Clear Sky Science · en
Treatment resistance to platinum-based chemotherapy in lung and ovarian cancer is driven by a targetable TGFβ senescent secretome
When Cancer Treatment Backfires
Platinum-based chemotherapy is one of the workhorses of modern cancer treatment, especially for lung and ovarian cancers. It is designed to damage tumor DNA so badly that cancer cells can no longer divide. Yet many patients see their tumors shrink at first, only to have the disease return, tougher than before. This study asks a simple but unsettling question: can the very drugs that save lives also help surviving cancer cells bounce back? The answer, the authors find, lies in a special group of damaged, aging-like cells and the powerful chemical messages they send.
The Double Life of Aging Cancer Cells
Chemotherapy does not treat all tumor cells the same way. Some are killed outright, but others enter a state called cellular senescence—permanently stopped from dividing, yet very much alive. These senescent cells swell in size and begin secreting a cocktail of proteins and signals known as the senescence-associated secretory phenotype, or SASP. Using human lung and ovarian cancer cells in the lab, as well as mouse models, the researchers showed that platinum drugs such as cisplatin and carboplatin create especially potent senescent cells. When fresh cancer cells were bathed in fluid taken from these senescent cultures, they grew faster, migrated more readily and formed larger, more aggressive clusters and spheres than control cells.
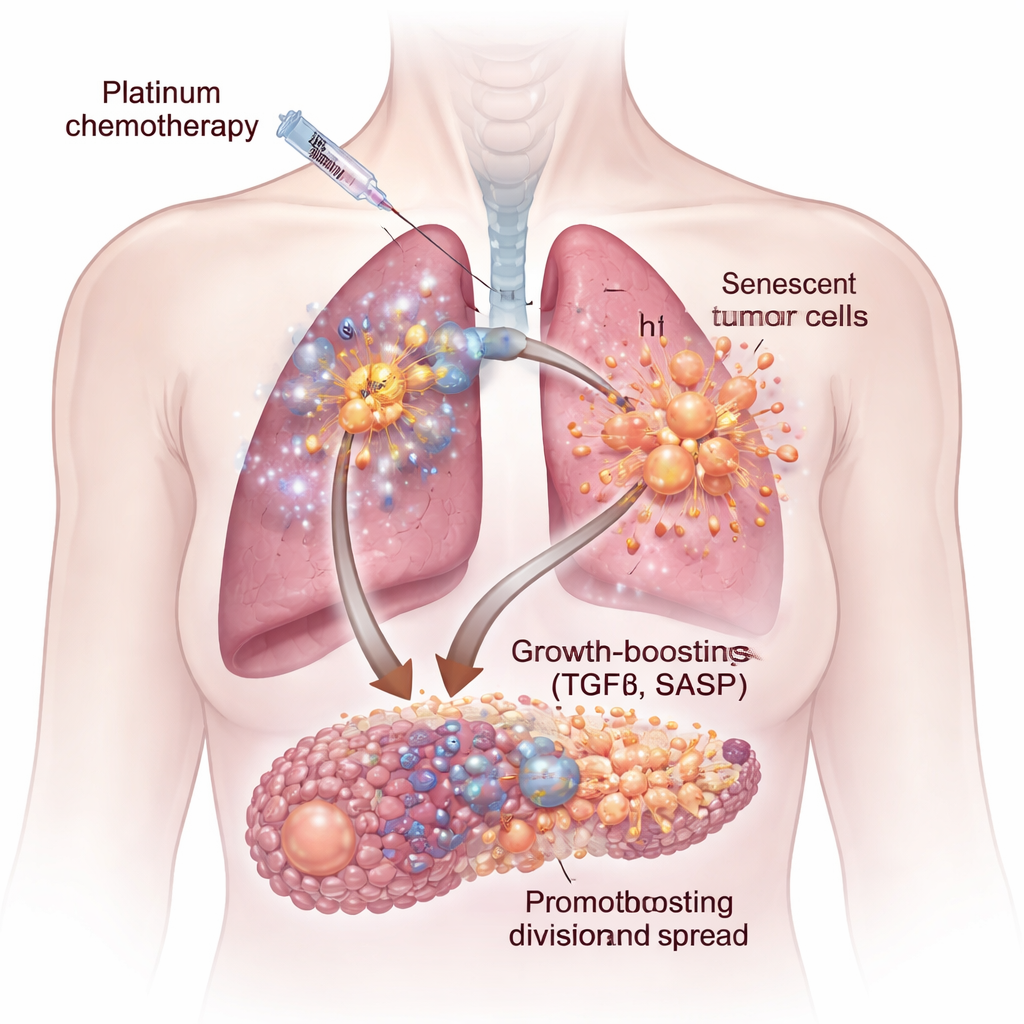
A Hidden Growth Signal: TGFβ
Not all chemotherapies triggered the same harmful secretions. When the team compared platinum drugs to two other common agents, docetaxel and palbociclib, only the platinum-treated cells produced a SASP that strongly boosted tumor growth. By combining gene expression studies, protein measurements and a high-throughput microenvironment microarray, they identified a key culprit: the signaling molecule transforming growth factor-beta, or TGFβ. Senescent cells induced by cisplatin were enriched for multiple TGFβ forms and related pathways. Adding purified TGFβ to cancer cells mimicked the growth boost seen with senescent-cell fluid, highlighting this cytokine family as a central driver of the pro-tumor secretome.
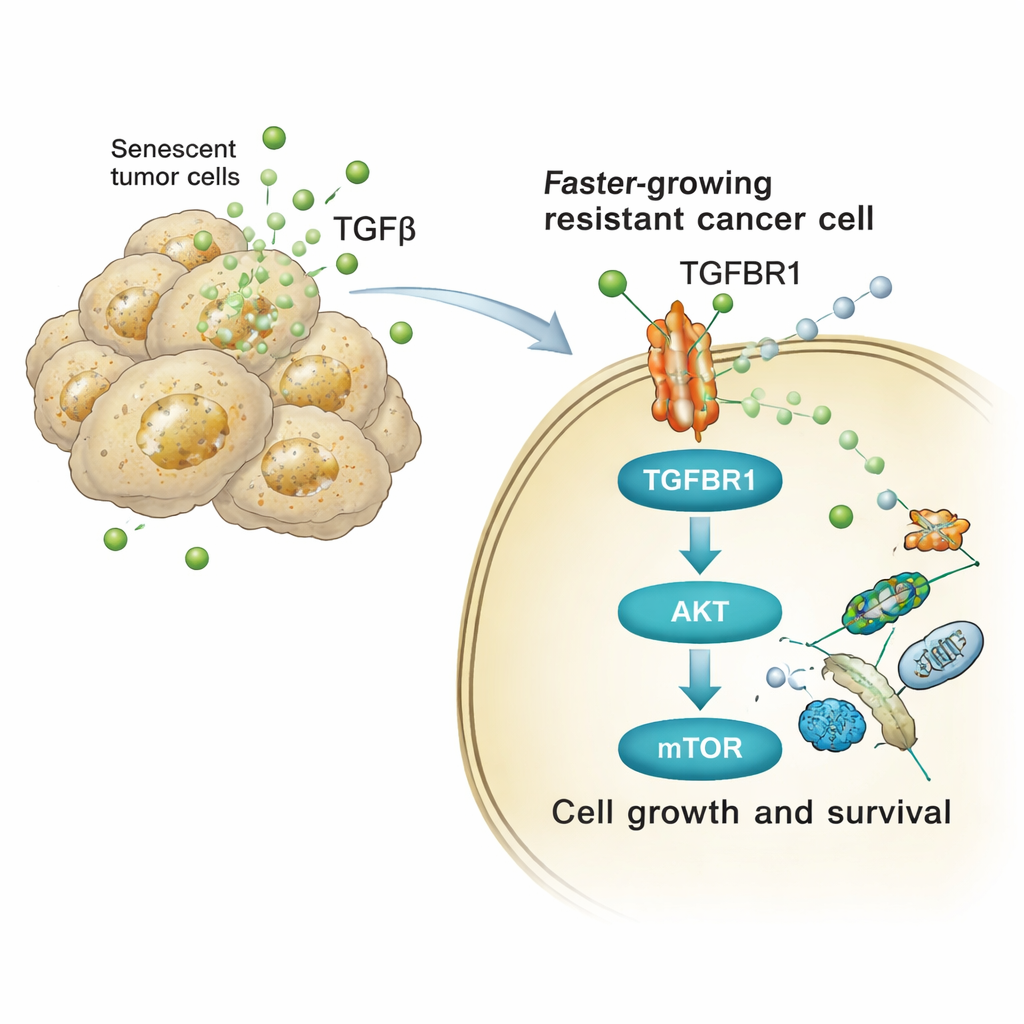
How the Signal Rewires Cancer Cells
The study then traced how these TGFβ-rich signals change the behavior of neighboring tumor cells. On the surface of recipient cells, TGFβ binds to a receptor called TGFBR1. This, in turn, switches on an internal chain of proteins—particularly the AKT and mTOR pathways—that are known to control cell growth, metabolism and survival. In both human and mouse lung cancer cells, exposure to cisplatin-derived secretions rapidly increased the activated (phosphorylated) forms of AKT and its downstream target p70S6K, and boosted the expression of cell-cycle genes. Blocking TGFBR1 with a drug called galunisertib, or directly inhibiting mTOR, largely shut down this signaling surge and reduced the extra proliferation, colony formation and invasive sphere growth triggered by the senescent secretome.
From Mice to Patients: A Shared Weak Spot
These mechanisms were not confined to petri dishes. In mice bearing lung tumors, mixing senescent and non-senescent cancer cells led to faster-growing tumors and shorter survival, while eliminating senescent cells or blocking TGFBR1 curtailed this effect. When standard cisplatin treatment was given to mice with genetically engineered lung cancers, tumors accumulated senescent markers alongside high AKT/mTOR activity in nearby, still-dividing cells. Combining cisplatin with TGFBR1 inhibition or senolytic drugs (which selectively kill senescent cells) reduced tumor burden and extended survival compared to chemotherapy alone. Crucially, analyses of human lung and high-grade serous ovarian cancer samples taken after platinum-based therapy revealed similar patterns: increased markers of senescence and heightened AKT/mTOR signaling in the tumor microenvironment, especially in regions rich in senescent cells.
Turning a Liability into an Advantage
For a layperson, the take-home message is that platinum chemotherapy can leave behind a population of damaged but dangerous “zombie” cells. These senescent tumor cells no longer divide themselves, but they pump out TGFβ-rich signals that awaken nearby cancer cells, helping tumors regrow and resist further treatment. The encouraging news is that this weakness can be targeted. In preclinical lung and ovarian cancer models, adding drugs that block the TGFβ receptor TGFBR1 or that selectively clear senescent cells made platinum chemotherapy more effective and improved survival without obvious extra toxicity. The work points toward future clinical trials that combine standard platinum treatment with senolytic or anti-TGFβ strategies, with the goal of preserving the benefits of chemotherapy while disarming its hidden, senescence-driven side effects.
Citation: González-Gualda, E., Reinius, M.A.V., Macias, D. et al. Treatment resistance to platinum-based chemotherapy in lung and ovarian cancer is driven by a targetable TGFβ senescent secretome. Nat Aging 6, 368–392 (2026). https://doi.org/10.1038/s43587-025-01054-2
Keywords: chemotherapy resistance, cellular senescence, TGFβ signaling, lung cancer, ovarian cancer