Clear Sky Science · en
The clinical significance of the Mediterranean fever gene MEFV variants in Castleman disease
Hidden Clues in a Rare Immune Storm
Some people develop a dangerous, full‑body “cytokine storm” in which their immune system spirals out of control, damaging organs and threatening their lives. One rare example is a group of disorders called Castleman disease, and an especially severe subtype in young people known as TAFRO. This study explores whether subtle inherited changes in a single immune‑related gene, called MEFV, help trigger or worsen this storm—and whether understanding those changes can point to better treatments.
A Rare Teenager’s Life‑Threatening Illness
The researchers followed a previously healthy 15‑year‑old boy who suddenly developed high fever, belly pain, massive fluid buildup, kidney failure, and swollen lymph nodes and spleen. Tests showed very high levels of inflammatory substances in his blood, including a signaling protein called interleukin‑6 (IL‑6). A lymph node biopsy confirmed Castleman disease of the TAFRO subtype, a particularly aggressive form that is seldom seen in children. The team treated him with an antibody drug that blocks IL‑6 together with steroids and immune‑modifying medicines. Within a few months, his symptoms, organ damage, and abnormal scans all improved dramatically, and he remained in complete remission even after treatment was stopped.
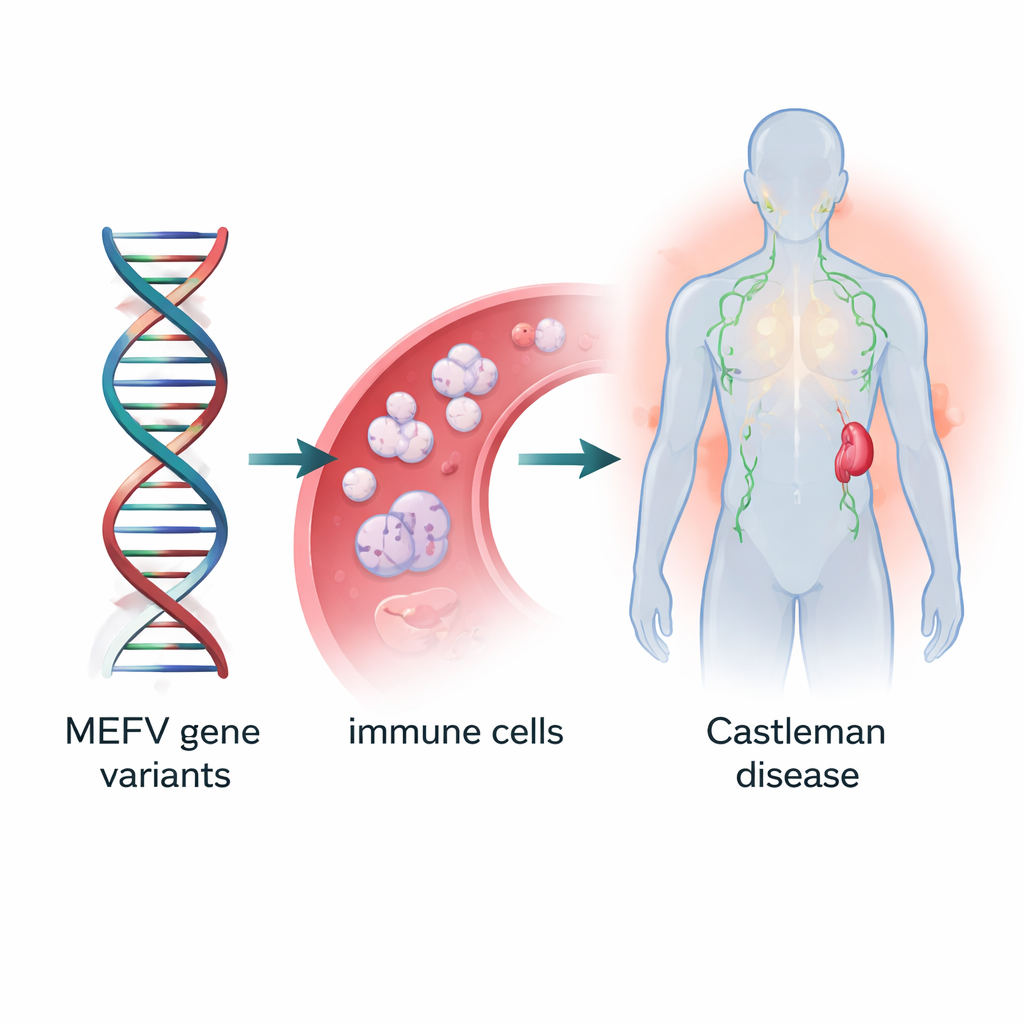
A Suspicious Gene in the Family
Because the cause of TAFRO and other Castleman disease types is still mysterious, the scientists sequenced the DNA from the boy, his parents, and later from tumor samples of 37 additional Castleman patients. They focused on the MEFV gene, already known for its role in another inflammatory illness called familial Mediterranean fever. The teenager carried a combination of three MEFV changes—named E148Q, P369S, and R408Q—on both copies of the gene, inherited from his symptom‑free parents who each carried fewer changes. In the larger group of 37 patients, three‑quarters had some MEFV variant, and about one in five carried the same triple combination. These variants were far more common than in the general East Asian population, suggesting they may help set the stage for Castleman disease.
How Gene Changes Push Immune Cells Into Overdrive
To understand how MEFV variants might translate into disease, the researchers isolated blood immune cells from the teenager, his parents, and a healthy person and exposed them to a bacterial component that strongly provokes inflammation. Cells from the boy, with the full triple‑variant MEFV, clumped more and produced much higher levels of IL‑6, another inflammatory messenger called IL‑1β, and several chemical “homing signals” (chemokines) than cells from the others. When they added colchicine, a long‑used drug for gout and familial Mediterranean fever, these cells released fewer inflammatory substances and showed less clumping, hinting that colchicine or related medicines might help calm similar storms in Castleman disease.
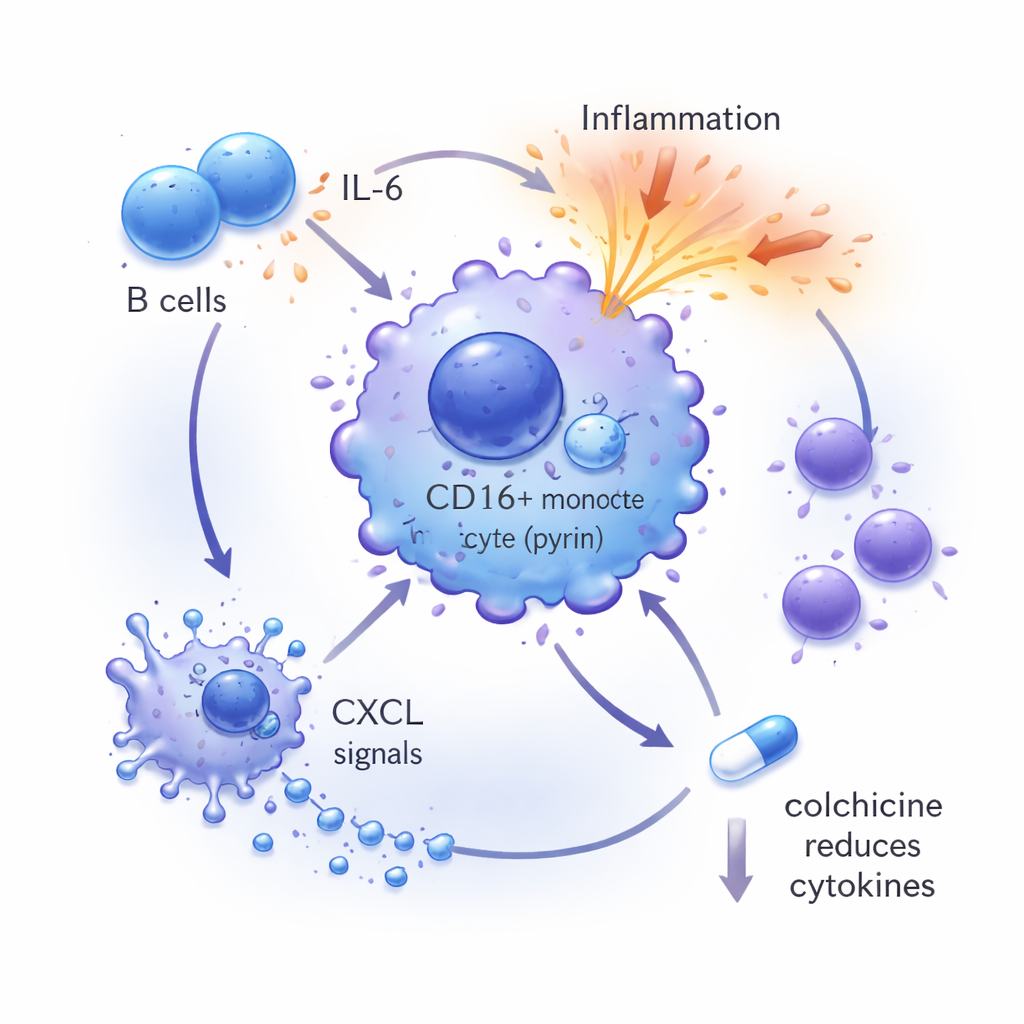
Zooming In on Single Cells
The team then used single‑cell RNA sequencing, a technique that reads the activity of thousands of genes in individual cells, to build a detailed map of the boy’s immune system during a disease flare and after recovery. They found that MEFV was most active in a subset of blood cells called CD16‑positive monocytes. During the flare, these cells showed strong signatures of inflammation and IL‑6‑related activity, and they appeared to be heavily influenced by signals from nearby B cells, which were themselves making a lot of IL‑6. Another cell type, megakaryocytes—best known for producing platelets—was unusually frequent and seemed to send out powerful chemokine signals that could further attract and activate other immune cells. After successful treatment, these abnormal patterns largely returned toward normal.
What This Means for Patients
This work suggests that inherited MEFV variants do not act alone to cause Castleman disease, but they may prime key immune cells to overreact to triggers and fuel a dangerous cytokine storm, especially in TAFRO. Blocking IL‑6 can be highly effective, as seen in the adolescent patient, and drugs like colchicine that target MEFV‑related pathways might offer an additional way to dial down the inflammation in some patients. For people living with Castleman disease or caring for someone who has it, the study provides hope that carefully reading the immune system’s genetic “spelling mistakes” could lead to more precise diagnoses and a broader toolkit of targeted, life‑saving treatments.
Citation: Du, Y., Xie, S., Dai, Z. et al. The clinical significance of the Mediterranean fever gene MEFV variants in Castleman disease. Commun Med 6, 121 (2026). https://doi.org/10.1038/s43856-026-01392-1
Keywords: Castleman disease, TAFRO syndrome, MEFV gene, cytokine storm, colchicine