Clear Sky Science · en
Attenuation of epicardial activation and myofibroblast abundance via the Fbln2–Nupr1b axis stimulates cardiac regeneration in zebrafish
Why some hearts can heal themselves
When a person has a heart attack, the damaged muscle is usually patched over with a stiff scar that never truly heals. Zebrafish, small striped aquarium fish, do something remarkable instead: they regrow their heart tissue and eventually clear most of the scar. This study asks a deceptively simple question with big medical implications: how does a healing heart know when to build scar tissue and when to remove it so new muscle can grow? By decoding this balance in zebrafish, the work points toward strategies that might one day help injured human hearts repair themselves rather than fail.
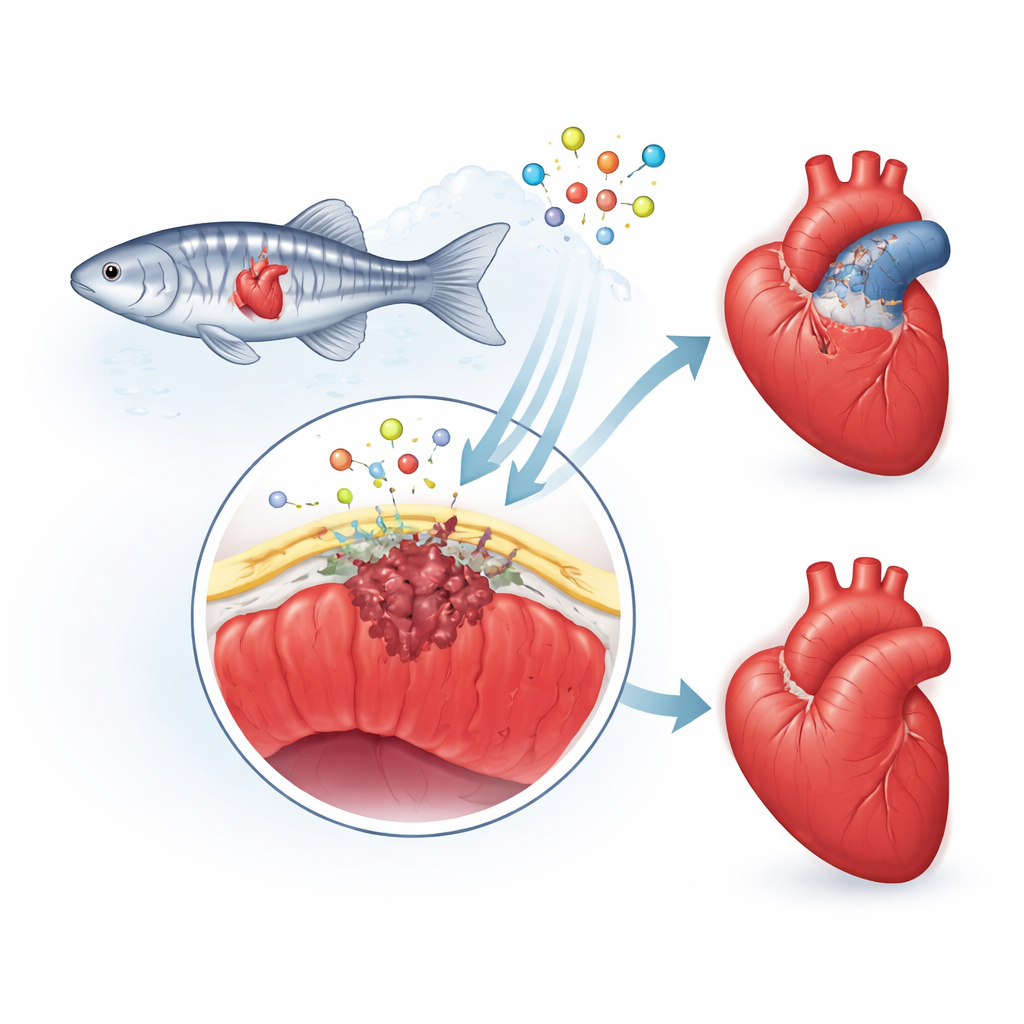
The heart’s protective outer skin
The researchers focus on the epicardium, a thin “skin” covering the heart. After injury, this layer wakes up, its cells change identity, and some dive into the damaged area. There they become support cells and scar-forming cells, and they release chemical signals that guide new blood vessels and heart muscle growth. In people, this repair response tends to overshoot, leaving behind a thick, permanent scar. In zebrafish, however, the scar is temporary and is gradually taken apart as healthy muscle returns. The team suspected that a protein called fibulin-2 (Fbln2), which lives in the heart’s surrounding scaffold, helps set the pace and intensity of this epicardial response.
A molecular dimmer switch on scarring
Using gene-editing in zebrafish, the scientists created two kinds of animals with different levels of Fbln2: one strain with reduced Fbln2 and another with the protein entirely removed. They injured the fish hearts with a freezing probe, mimicking a heart attack, and followed healing over weeks to months. When Fbln2 levels were lowered, early cell division in heart muscle and blood vessel lining dipped, but the fibrous scar that formed was smaller and more easily remodeled. By three months, these hearts had regenerated well and retained little collagen, the main scar material. In contrast, fish that completely lacked Fbln2 showed a stronger shutdown of a key growth-and-scarring pathway, involving a family of signals known as TGFβ, and had far fewer scar-forming cells right under the epicardium. Their early scars were also smaller—yet later on, these hearts failed to clear collagen and ended up with large, lasting scars and poor regeneration.
Zooming in on epicardial state changes
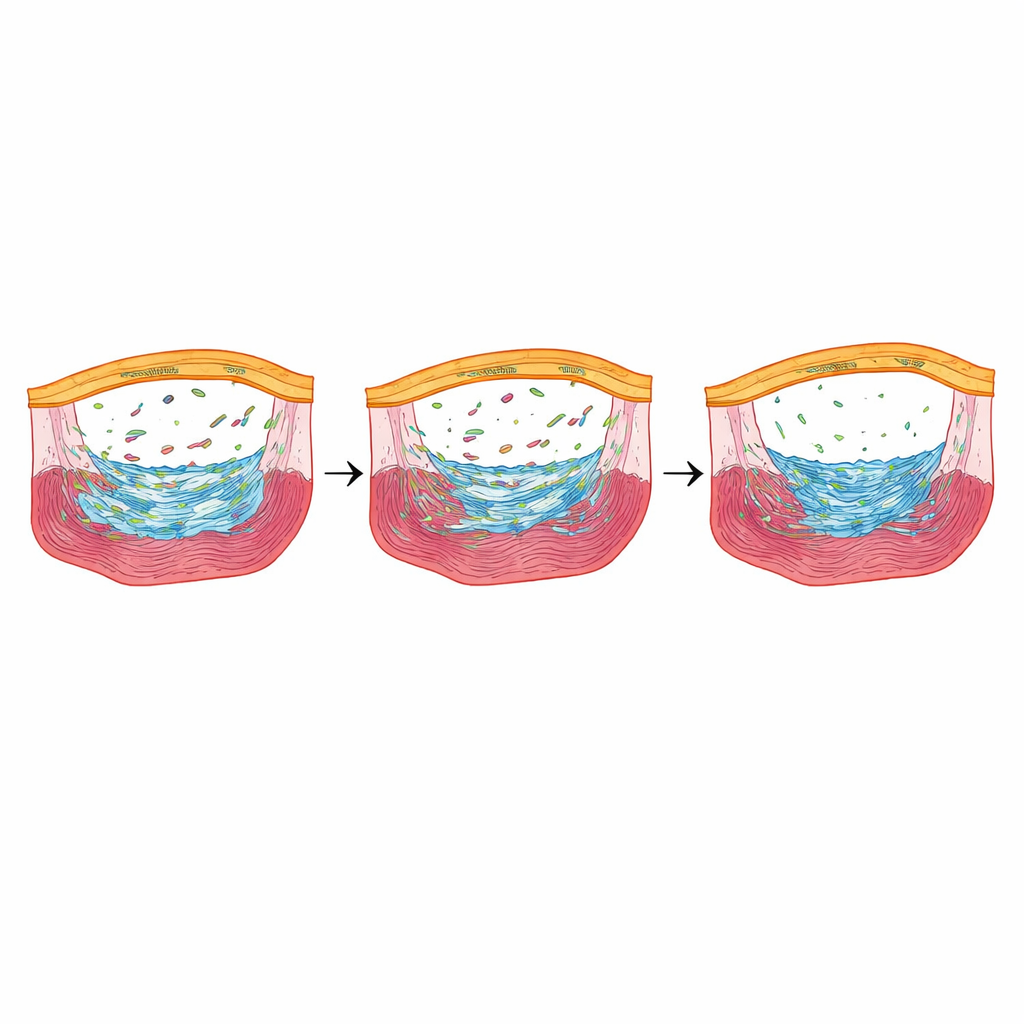
To see how epicardial cells were behaving at single-cell resolution, the team used single-cell RNA sequencing shortly after injury. They identified several epicardial cell “states,” including quiet cells, newly activated cells, stressed cells adapting to low oxygen, and cells that were actively dividing. In fish with normal Fbln2, epicardial cells smoothly progressed from quiet to activated to mature states. In fish with reduced or absent Fbln2, this progression stalled: cells lingered in early stages and the more mature activated groups were underrepresented, especially in the complete-loss animals. Multiple genes that normally turn on during epicardial activation and in response to TGFβ signaling were dialed down, pointing to Fbln2 as a tuner of this pathway that decides how strongly the epicardium commits to forming and remodeling scar tissue.
A stress responder that fine-tunes repair
Among the genes most sharply reduced when Fbln2 was lowered, one stood out: nupr1b, a stress-responsive regulator active in epicardial cells during early healing. When the researchers deleted nupr1b, zebrafish showed fewer epicardial scar-forming cells and reduced heart muscle cell division, and they retained large collagen deposits months after injury—much like fish lacking Fbln2 entirely. Strikingly, when they briefly forced nupr1b back on only in epicardial cells of Fbln2-reduced fish, the number of scar-forming cells and the proliferation of heart muscle cells bounced back, and late scarring increased. Blocking TGFβ receptors chemically reduced nupr1b levels, placing nupr1b downstream of a Fbln2–TGFβ signaling axis.
What this means for healing the human heart
Together, these findings reveal an “epicardial Fbln2–Nupr1b axis” that helps zebrafish hearts walk a tightrope between too little and too much fibrosis. Turning this axis down slightly tames the early burst of scar and encourages efficient scar removal, while turning it off completely derails regeneration and leaves a rigid scar behind. For human medicine, the lesson is that the goal is not to block scarring outright, but to tune it—keeping enough structure to prevent rupture while ensuring that scar tissue is temporary and can be replaced by healthy muscle. Understanding and eventually harnessing this zebrafish control system could inform future therapies that coax the injured human heart toward true regeneration instead of chronic scarring.
Citation: Kayman Kürekçi, G., Kaur Bajwa, G., Zhang, S. et al. Attenuation of epicardial activation and myofibroblast abundance via the Fbln2–Nupr1b axis stimulates cardiac regeneration in zebrafish. Nat Cardiovasc Res 5, 218–233 (2026). https://doi.org/10.1038/s44161-026-00785-8
Keywords: cardiac regeneration, fibrosis, zebrafish, epicardium, TGF-beta signaling