately support image-guided therapies such as targeted drug delivery, where doctors selectively activate contrast or treatment agents only in the regions that need them.
What This Means for Patients
In simple terms, this work turns ultrasound imaging into something more like a controllable flashlight inside the bloodstream: tiny droplets stay quiet until a pulse of light tells them to "shine" for ultrasound. That control makes it possible to see smaller vessels more clearly, watch blood flow for longer, and potentially guide precision treatments with fewer injections and lower energy levels. While further testing is needed before use in people, the approach points toward safer, more informative scans of the body’s smallest and most important highways—the microvessels that feed our organs and tumors alike.
Citation: Zhao, S., Yi, J., Qiu, Y. et al. Photo-activated ultrasound localization imaging with laser-activated nanodroplets. Commun Eng 5, 43 (2026). https://doi.org/10.1038/s44172-026-00592-w
Keywords: ultrasound imaging, microvasculature, nanodroplets, photoacoustic imaging, brain blood flow