Clear Sky Science · en
Surface-engineered dual drug-loaded tumor-targeted liposomal nanoparticles to overcome the therapeutic resistance in glioblastoma multiforme
Why this brain cancer research matters
Glioblastoma is one of the deadliest forms of brain cancer. Even with surgery, radiation, and chemotherapy, most patients live little more than a year after diagnosis. A major problem is that standard drugs often cannot reach the tumor in the brain, and even when they do, the tumor quickly adapts and becomes resistant. This study explores a new way to sneak multiple drugs across the brain’s natural defenses and concentrate them inside the tumor, with the goal of making existing treatments like radiation work much better.
A tiny delivery truck for cancer drugs
The researchers built ultra-small drug carriers called tumor-targeted liposomes. These are soft, fat-based bubbles on the scale of billionths of a meter that can hold medicines inside. The team modified the surface of these bubbles with a special peptide that recognizes and sticks to glioblastoma cells, helping the particles home in on tumors instead of healthy tissue. Each bubble was designed to carry a pair of cancer drugs at the same time: either everolimus plus vinorelbine, or rapamycin plus vinorelbine. The idea is that the two drugs attack tumor cells in complementary ways, while the protective shell helps the drugs survive in the bloodstream and slip into the brain.
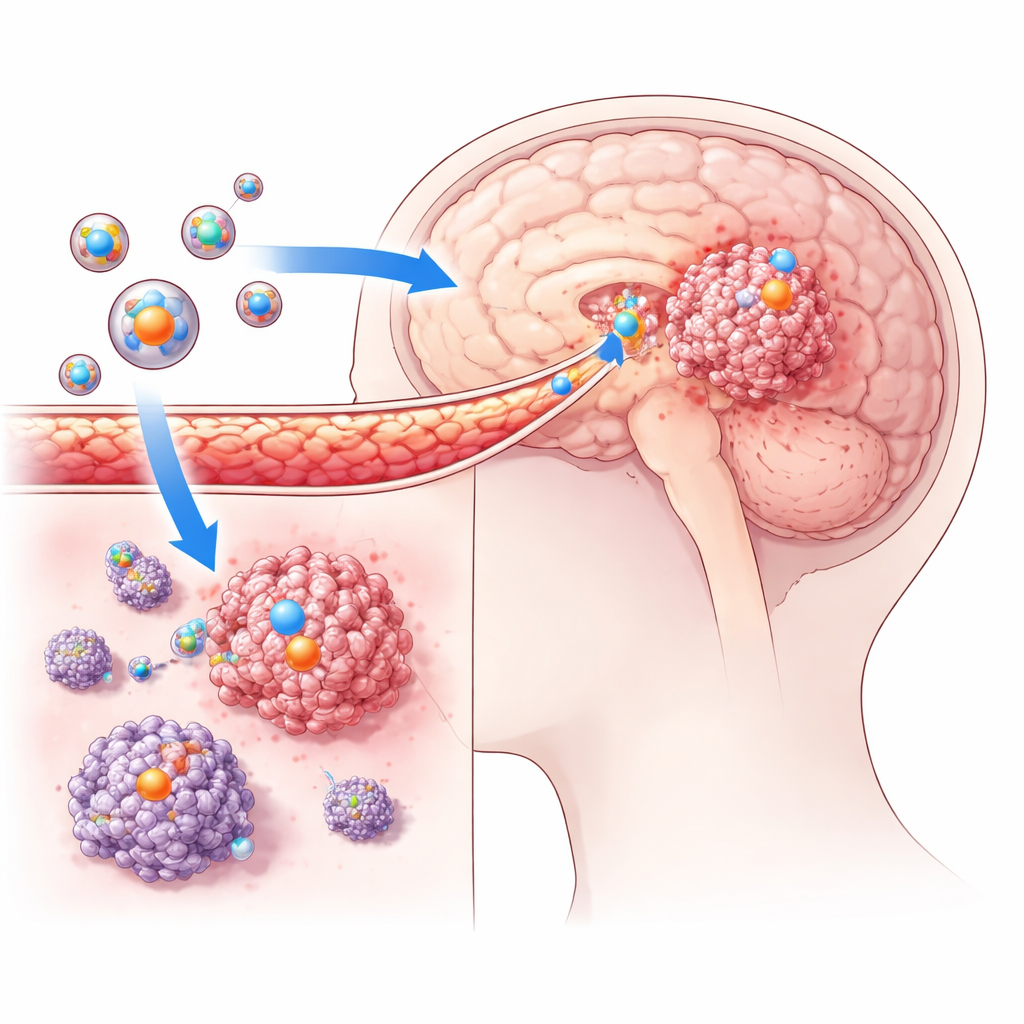
Crossing the brain’s protective wall
Using mouse models implanted with human glioblastoma cells, the team tested whether these liposomes could actually reach tumors inside the brain. They labeled the particles with fluorescent dyes and followed their journey using advanced imaging tools. Compared with non-targeted liposomes, the tumor-targeted versions showed much stronger signals inside the brain tumors and little signal in normal brain regions. This confirmed that the engineered particles were able to cross the blood–brain barrier and accumulate specifically where the cancer cells were growing. In cell culture experiments, glioblastoma cells also took up far more of the targeted liposomes than the control ones, reinforcing the idea that the surface peptide greatly improves tumor seeking and entry.
Hitting the tumor harder while sparing the rest of the body
The scientists first compared how well single-drug and dual-drug liposomes killed cancer cells in dishes. The dual-drug versions, especially the everolimus–vinorelbine combination, were more potent than either drug alone, and they worked better than the same drugs given without the liposomal carrier. When combined with radiation, the effect was even stronger: cancer cells formed far fewer colonies, moved less, and showed signs of increased damage. In mouse brain tumors, animals treated with dual-drug liposomes plus radiation had slower tumor growth and lived longer than those given radiation alone, liposomes alone, or standard chemotherapy with temozolomide. Importantly, the researchers did not see obvious harm in other organs, suggesting that focusing treatment into the tumor may reduce side effects.
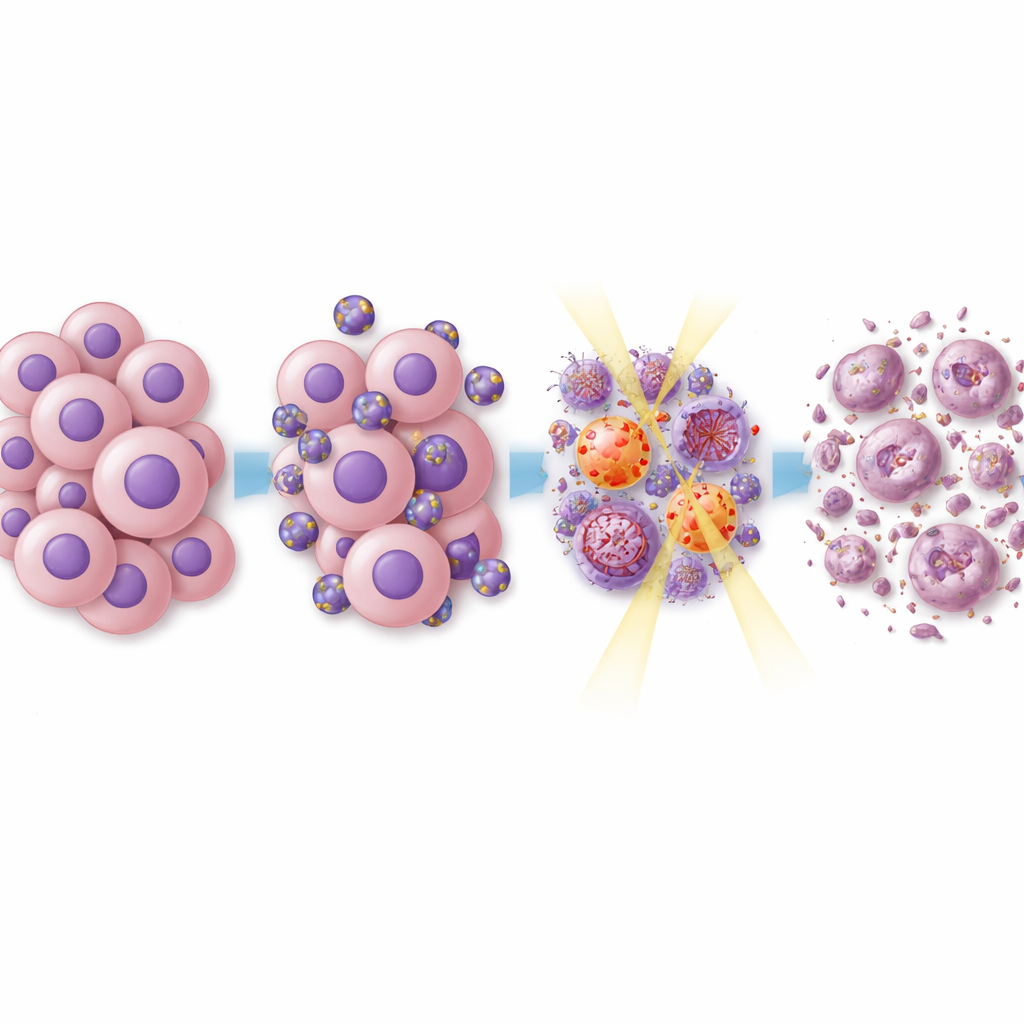
What is happening inside the cancer cells
To understand why this approach made tumors more sensitive to treatment, the team examined key signaling systems inside the cancer cells. Everolimus and rapamycin are known to block a pathway called mTOR, which helps cells grow and resist stress. The dual-drug liposomes suppressed mTOR-related signals, as well as other growth pathways that drive glioblastoma cell division and movement. When radiation was added, proteins involved in DNA damage repair were also dampened. That means the tumor cells were less able to fix the genetic injury caused by radiation, pushing them toward death instead of recovery. Detailed gene activity analyses of treated tumors showed broad changes in networks linked to cell cycle control, DNA repair, and the tumor’s interaction with the immune system, and many genes associated with treatment resistance were turned down.
What this could mean for future patients
This work shows that carefully engineered, tumor-seeking nanoparticles can ferry two cooperating drugs across the brain’s protective barrier, concentrate them inside glioblastoma, and make radiation therapy more effective. In mice, this strategy slowed tumor growth and extended survival without obvious added toxicity. While these results are still preclinical and much more testing is needed before use in people, the study points to a practical way to combine targeted drug delivery with existing treatments to outsmart a highly resistant cancer. If similar benefits are seen in humans, such dual drug-loaded liposomes might one day offer glioblastoma patients longer and better-quality lives.
Citation: Angom, R.S., Rachamala, H.K., Nakka, N.M.R. et al. Surface-engineered dual drug-loaded tumor-targeted liposomal nanoparticles to overcome the therapeutic resistance in glioblastoma multiforme. Commun Med 6, 152 (2026). https://doi.org/10.1038/s43856-025-01279-7
Keywords: glioblastoma, nanoparticles, liposomes, brain cancer treatment, radiation sensitization