Clear Sky Science · en
Intraoperative biopsy imaging of lung cancer risk
Seeing Cancer’s Hidden Footprints
Lung cancer surgeons face a high‑stakes puzzle: during an operation, they must quickly find tiny tumors and their exact edges, yet today’s tools can miss dangerous spots or take too long. This study introduces a new way to "see" cancer by looking not at the tumor cells themselves, but at the microscopic scaffolding that surrounds them. By combining advanced laser imaging with artificial intelligence, the authors show it is possible to spot lung cancer and its boundaries rapidly and with remarkable accuracy.
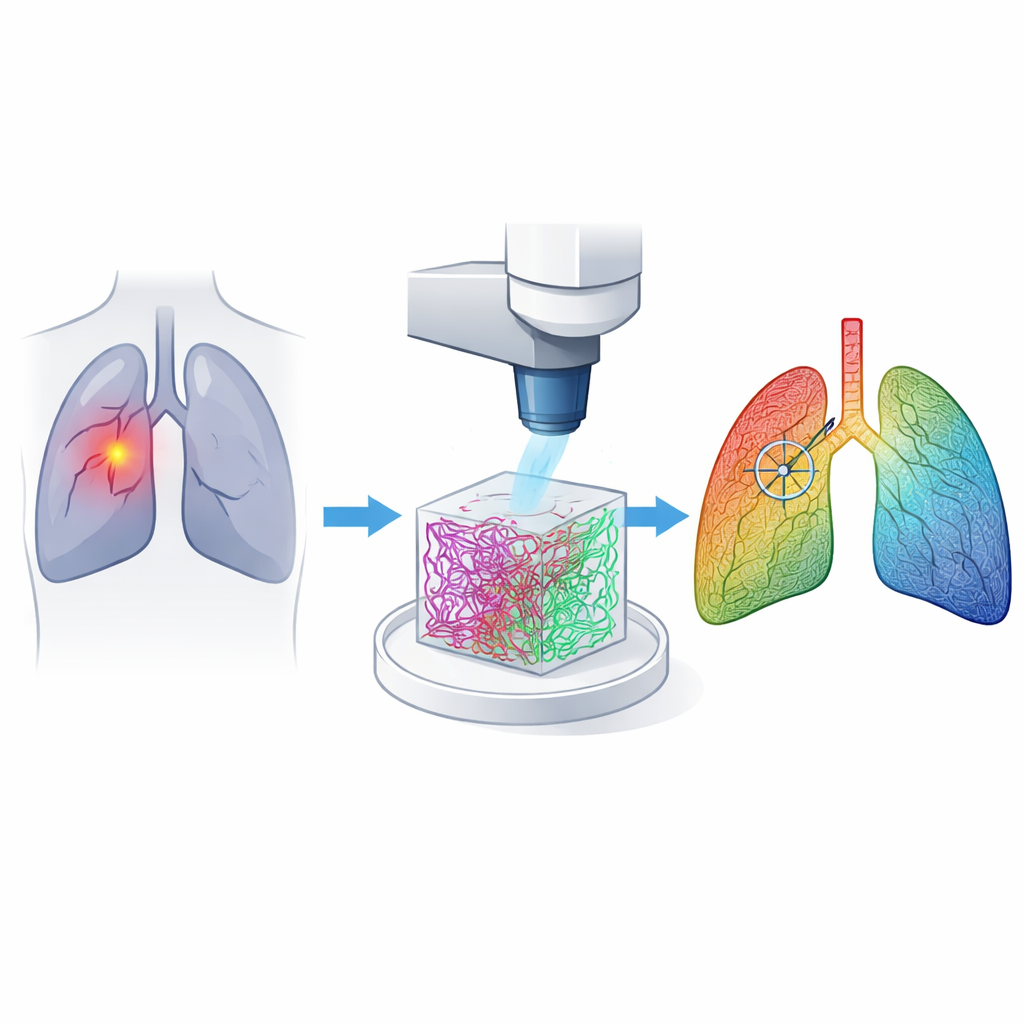
The Supporting Tissue That Tells a Story
Our lungs are held together by a mesh of protein fibers called the extracellular matrix. Two key players, collagen and elastin, give lung tissue strength and stretchiness. When cancer develops, these fibers are remodeled: they change how they are arranged, how dense they are, and how they interact with each other. The researchers reasoned that these subtle structural shifts could serve as reliable fingerprints of cancer, even when tumors are hard to see with ordinary surgical cameras.
A Gentle Laser Microscope in the Operating Room
The team built a custom multiphoton microscope, a type of laser system that can peer hundreds of micrometers into fresh tissue without adding any dyes. With a single near‑infrared laser setting, it records two signals at once: one that highlights collagen and another that highlights elastin. This dual imaging produces crisp 3D views of the lung’s fiber network within a couple of minutes per site, fast enough to be practical during surgery. The method was tested on tissue taken from 222 patients with lung adenocarcinoma, capturing hundreds of small 3D image stacks from both tumor and normal regions.
Teaching AI to Read the Fiber Code
Rather than judging these images by eye, the scientists measured each tiny volume element, or voxel, across several features: how aligned the fibers are, how wavy they appear, how densely they fill space, how thick elastin strands are, and how similar nearby collagen and elastin fibers are in position and direction. They introduced a new "similarity coefficient" that rises when collagen and elastin lie close together and point the same way. Using unsupervised learning, they grouped millions of voxels into recurring fiber "vocabularies" that capture common structural patterns. A standard machine‑learning classifier then learned how the mix of these vocabularies differs between normal and cancerous tissue.
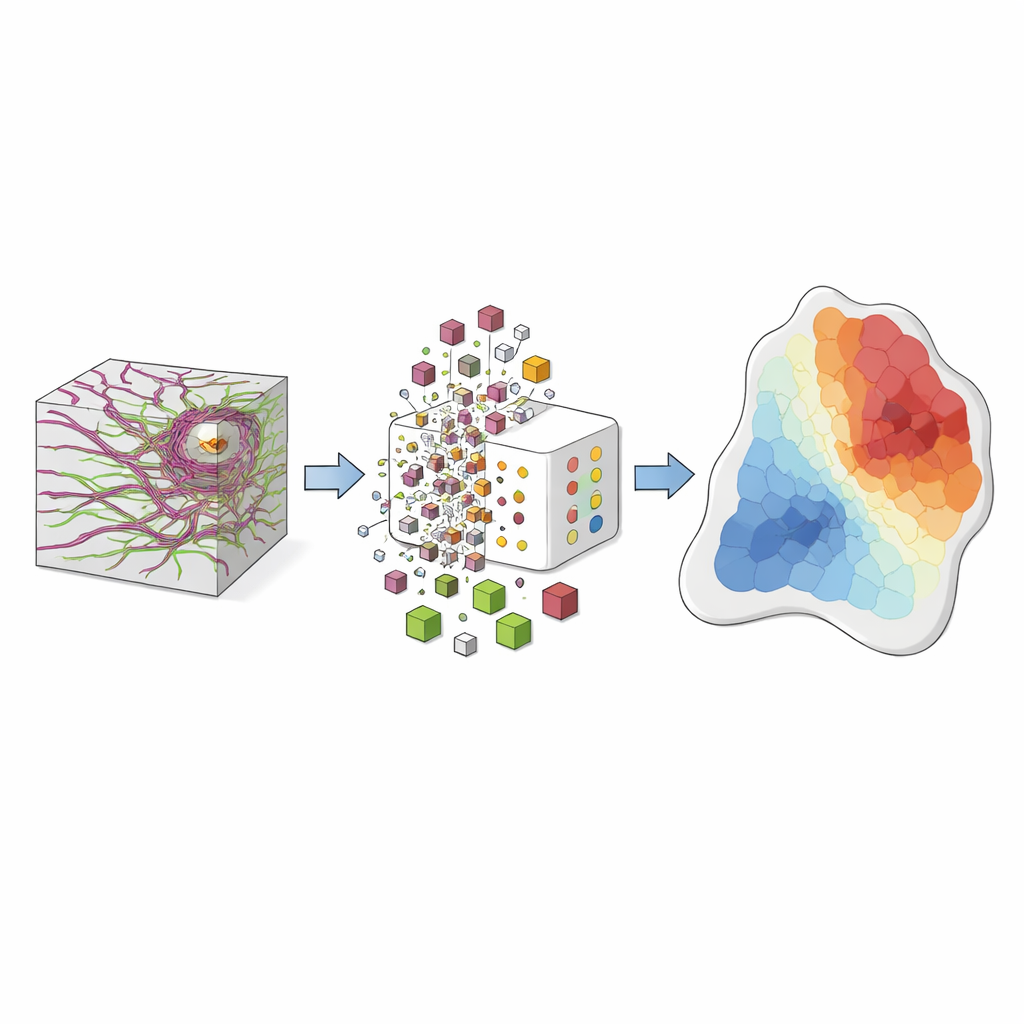
From Invisible Patterns to Risk Maps
When this framework was tested on an independent set of lung samples, it identified cancer with a sensitivity of about 99%, meaning it almost never missed a tumor. The researchers went further and created a "cancer risk index" for each fiber vocabulary, indicating whether it tends to appear in healthy or diseased tissue. By coloring every voxel according to this index, they generated intuitive risk maps: tumor regions glow in warm hues, normal areas in cool hues, and boundary zones show a mix. These maps successfully outlined the tumor‑normal border in human specimens and showed the same remodeling trends in a mouse model imaged live, suggesting the approach could translate into real‑time guidance in the operating room.
What This Could Mean for Patients
Instead of waiting for frozen‑section pathology, surgeons could one day scan suspicious lung areas with a small laser probe and immediately see where cancer is most likely hiding, as revealed by the reorganized collagen and elastin around it. This study shows that the tumor’s supporting framework carries a strong, readable signal of risk and that AI can turn that signal into clear visual cues. If developed into clinical tools, such imaging could make biopsies more accurate, reduce repeat surgeries, and help ensure that dangerous tissue is removed while preserving as much healthy lung as possible.
Citation: Qian, S., Yang, L., Meng, J. et al. Intraoperative biopsy imaging of lung cancer risk. Commun Med 6, 143 (2026). https://doi.org/10.1038/s43856-026-01406-y
Keywords: lung cancer surgery, intraoperative imaging, extracellular matrix, multiphoton microscopy, artificial intelligence diagnostics