Clear Sky Science · en
Subtyping Alzheimer’s disease and Parkinson’s disease using longitudinal electronic health records
Why this matters for families and patients
Alzheimer’s and Parkinson’s disease are often spoken of as if they were single, uniform illnesses. Yet anyone who has watched a loved one decline knows that no two journeys look the same. This study asks a simple but powerful question: can the medical records we already collect over many years reveal hidden “flavors” of these brain diseases, in a way that might eventually guide more tailored care? Using modern pattern-finding tools on the health histories of over 100,000 people in the United Kingdom, the authors show that Alzheimer’s and Parkinson’s can each be broken down into five distinct subtypes, with different risks, symptoms, and likely futures.
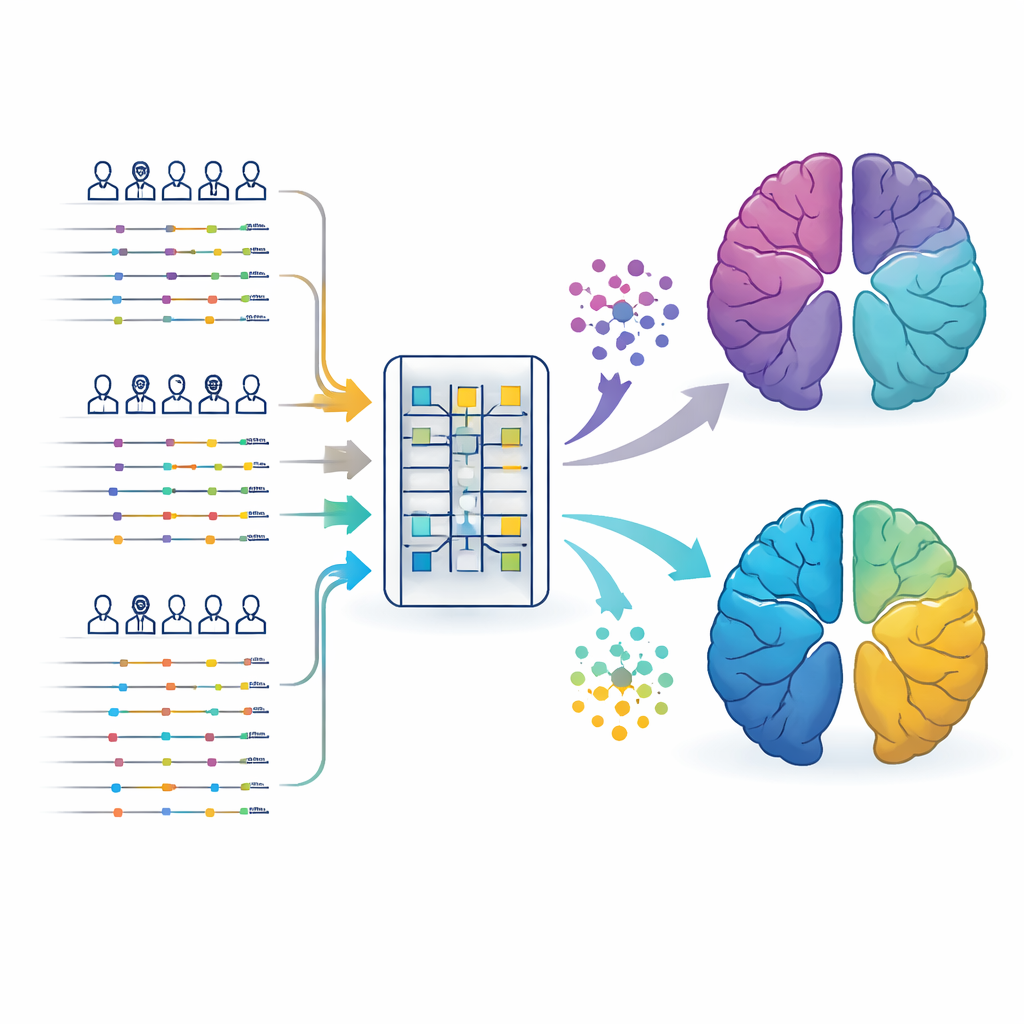
Following people’s health stories over decades
The researchers drew on anonymized electronic health records from two large UK resources: the Clinical Practice Research Datalink, which holds information from general practitioners, and the UK Biobank, a long-running research project that links clinic visits, hospital stays, prescriptions, and genetic data. They focused on more than 113,000 people with Alzheimer’s disease and over 45,000 with Parkinson’s disease, each with around two to three decades of medical history before their diagnosis. Instead of looking only at test results taken after the brain disease was recognized, the team fed these long, time-stamped health “stories” into a transformer, a type of artificial intelligence that is good at understanding sequences such as language or, in this case, a timeline of doctor visits.
Letting the data sort patients into hidden groups
From every person’s record, the model created a compact numerical “fingerprint” that captured patterns of illnesses, prescriptions, and timing. A clustering method then sorted these fingerprints into groups without being told in advance what to look for. For both Alzheimer’s and Parkinson’s, the most stable and reproducible solution was five subtypes each. One group in each disease looked like a classic, mostly “pure” brain disorder: people carried stronger genetic risk scores but had relatively few other medical problems and fared better after diagnosis. Other groups were shaped by patterns that appeared years earlier, such as heavy vascular disease, metabolic problems like diabetes and obesity, or a long history of anxiety and depression.
Different paths, different outcomes
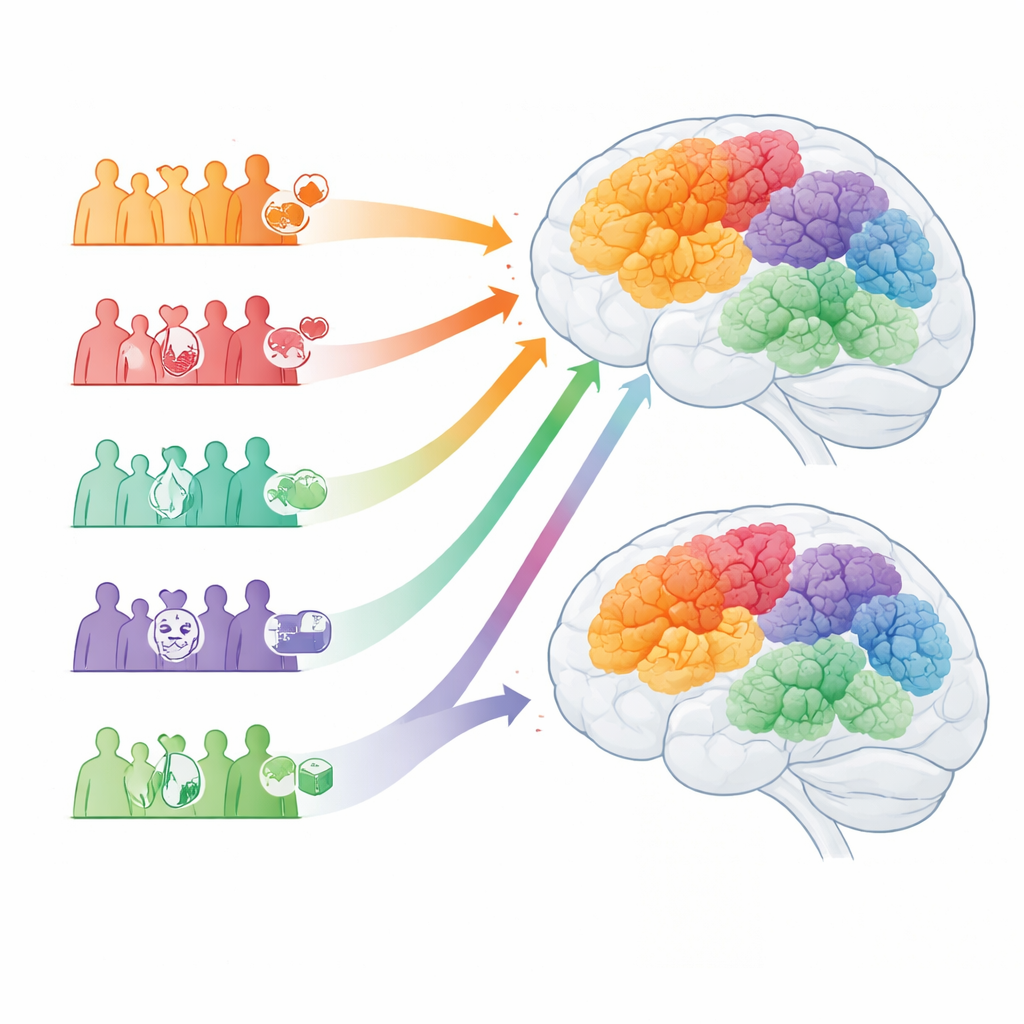
The five Alzheimer’s subtypes ranged from a common late-onset form with fewer other illnesses, to a vascular-dominated pattern with widespread high blood pressure, to a “mental health” form marked by prominent depression, anxiety, and faster loss of thinking skills. Another subtype was strongly tied to diabetes, kidney disease, and signs of chronic inflammation, despite having lower inherited risk; these patients tended to become ill earlier and die sooner. A final Alzheimer’s group showed more heart disease and movement problems and had the highest rates of hospital admission and death. The Parkinson’s subtypes echoed these themes: one group with relatively “clean” health histories and higher genetic risk, one dominated by high blood pressure and blood vessel disease, one with severe mood and movement difficulties, one with strong metabolic and inflammatory features, and one combining serious heart and movement problems with particularly poor survival.
Genes and shared risk factors across brain diseases
Because the UK Biobank includes DNA data, the authors could ask whether these clinically defined subtypes also differ biologically. In both conditions, all patient groups had higher disease-related genetic risk scores than people without the diagnosis, as expected. Yet some clusters carried especially strong or weak genetic signals. For instance, a “metabolic–inflammatory” Alzheimer’s subtype had lower scores for classic Alzheimer’s risk genes and was relatively enriched for a protective form of the APOE gene, suggesting that lifelong conditions such as diabetes and obesity can drive a dementia-like picture even when inherited risk is modest. Across both Alzheimer’s and Parkinson’s, subtypes shaped by high blood pressure, stroke risk, or mood disorders recurred in similar ways, hinting that blood vessel health, metabolism, and mental health may nudge the brain toward different patterns of degeneration rather than being mere side effects of aging.
What this could mean for future care
This work does not yet change how doctors diagnose patients at the bedside, and the authors stress that medical records cannot, on their own, prove the exact biological causes behind each subtype. Still, by showing that long-term routine data naturally fall into consistent, meaningful groups that match differences in symptoms, survival, and genetics, the study offers a blueprint for more precise, early risk profiling. In the future, people with certain mixes of blood vessel disease, diabetes, or mood problems might be monitored more closely for specific patterns of Alzheimer’s or Parkinson’s, and drug trials could target subtypes most likely to benefit. In short, the findings support a shift from treating these conditions as single monolithic diseases toward viewing them as families of related but distinct paths—opening the door to more personalized prevention and therapy.
Citation: Lian, J., Fan, Z., Petrazzini, B.O. et al. Subtyping Alzheimer’s disease and Parkinson’s disease using longitudinal electronic health records. Nat Aging 6, 612–625 (2026). https://doi.org/10.1038/s43587-026-01085-3
Keywords: Alzheimer’s disease subtypes, Parkinson’s disease subtypes, electronic health records, machine learning in neurology, neurodegeneration risk factors