Clear Sky Science · en
Advancing towards cancer theragnostic by probing the 225Ac decay chain with ultra-high-resolution metallic magnetic calorimeter based detectors
Sharper Eyes on Cancer-Fighting Radiation
Targeted alpha therapy is an emerging way to fight cancer by sending tiny bursts of high-energy radiation directly into tumor cells. One of the most promising radioactive materials for this approach is actinium-225, which breaks down into several “daughter” elements that can also release damaging radiation. To use this powerful tool safely and effectively, doctors must know exactly where each radioactive fragment goes in the body. This study tests a new kind of ultra-precise radiation detector to “see” almost the entire decay chain of actinium-225 in far greater detail than was previously possible.
Why Following Every Fragment Matters
Actinium-225 is attractive for cancer treatment because it emits alpha particles—heavy, energetic particles that travel only about the width of a few cells. That makes them ideal for destroying tumors while sparing most healthy tissue. But there is a catch: as actinium-225 decays, it turns into a series of new radioactive elements, such as francium-221 and bismuth-213. These daughters do not always stay attached to the original drug molecule. Once freed, they can drift to other organs, delivering unwanted radiation doses to places like the kidneys or bone marrow. At present, standard medical imaging tools can reliably detect only two of these daughter elements inside the body, leaving much of the decay chain effectively invisible. Better tracking of all the decay products would allow clinicians to calculate organ doses more accurately and fine-tune treatments for each patient.
A New Kind of Ultra-Precise Radiation Thermometer
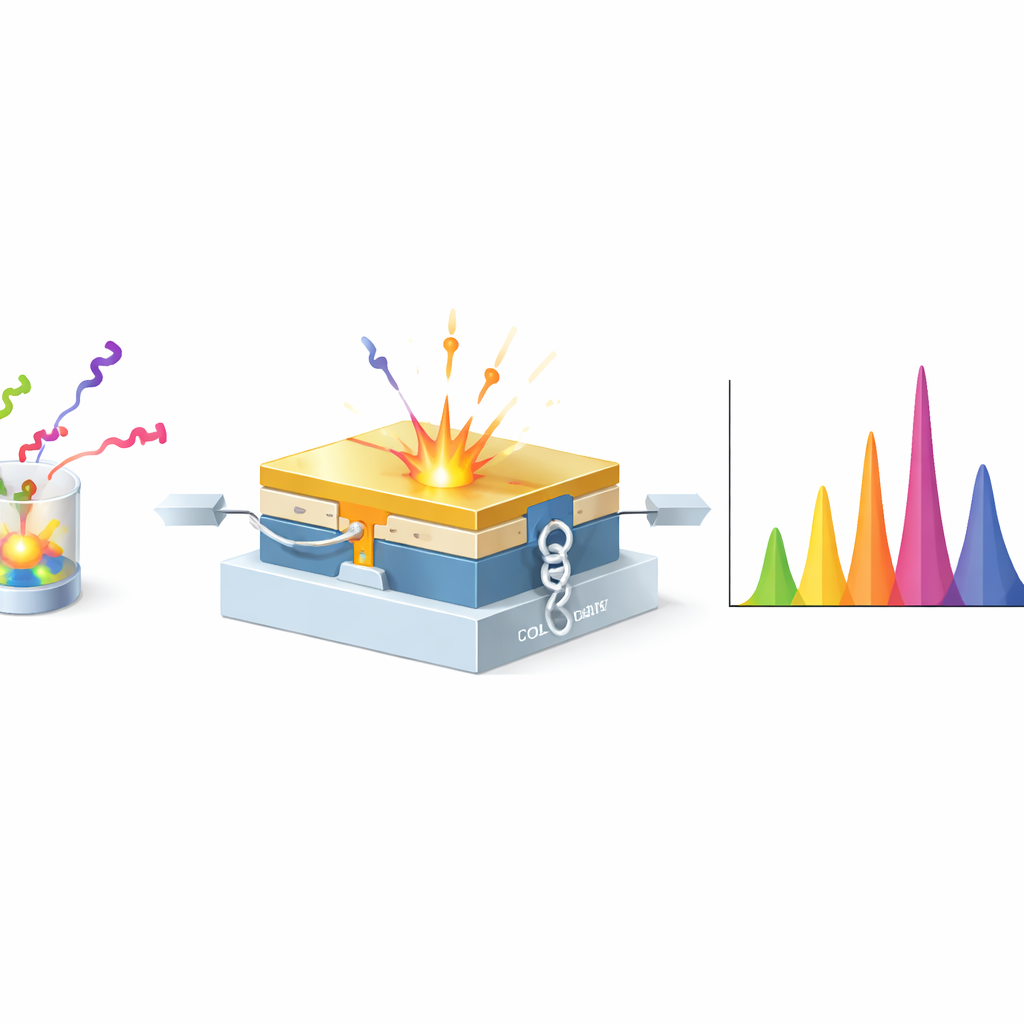
The researchers turned to a special device called a metallic magnetic calorimeter, which acts like an extremely sensitive thermometer for tiny bursts of energy. The detector is cooled to a fraction of a degree above absolute zero. When an X-ray or gamma ray from actinium-225 (or one of its daughters) hits the detector’s absorber, it warms up ever so slightly. That temperature rise changes the magnetization of a sensor, which is read out by a superconducting circuit. Because the incoming energy is converted directly into heat rather than light or electrical charge, the energy can be measured with extraordinary precision—dozens of times better than common hospital detectors—over a broad range of X-ray and gamma-ray energies.
Separating the Voices in a Crowded Signal
In the study, the team placed a sealed actinium-225 sample in front of the calorimeter and recorded its X-ray and gamma-ray spectrum for about two days, alongside calibration measurements using well-known reference sources. They then used advanced software to clean the signals, correct for slow drifts in detector behavior, and match the measured energies to theoretical values from nuclear databases. Thanks to the exceptional sharpness of the calorimeter, what used to look like single broad bumps in older detectors split into many narrow, well-separated peaks. The researchers could clearly pick out the fingerprints of actinium-225 itself, as well as multiple daughters such as francium-221, bismuth-213, thallium-209, astatine-217, polonium-213, and lead-209. Only two very short-lived steps in the decay chain remained out of reach, largely because they are present in vanishingly small amounts.
Hints of New Physics Opportunities
Beyond resolving known decay lines, the detector also picked up subtle X-ray signals that likely stem from a process called particle-induced X-ray emission. Here, the intense alpha particles from actinium-225 excite nearby atoms, causing them to emit their own characteristic X-rays. This effect has usually been studied only in lighter elements, but the calorimeter’s combination of high sensitivity and very fine energy resolution appears to extend this technique into the heavy-element region where actinium sits. That opens the door not just to better counting of nuclides, but also to new types of elemental and chemical analysis in radioactive samples that were previously too complex to study in detail.
From Lab Bench to Personalized Cancer Care
By showing that almost every step in the actinium-225 decay chain can be separated and identified with a single, ultra-precise detector, this work lays a foundation for more accurate dosimetry and quality control in targeted alpha therapy. In the near term, such detectors could help verify the purity of medical actinium samples and track tiny impurities or side products. With further development—such as thicker absorbers, more detector pixels, and integration with imaging setups—the same technology could one day be used to map where actinium-225 and its daughters actually travel in tissues or small animals, and eventually in patients. In simple terms, the study demonstrates a new way to “zoom in” on the radiation from actinium-based cancer treatments, giving clinicians the detailed information they need to better balance tumor killing power against the protection of healthy organs.
Citation: Maurer, K., Unger, D., Behe, M. et al. Advancing towards cancer theragnostic by probing the 225Ac decay chain with ultra-high-resolution metallic magnetic calorimeter based detectors. Commun Med 6, 169 (2026). https://doi.org/10.1038/s43856-026-01377-0
Keywords: targeted alpha therapy, actinium-225, nuclear medicine imaging, radiation detectors, dosimetry