Clear Sky Science · en
Structural signature of plasma proteins classifies the status of Alzheimer’s disease
Why a Blood Test for Memory Loss Matters
Alzheimer’s disease is often well underway long before memory problems become obvious, but today’s tests to spot it early can be invasive, expensive or hard to access. This study explores a different idea: whether tiny changes in the three-dimensional shapes of proteins circulating in our blood can reveal where someone sits on the path from healthy aging to mild forgetfulness to full Alzheimer’s disease. If successful, such a blood test could make screening simpler, support earlier treatment and help researchers track who benefits from new therapies.
Looking at Protein Shape, Not Just Amount
Most blood tests measure how much of a given molecule is present. Here, scientists instead focused on protein shape. Inside our cells, a quality-control system keeps proteins properly folded; when this system falters with age, misfolded proteins can pile up and damage brain cells. The team wondered if this breakdown in “protein housekeeping” leaves a structural fingerprint in blood proteins. From 520 volunteers who were either cognitively healthy, had mild cognitive impairment (MCI) or had Alzheimer’s disease, they drew blood and used a chemistry technique called covalent protein profiling to tag exposed parts of proteins. The more exposed a site, the more easily it is tagged, giving a numerical readout of protein shape that is largely independent of how much protein is present.
Finding Structural Fingerprints in the Blood
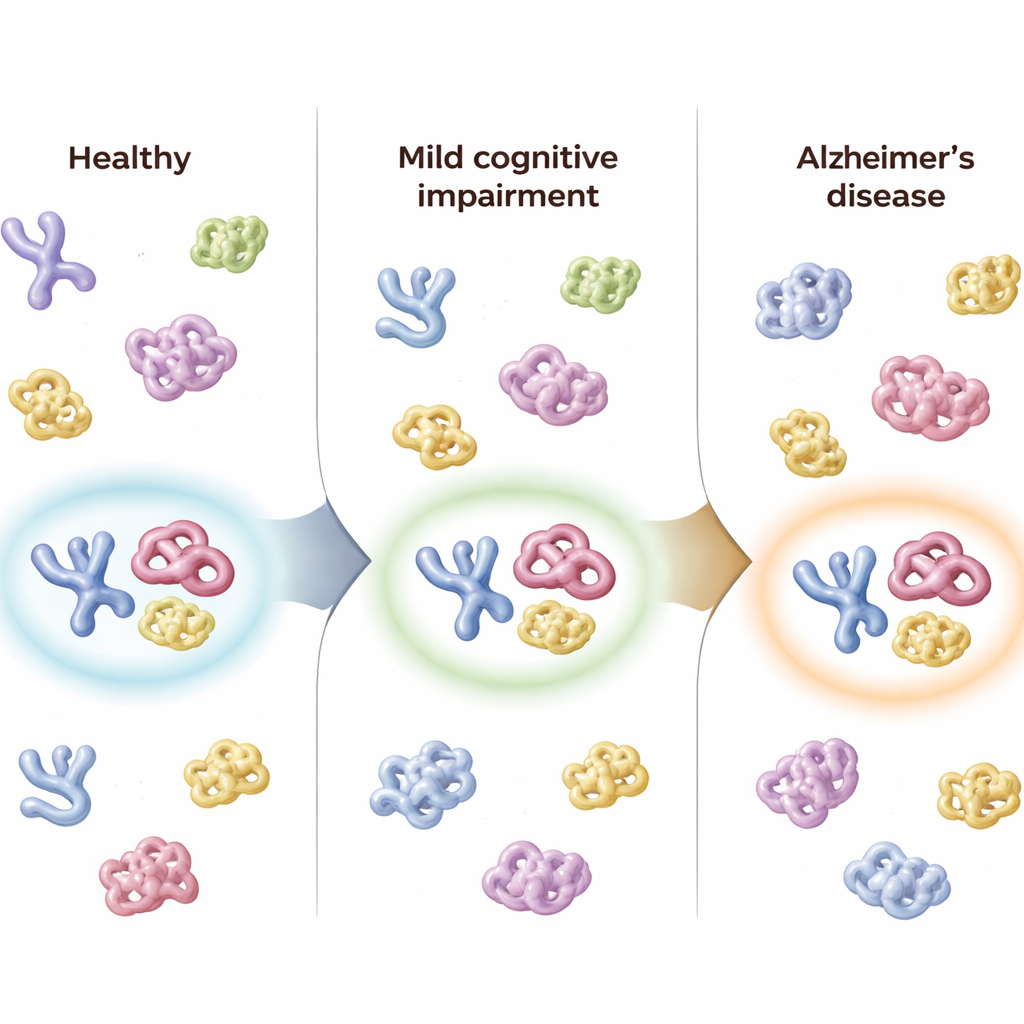
Across nearly 900 tagged protein fragments, the researchers saw a subtle but consistent trend: as people moved from healthy to MCI to Alzheimer’s, certain protein surfaces became less exposed and more variable from person to person. This pattern fits with the idea that the body’s protein-folding control worsens as the disease advances. The team also examined how a major genetic risk factor for Alzheimer’s, the APOE ε4 variant, influenced protein shape. They found that people carrying two copies of ε4 showed distinctive structural shifts in several proteins that interact with the APOE protein, suggesting that this risky gene alters not just which proteins are present, but how they are folded and arranged.
Linking Blood Proteins to Mood and Behavior
Alzheimer’s affects far more than memory; symptoms such as agitation, depression and hallucinations are common and often differ between men and women. The researchers combined clinical ratings of 12 types of neuropsychiatric symptoms with their protein-shape measurements. In both sexes, worse symptoms tended to go hand in hand with proteins becoming more structurally “closed off.” Some proteins, including clusterin and several others linked to amyloid buildup, tracked symptom severity similarly in men and women, while others showed sex-specific patterns. These findings hint that blood protein structure may reflect not only the presence of disease, but also how it manifests in behavior and mood.
A Three-Protein Panel to Classify Disease Stage
To turn these complex measurements into something clinically useful, the team fed the structural data into 18 different machine-learning approaches. The best-performing model, based on deep learning, relied on just three protein fragments drawn from C1QA, clusterin (also called CLUS) and apolipoprotein B (ApoB). Using only the structural readouts from these three fragments, the model correctly sorted people into healthy, MCI or Alzheimer’s groups about 83% of the time in an independent test set. When asked easier two-way questions—such as distinguishing healthy from MCI, or MCI from Alzheimer’s—the panel’s performance was even stronger, with measures of accuracy comparable to or better than many current blood-based approaches that focus on protein amounts rather than structure.
Following People Over Time
The researchers also had follow-up samples from 50 participants taken up to about eight months later. In people whose diagnosis worsened—for example, from healthy to MCI, or from MCI to Alzheimer’s—the combined score from the three-protein panel shifted in the same direction, reflecting disease progression. In contrast, individuals whose clinical status stayed the same showed little change in their panel scores. The panel’s overall “Alzheimer’s confidence” score tracked closely with standard cognitive tests, with measures of daily functioning, with brain shrinkage seen on MRI scans and with established spinal fluid markers of amyloid and tau, suggesting that the blood-based structural signal mirrors underlying brain changes.
What This Could Mean for Patients
Taken together, the work shows that small, coordinated changes in the shapes of just a few abundant blood proteins can reliably signal whether someone is cognitively healthy, mildly impaired or has Alzheimer’s disease. Because the test uses blood rather than spinal fluid or brain imaging, it could, in principle, be scaled for routine screening or to select and monitor participants in clinical trials. The authors caution that larger and longer-term studies are needed, and the chemistry involved is still specialized. Nonetheless, their results point to protein structure—not only protein levels—as a promising new source of information for detecting and tracking Alzheimer’s disease earlier and more precisely.
Citation: Son, A., Kim, H., Diedrich, J.K. et al. Structural signature of plasma proteins classifies the status of Alzheimer’s disease. Nat Aging 6, 597–611 (2026). https://doi.org/10.1038/s43587-026-01078-2
Keywords: Alzheimer’s disease biomarkers, blood test for dementia, protein folding changes, machine learning diagnosis, clusterin and C1QA