Clear Sky Science · en
Induction of senescence during postpartum mammary gland involution supports tissue remodeling and promotes postpartum tumorigenesis
Why this matters for new mothers
After childbirth, the breast must quickly shift from producing milk to returning to a resting state. This dramatic makeover, called involution, has long been linked to a temporary spike in breast cancer risk in the years after pregnancy, but the biological reasons were unclear. This study in mice reveals that a special "retirement" state of cells, known as senescence, helps the breast heal after weaning—yet the same program can be co‑opted by budding tumors to grow and spread. Understanding this double‑edged process could open doors to new ways of protecting women from postpartum breast cancer.
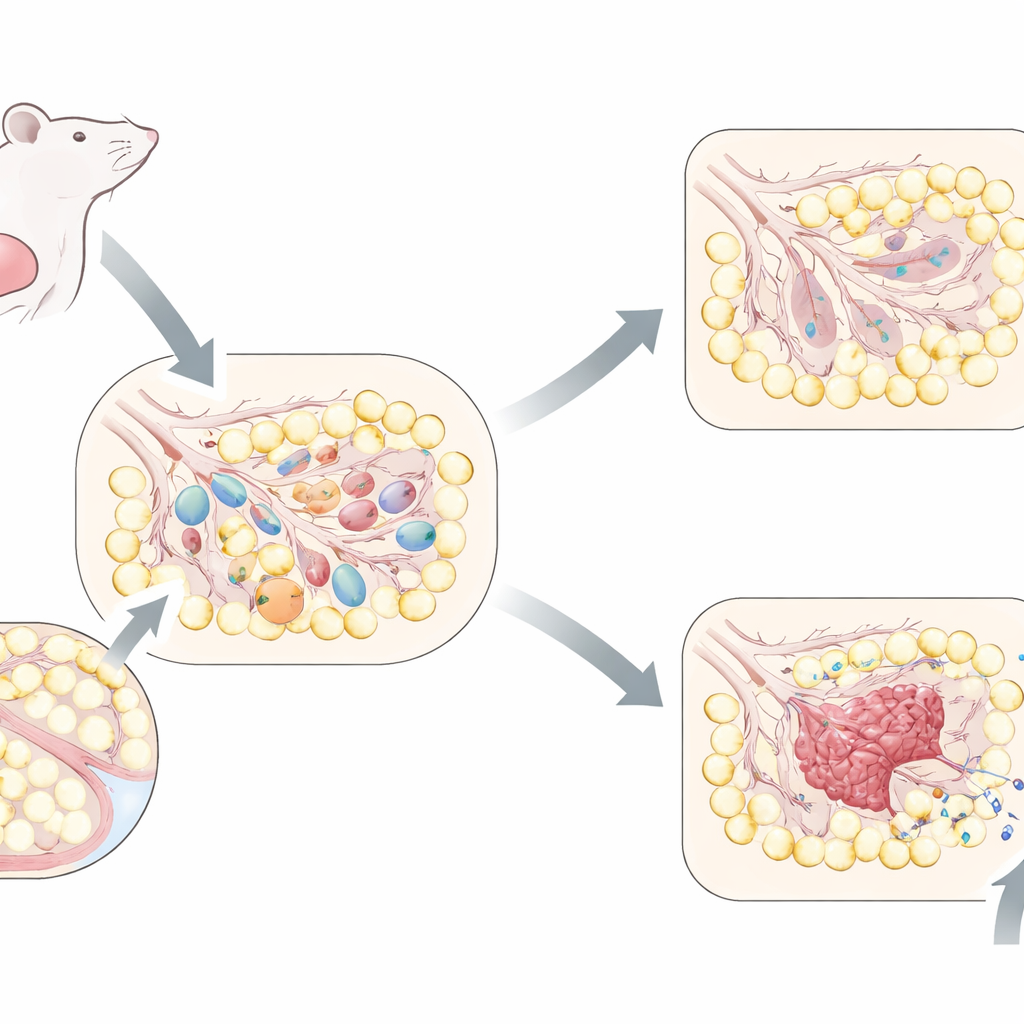
A dramatic remodel after breastfeeding ends
During pregnancy and breastfeeding, the mammary gland expands into a dense network of tiny sacs that produce and store milk. Once nursing stops, most of these milk‑making structures are no longer needed. In mice, the tissue undergoes a tightly timed sequence: first, many milk‑producing cells die, then the gland is rebuilt as fat cells refill the space and supporting structures are reshaped. The authors tracked this process at many time points and found that, a few days after weaning, a wave of cells with hallmarks of senescence appears specifically in the milk‑producing units. These cells gradually vanish as the gland finishes returning to its pre‑pregnancy form, indicating that senescence is a short‑lived but programmed part of normal remodeling rather than an age‑related buildup.
Retired cells that help reshape the breast
Senescent cells are often viewed as culprits of aging because they stop dividing yet release many signaling molecules that can inflame or damage nearby tissues. Here, however, senescent cells appear to serve a constructive purpose. Using genetic tools, the researchers disabled a key senescence switch, the gene p16, only in the milk‑producing cells. They also used a drug that selectively kills senescent cells. In both cases, when senescence was reduced, the early stages of involution slowed down: milk‑producing structures lingered longer than normal, fat cells refilled less efficiently, and the overall architecture of the gland took more time to reset. The team also showed that these senescent cells release signals that attract immune cells called macrophages, which are known to help clear dying cells and guide tissue repair. In this context, senescence behaves like a temporary "foreman" that orchestrates cleanup and rebuilding after lactation.
How helpful repair becomes a cancer ally
The same features that make senescent cells powerful coordinators of repair also make them risky when cancerous cells are present. Postpartum breast cancers are more likely to spread and to be deadly than those diagnosed in women who have never given birth. In mouse models prone to developing mammary tumors, a single pregnancy followed by involution caused tumors to appear sooner. When the researchers either dampened senescence genetically or cleared senescent cells with the senolytic drug ABT‑263 during the involution period, tumors appeared later and were less frequent. In laboratory-grown tumor organoids, fluids collected from involuting glands rich in senescent cells made cancer cells more invasive, while fluids from glands with reduced senescence had a weaker effect. These experiments suggest that senescent cells, through their cocktail of secreted factors, increase the plasticity and aggressiveness of nearby tumor‑initiating cells.
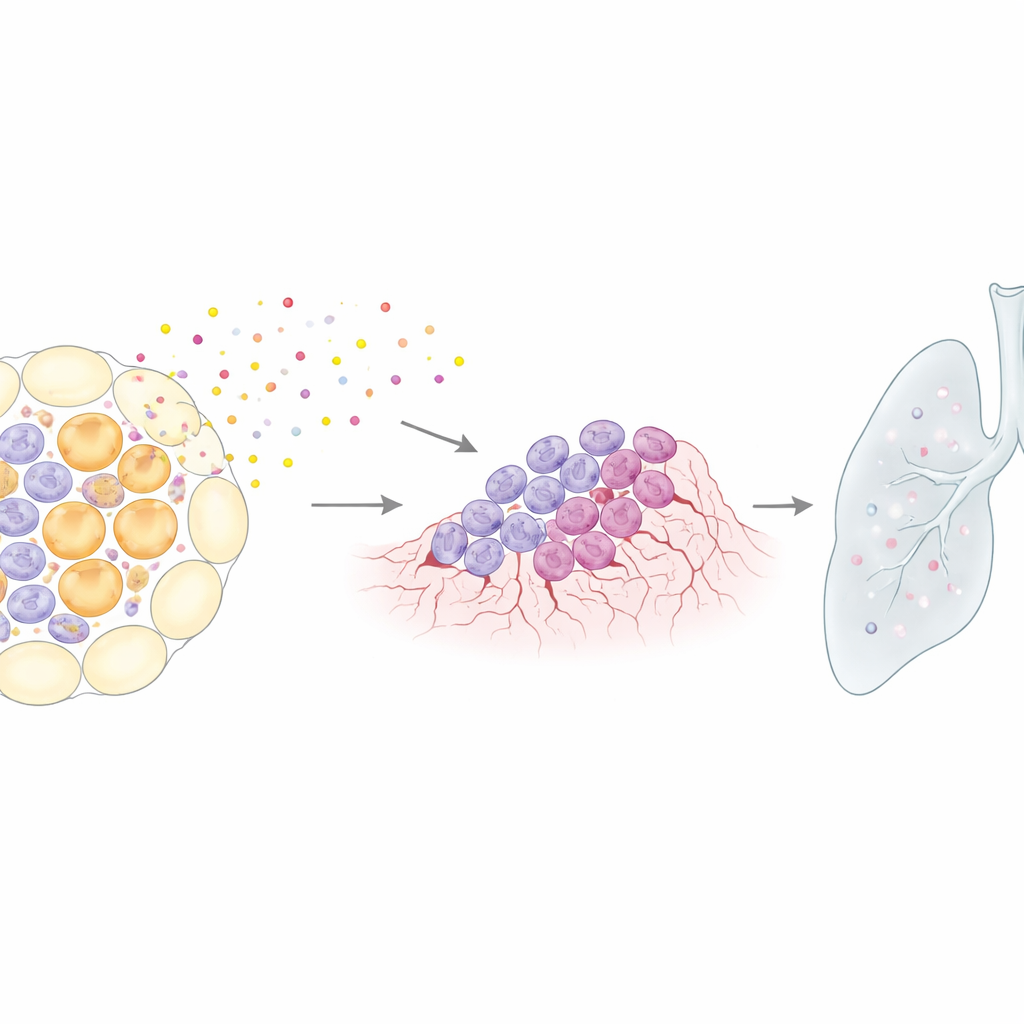
From local changes to distant spread
The team then examined whether this senescence‑driven environment could encourage cancer cells to travel beyond the breast. They implanted aggressive breast tumor cells into either resting or involuting mammary glands. In involuting glands, lung metastases were common, but when senescent cells were removed with ABT‑263 during involution, lung and other organ metastases were markedly reduced, even though the drug did not directly harm tumor cells in non‑involuting animals. This indicates that the postpartum remodeling period creates a transient window in which senescent cells help tumors seed and colonize distant sites. Intriguingly, when females became pregnant again during nursing—thereby delaying full involution and its senescence wave—the usual acceleration of tumor formation after pregnancy was blunted, consistent with the idea that the senescent remodeling phase is the risky interval.
What this means for postpartum breast cancer
Altogether, the study reveals that senescence during postpartum breast involution is a programmed, hormone‑dependent step that helps the gland safely transition from milk factory back to resting organ. Yet if cancer‑prone or already abnormal cells are present, the same senescent cells can fuel tumor growth, invasion and spread by showering the tissue with potent signaling molecules. For a lay reader, the takeaway is that the body’s own wound‑healing tools can be hijacked by cancer at vulnerable moments such as after childbirth. In the future, carefully timed treatments that target senescent cells—or the signals they release—might preserve the benefits of normal tissue repair while lowering the heightened risk of postpartum breast cancer.
Citation: Chiche, A., Djoual, L., Charifou, E. et al. Induction of senescence during postpartum mammary gland involution supports tissue remodeling and promotes postpartum tumorigenesis. Nat Aging 6, 541–559 (2026). https://doi.org/10.1038/s43587-025-01058-y
Keywords: postpartum breast cancer, cellular senescence, mammary gland involution, tumor metastasis, senolytic therapy