Clear Sky Science · en
Machine learning–based cfDNA fragmentation profiling using automated capillary electrophoresis for early detection of hepatocellular carcinoma
Why this matters for people with liver disease
For millions of people living with chronic hepatitis or liver scarring, the biggest danger is a silent one: liver cancer that grows unnoticed until treatment options are limited. This study presents a blood-based test, called CEliver, that aims to spot liver cancer early by reading subtle patterns in tiny bits of DNA circulating in the bloodstream. Because it uses equipment that many hospitals already own and avoids expensive genome sequencing, it could make advanced cancer screening more accessible in everyday clinics.
Tiny DNA clues floating in blood
Our blood carries small fragments of DNA released when cells die and break apart. In healthy people, most of these fragments are similar in size, but cancer cells tend to shed shorter, more irregular pieces. The researchers focused on hepatocellular carcinoma, the most common form of primary liver cancer, which often arises in people with long-standing liver disease such as chronic hepatitis B. They reasoned that if they could measure the size distribution of these DNA fragments precisely, they might be able to distinguish people with early liver cancer from those who are simply at high risk.
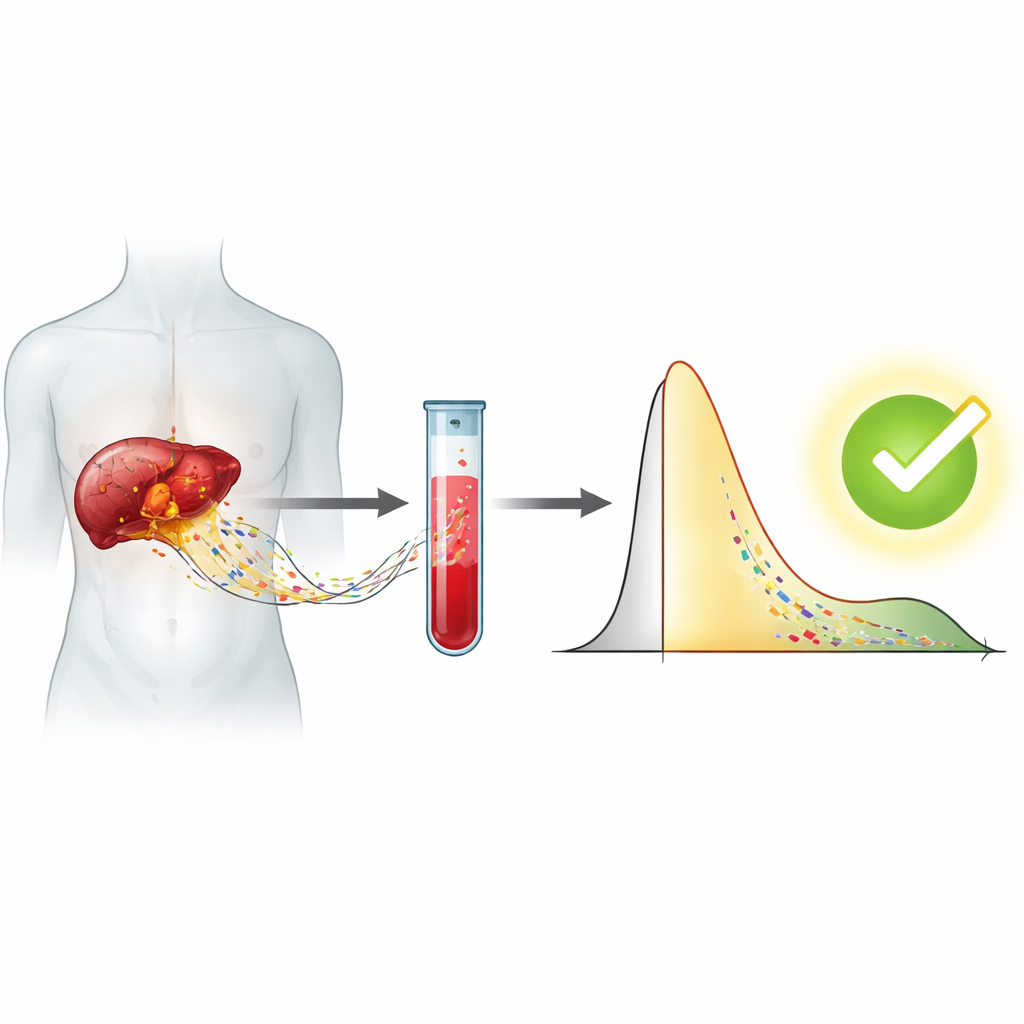
Turning a standard lab machine into a smart detector
Instead of relying on whole-genome sequencing, the team used automated capillary electrophoresis, a routine lab technique that separates DNA by size and reports the results as a curve showing how much DNA appears at each length. From each blood sample, they generated a detailed profile of cell-free DNA, including overall DNA concentration, the most common fragment size, and how strongly the signal appeared in 20 size “windows” between about 50 and 250 base pairs. They then built more than 300 numerical features describing the balance of short versus long fragments in many different ways, capturing subtle shifts that might be missed by a single summary measure.
Teaching a model to spot early liver cancer
To turn these patterns into a practical test, the researchers combined the fragment features with levels of alpha-fetoprotein, a blood marker already used for liver cancer screening, and trained a machine-learning model called CEliver. They developed this model using samples from 111 people: 71 high-risk individuals with chronic liver disease but no cancer, and 40 patients with hepatocellular carcinoma at different stages. The model learned which combinations of fragment patterns and alpha-fetoprotein values best separated the two groups. In this development set, CEliver correctly identified 98% of liver cancer cases overall and 96% of early-stage cases, while misclassifying only 1% of high-risk but cancer-free individuals.

Testing performance in real-world-like conditions
The team then challenged the fixed CEliver model on an independent group of 69 people who had not been used in training: 27 with liver cancer, 30 at high risk, and 12 healthy volunteers. Using a single cutoff score, the model detected 85% of all liver cancer patients and 88% of those in the earliest stage, while correctly labeling every high-risk and healthy person as cancer-free. By comparison, the standard blood marker alone detected only about half of cancer cases and missed most early-stage tumors, especially those smaller than 2 centimeters. In several patients, CEliver flagged likely cancer months before imaging confirmed a tumor, suggesting that fragment patterns may change before growths are clearly visible.
What this could mean for patients
The study shows that a relatively simple blood test, built on widely available lab hardware and advanced data analysis, can detect liver cancer at an early, more treatable stage with high accuracy. For people living with chronic hepatitis or liver scarring, this approach could eventually offer a more sensitive and scalable way to monitor for cancer than today’s ultrasound scans and single blood markers. While larger and more diverse studies are still needed, CEliver points toward a future where careful reading of cell-free DNA patterns becomes a routine part of protecting high-risk patients from one of the deadliest liver diseases.
Citation: Udomruk, S., Sutthitthasakul, S., Bunsermvicha, N. et al. Machine learning–based cfDNA fragmentation profiling using automated capillary electrophoresis for early detection of hepatocellular carcinoma. Commun Med 6, 166 (2026). https://doi.org/10.1038/s43856-026-01437-5
Keywords: liver cancer screening, cell-free DNA, hepatocellular carcinoma, early cancer detection, machine learning diagnostics