Clear Sky Science · en
Selective blockade of latent TGF-β1 activation suppresses tissue fibrosis with good safety
Why hidden scarring matters
Many chronic diseases quietly damage our organs by laying down stiff scar tissue, a process called fibrosis. This scarring can slowly choke off normal function in the liver, kidneys, lungs, and even around tumors, where it can also blunt the effects of modern cancer immunotherapies. The protein TGF-β1 sits at the heart of this scarring process, but shutting it off everywhere in the body has proved dangerous. This study describes a new antibody, called SOF10, designed to block only the harmful activation of TGF-β1 while preserving its essential roles, showing promising benefits in animal models of liver disease, kidney failure, and cancer.
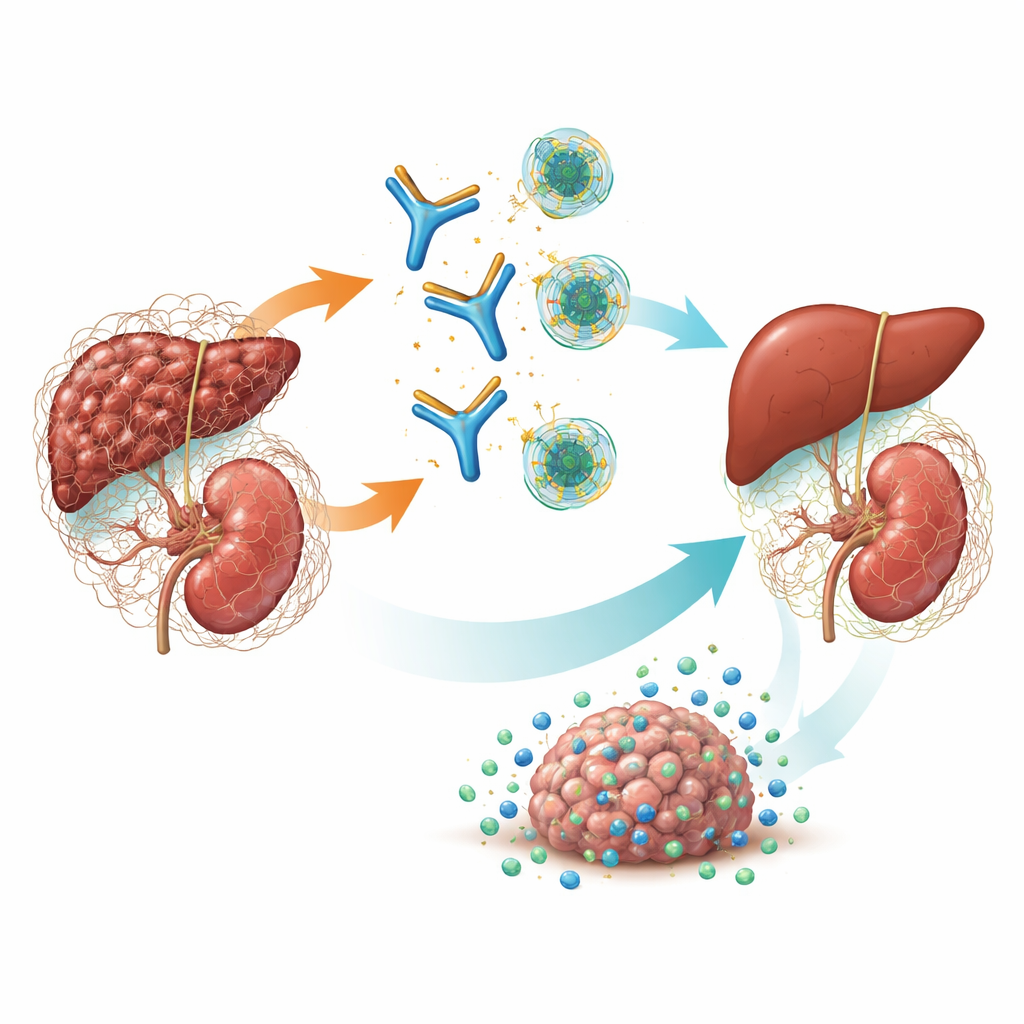
A master switch for scarring and cancer
TGF-β is a powerful signaling molecule that helps control how cells grow, heal, and interact with the immune system. In chronic disease, one form—TGF-β1—drives fibroblasts to build excess connective tissue, leading to organ stiffening and failure. It also shapes an immune-suppressive, fibrotic shell around tumors that keeps immune cells and drugs from reaching cancer cells. Earlier drugs tried to shut down all three TGF-β forms at once, but this “pan-blockade” caused serious side effects such as heart valve damage, bleeding, and skin tumors. Genetic studies and prior models suggested that TGF-β1 is the main culprit in fibrotic kidney and many solid tumors, raising the possibility that carefully targeting just this isoform could be both effective and safer.
Locking TGF-β1 in its inactive form
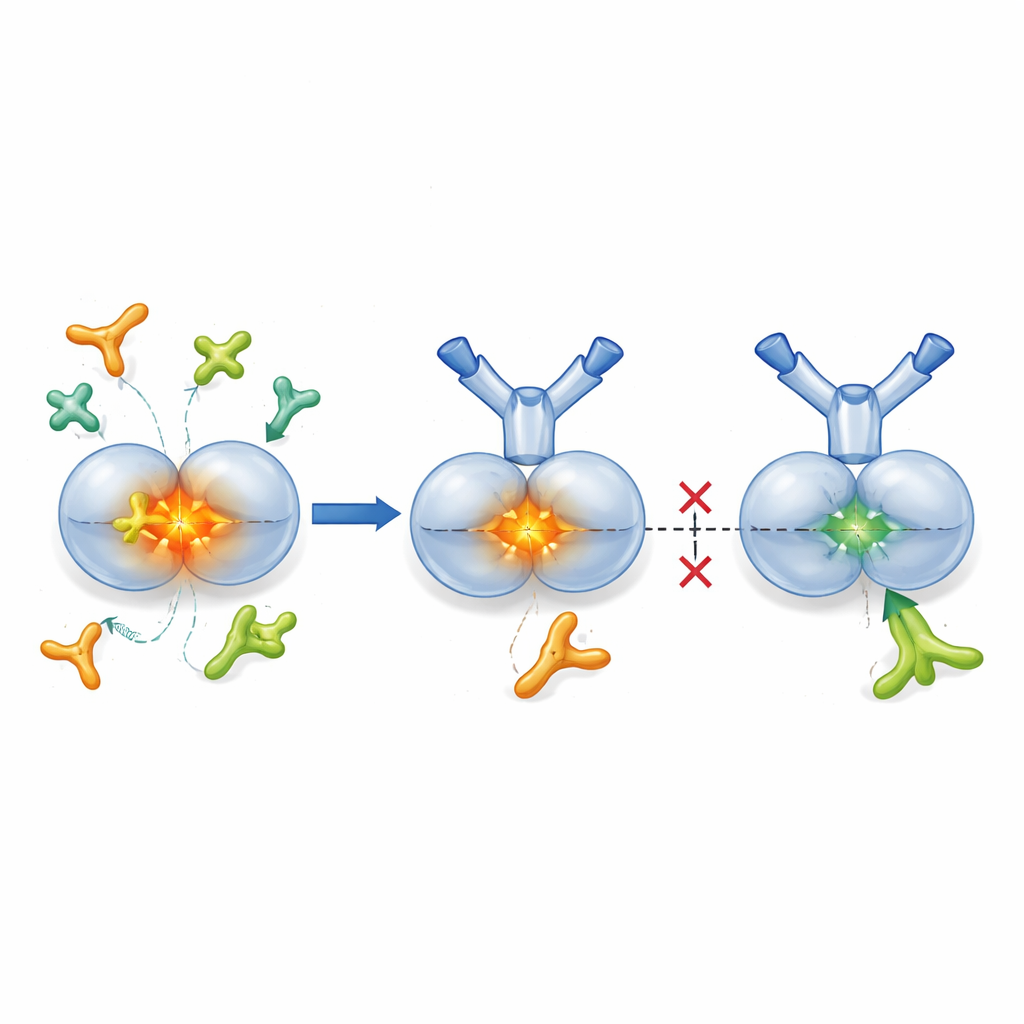
TGF-β1 is normally produced in a “latent” package, where the active core is wrapped by a partner protein that keeps it off until needed. Enzymes called proteases and certain cell-surface gripping proteins known as integrins can open this package, releasing active TGF-β1 into nearby tissue. The researchers engineered SOF10, a humanized antibody that binds only to the latent form of TGF-β1, not the active hormone or other isoforms. Structural studies using X-ray crystallography showed that SOF10 wedges into the interface between two halves of the latent complex, stabilizing it like a clamp. In lab tests, this prevented proteases and one integrin (αvβ8) from freeing active TGF-β1, yet left another integrin (αvβ6)—important for maintaining immune balance in healthy tissues—largely untouched. In effect, SOF10 selectively silences the damaging activation routes while sparing a key homeostatic pathway.
Protecting liver and kidneys from slow damage
To see whether this selective clamp on TGF-β1 could actually limit scarring, the team tested SOF10 in several mouse models. In a rapid diet-induced model of fatty liver disease that progresses to liver fibrosis, SOF10 treatment reduced the liver’s expression of TGF-β–responsive genes and collagen-building genes, and it lowered hydroxyproline, a chemical marker of accumulated scar tissue. In two different kidney models—a surgical obstruction model that drives interstitial fibrosis and a genetic model of Alport syndrome that develops both glomerular scarring and kidney failure—SOF10 cut fibrotic gene activity, reduced collagen content, and visibly shrank scarred areas under the microscope. In the chronic Alport model, blood tests showed improved kidney function, and these benefits were on par with a broader TGF-β–blocking antibody, but without blocking other TGF-β isoforms.
Freeing the immune system inside tumors
Because fibrotic tissue around tumors can act as both a physical barricade and an immune brake, the researchers asked whether SOF10 could improve responses to checkpoint inhibitor drugs. In mouse breast and colon cancer models that normally respond poorly to anti–PD-L1 therapy, adding SOF10 significantly slowed tumor growth, whereas SOF10 or anti–PD-L1 alone had little effect. Tumors from combination-treated animals contained more killer CD8 T cells and more of the toxic enzymes they use to destroy cancer cells. Fibroblasts taken from these tumors showed lower activity in collagen and matrix-building pathways but higher activity in interferon and antigen-presentation pathways, a shift toward a more immune-supportive stroma. These changes closely mirrored what had been seen with broad TGF-β blockade, suggesting that targeting latent TGF-β1 alone can reprogram the tumor environment in a similar way.
Safety signals in animals
Because earlier attempts to block TGF-β broadly ran into safety problems, the team carried out 13-week toxicity studies in mice and cynomolgus monkeys, giving repeated high doses of SOF10. The drug behaved like a typical antibody in the bloodstream, and across a wide dose range there were no signs of damage in major organs, blood cell counts, or heart and lung function. The highest doses tested—up to 200 mg/kg every two weeks in mice and 100 mg/kg in monkeys—were designated as levels with no observed harmful effects. This safety profile supports the idea that sparing TGF-β2, TGF-β3, and αvβ6-mediated activation reduces the risks seen with earlier, less selective approaches.
What this could mean for patients
Taken together, the findings suggest that locking TGF-β1 in its inactive shell—rather than wiping out all TGF-β signaling—can ease scarring in multiple organs, improve kidney function, and make stubborn tumors more vulnerable to immunotherapy, all with encouraging safety in animal tests. While human trials are needed to confirm these benefits and monitor long-term effects, SOF10 and similar strategies may open a new way to treat chronic fibrotic diseases and certain cancers, turning down harmful scarring while preserving the healing and immune-balancing roles our tissues still require.
Citation: Kanamori, M., Sato, I., Koo, C.X. et al. Selective blockade of latent TGF-β1 activation suppresses tissue fibrosis with good safety. Commun Med 6, 131 (2026). https://doi.org/10.1038/s43856-026-01408-w
Keywords: fibrosis, TGF-beta1, monoclonal antibody, kidney and liver disease, cancer immunotherapy