Clear Sky Science · en
Pembrolizumab and epigenetic modification with azacitidine reshapes the tumor microenvironment of platinum-resistant epithelial ovarian cancer: a phase 2 non-randomized clinical trial
Why this study matters
For many women with advanced ovarian cancer, standard chemotherapy eventually stops working, leaving few good options. This study explores a new strategy: pairing an immune-based treatment with a drug that can "reprogram" cancer cells, in hopes of turning a quiet, hard-to-recognize tumor into one the immune system can see and attack. Understanding whether this approach is safe, how well it controls cancer growth, and what it does inside tumors could shape future treatments for one of the deadliest gynecologic cancers.
A hard-to-treat form of ovarian cancer
Ovarian cancer is often diagnosed at an advanced stage, and many tumors become resistant to a key chemotherapy drug, platinum. Once that happens, responses to further chemotherapy are usually short-lived. Immune checkpoint drugs, which release the brakes on immune cells, have transformed care in several other cancers but have had modest success in ovarian cancer. One reason is that many ovarian tumors are "cold": they contain relatively few active immune cells, making it difficult for checkpoint drugs to work. The researchers behind this trial asked whether they could warm up these tumors by first altering how genes are switched on and off inside cancer cells.
Pairing immune therapy with an epigenetic drug
The team tested a combination of pembrolizumab, a widely used immune checkpoint drug, with oral azacitidine, a medicine that affects the chemical tags on DNA that help control gene activity. By changing these tags, azacitidine can reawaken silenced genes and trigger antiviral-like alarm signals inside tumor cells. In laboratory models, this kind of "viral mimicry" attracts immune cells into previously quiet tumors. In this phase II clinical trial, 34 women with platinum-resistant or platinum-refractory epithelial ovarian cancer received pembrolizumab along with one of four dosing schedules of azacitidine. All had measurable disease and had already been through standard surgery and chemotherapy.
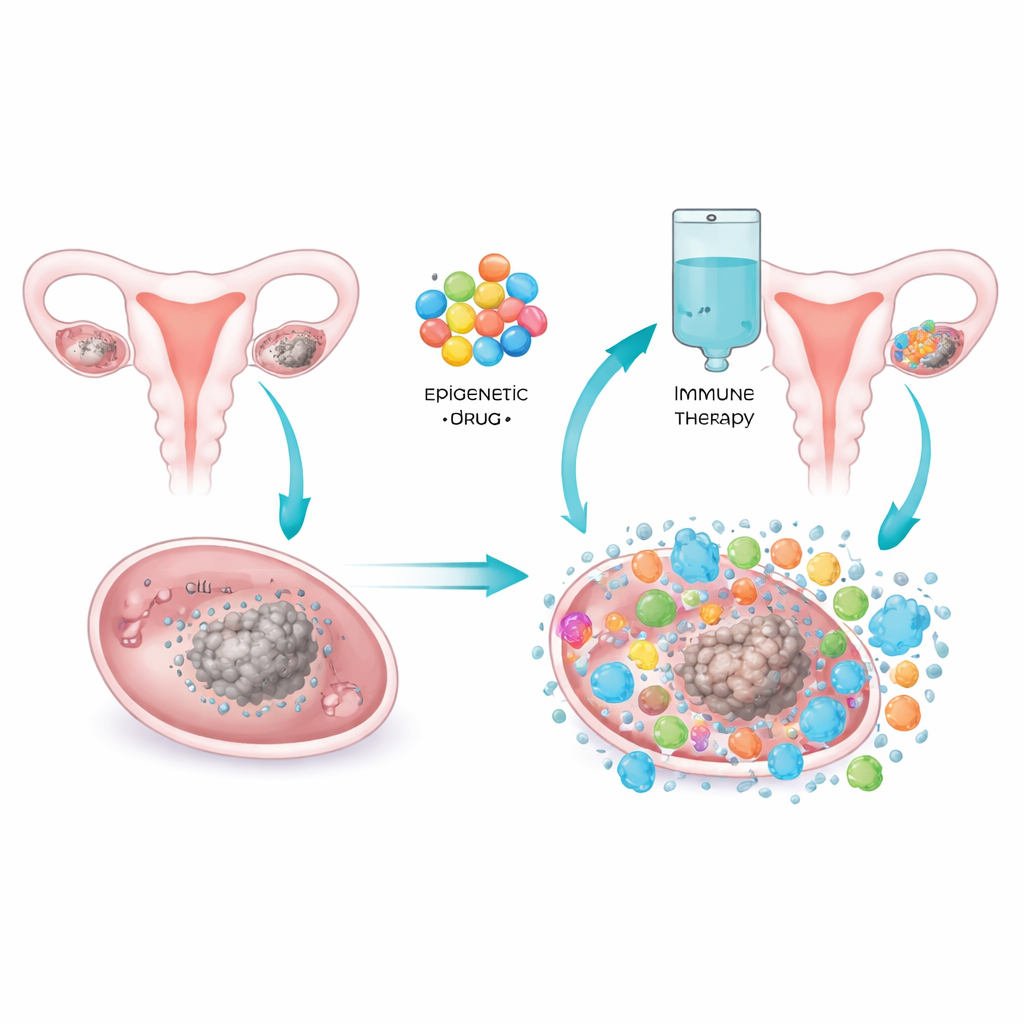
What happened in patients
The combination was considered moderately well tolerated. Common side effects included nausea, vomiting, diarrhea, fatigue, anemia, and low white blood cell counts, especially at higher azacitidine doses. Most of these issues were manageable with dose adjustments, and no unexpected safety problems emerged beyond what is known for each drug alone. In terms of tumor shrinkage, only one patient had a partial response, and none had a complete response, for an overall response rate of about 3%. However, about half of the women experienced either tumor shrinkage or stable disease, giving a disease control rate of 50%. A small number stayed on treatment for more than a year, including one woman who remained in remission after switching to pembrolizumab alone.
How the tumor environment was reshaped
To see what the drug pair was doing inside tumors, the researchers took tissue samples before treatment and again about six weeks after therapy began. They measured activity levels of hundreds of immune-related genes and sequenced RNA from the tumors to survey broader changes. On treatment, tumors showed higher activity in genes linked to inflammation, interferon signaling, and the machinery that displays tumor fragments to immune cells. Pathways that help immune cells move into and stick to tumors were also more active. At the same time, the relative number of tumor-invading T cells, especially the killer subtype, increased. These patterns suggest the tumor surroundings were shifting from a quiet state toward a more inflamed, immune-aware condition.
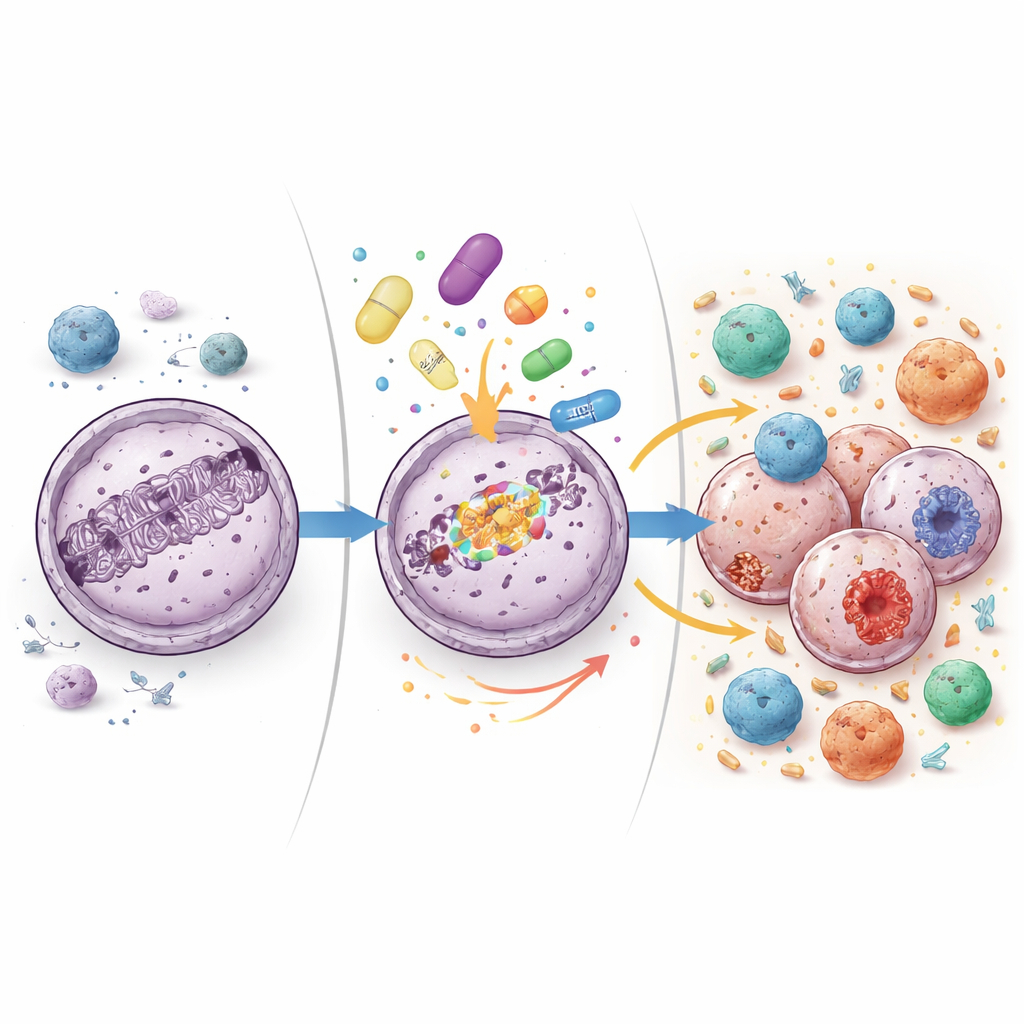
Clues from responders and immune cell fingerprints
The investigators also examined patterns in patients who showed signs of benefit, either through drops in the blood marker CA-125 or longer-lasting disease control. Tumors from these women tended to have higher baseline activity in inflammatory gene programs and, after treatment began, showed stronger boosts in adaptive immune responses—those driven by T and B cells that recognize specific targets. By reconstructing the rearranged receptors on T cells from tumor RNA, the team found that T cell populations inside tumors frequently shifted under treatment, with some specific cell clones expanding or shrinking. This remodeling of the T cell landscape is another sign that the drug combination was actively reshaping the immune battle within tumors.
What this means for patients
For women with platinum-resistant ovarian cancer, this particular pairing of pembrolizumab and oral azacitidine did not dramatically increase the number of tumors that shrank on scans compared with immune therapy alone. However, it did control disease in about half of the participants and produced clear molecular signs that tumors were becoming more inflamed and more densely populated by immune cells. To a lay reader, the takeaway is that the drugs appear capable of "waking up" the immune system inside these otherwise quiet tumors, even if this did not always translate into major shrinkage in this small, heavily pretreated group. The authors argue that refining drug dosing, improving how azacitidine is delivered, and using molecular markers to select patients more likely to respond could make this immune-warming strategy an important building block of future ovarian cancer treatments.
Citation: Landon, B.V., Boland, J.L., Wahner Hendrickson, A.E. et al. Pembrolizumab and epigenetic modification with azacitidine reshapes the tumor microenvironment of platinum-resistant epithelial ovarian cancer: a phase 2 non-randomized clinical trial. Commun Med 6, 142 (2026). https://doi.org/10.1038/s43856-026-01404-0
Keywords: ovarian cancer, immunotherapy, epigenetic therapy, tumor microenvironment, checkpoint inhibitors