Clear Sky Science · en
Plasma and pericardial fluid metabolomic signatures of patients with ischemic heart disease
Why the Heart’s Chemistry Matters
Ischemic heart disease, in which parts of the heart muscle do not receive enough blood and oxygen, is a leading cause of death worldwide. Yet standard tests often miss the earliest warning signs that heart cells are in trouble. This study asks a simple but powerful question: can we read the heart’s “chemical fingerprints” in blood and in the fluid that bathes the heart to better understand what happens during this kind of damage, and someday help doctors spot it earlier?
Looking Inside the Heart’s Surroundings
Most heart tests focus on images of the arteries or on electrical activity. Here, researchers instead examined tiny molecules that act as fuels and by‑products of energy use in the body. They studied two fluids taken from people undergoing heart surgery: ordinary blood plasma, which reflects the body’s overall state, and pericardial fluid, the liquid that directly surrounds the heart. By comparing patients with ischemic heart disease to patients with a valve problem but no blocked arteries, the team could tease apart changes specific to poor blood flow in the heart muscle.
Reading the Chemical Fingerprints
To measure many molecules at once, the scientists used a technique called magnetic resonance spectroscopy, a cousin of MRI that works on fluids in test tubes. This allowed them to build a broad “metabolic profile” for each sample. Advanced statistical methods were then applied to see whether the overall patterns in these profiles could separate ischemic from non‑ischemic patients, and to highlight which molecules contributed most to the differences. Even without telling the computer which samples came from which group, the patterns in both plasma and pericardial fluid tended to cluster into two distinct clouds, indicating that the underlying chemistry differed in a consistent way between the two sets of patients.
A Shift in the Heart’s Fuel Choices
The clearest signal came from molecules related to how the heart powers itself. Patients with ischemic heart disease had higher levels of certain “ketone bodies,” especially 3‑hydroxybutyrate and acetoacetate, in their blood. These are alternative fuels that the body typically turns to during fasting or when sugar use is limited. Their rise here suggests that the oxygen‑starved heart is switching fuel sources, leaning more on these backup energy carriers. In the pericardial fluid, 3‑hydroxybutyrate was also elevated, hinting that this shift in fuel use is not just a whole‑body response but is playing out locally around the heart itself.
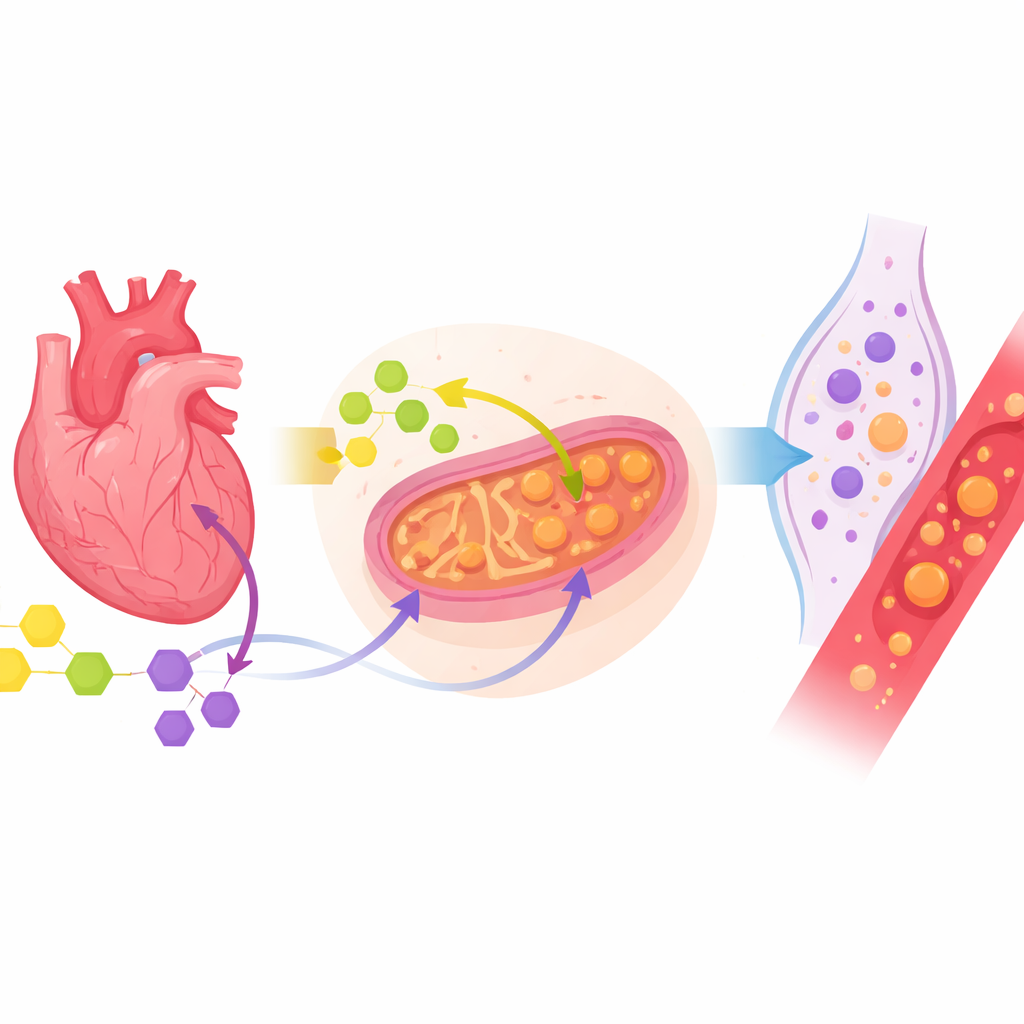
Signals of Mitochondrial Stress
Another standout change involved succinate, a molecule formed inside the cell’s powerhouses, the mitochondria. Succinate was especially increased in the pericardial fluid of ischemic patients. Earlier research has shown that when oxygen is scarce, succinate can build up in heart tissue and then spill out when blood flow returns, driving bursts of harmful reactive molecules and triggering inflammation. Finding more succinate in the fluid around the heart supports the idea that mitochondrial stress and disturbed energy flow are central features of ischemic heart disease, and that the pericardial space faithfully mirrors this hidden struggle.
Beyond Fuel: Amino Acids and Networked Pathways
The study also uncovered changes in several amino acids, the building blocks of proteins. Levels of branched‑chain amino acids, which can be broken down to feed the heart’s energy cycles, were higher in ischemic patients, while some aromatic amino acids, such as tyrosine and phenylalanine, tended to be lower. When the team mapped all altered molecules onto known metabolic pathways, they saw coordinated shifts in routes tied to fat use, ketone handling, and the central energy‑producing cycle, especially in the pericardial fluid. This pattern suggests that the chemistry around the heart is not simply a diluted copy of blood, but a focused snapshot of how the stressed heart is re‑wiring its metabolism.
What This Means for Patients
In everyday terms, this work shows that an oxygen‑starved heart changes the way it burns fuel, leaning more on emergency energy sources and accumulating telltale by‑products in both the blood and the fluid surrounding it. Molecules such as 3‑hydroxybutyrate and succinate stand out as promising markers of that shift. While the study is small and not yet ready to change clinical practice, it demonstrates that the heart’s chemical environment carries rich information about disease that today’s standard tests do not capture. In the future, refining and validating such metabolic fingerprints could help doctors detect heart damage earlier, better gauge its severity, and tailor treatments to the hidden chemistry of each patient’s heart.
Citation: De Castro, F., Coppola, C., Scoditti, E. et al. Plasma and pericardial fluid metabolomic signatures of patients with ischemic heart disease. Commun Med 6, 162 (2026). https://doi.org/10.1038/s43856-025-01353-0
Keywords: ischemic heart disease, metabolomics, pericardial fluid, ketone bodies, mitochondrial metabolism