Clear Sky Science · en
DNA damage in macrophages drives immune autoreactivity via nuclear antigen presentation
When Cellular Wear and Tear Turns the Immune System Against Us
Aging brings a higher risk of autoimmune diseases, in which the immune system begins to attack the body it is meant to protect. This study explores a surprising culprit: everyday DNA damage inside immune cells called macrophages. By tracing how this damage changes what macrophages show to other immune cells, the researchers uncover a possible missing link between getting older and developing illnesses like lupus.
Guardians of the Body Gone Off Script
Macrophages are frontline defenders that swallow microbes and debris and then display protein fragments to T cells, helping the immune system decide what to attack. The team engineered mice whose macrophages were defective in a key DNA repair protein, ERCC1-XPF, so that these cells carried persistent DNA damage. As these mice aged, they developed hallmarks of autoimmunity: inflammatory foci in the kidneys, deposits of immune complexes and complement proteins, enlarged spleens, and high levels of antinuclear antibodies similar to those seen in naturally old animals. Detailed immune profiling showed an expansion of plasma cells, activated T cells, and natural killer cells, all signs of a chronically stimulated immune system.
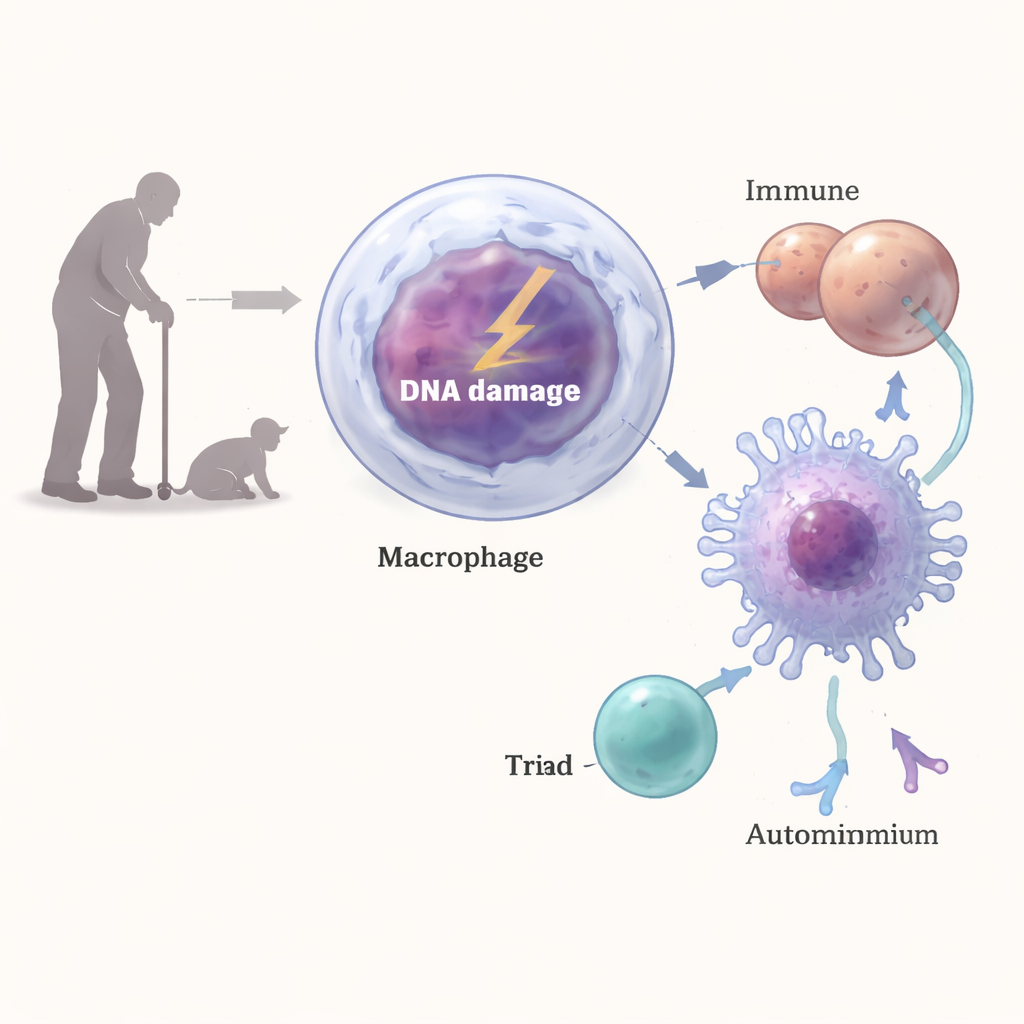
From Broken DNA to Self-Targeting T Cells
How could damaged DNA in macrophages trigger such a strong autoimmune-like response? The researchers found that DNA breaks activated a classic damage-response pathway in these cells, involving enzymes such as ATM, ATR and DNA-PK. This signaling boosted the surface levels of MHC class II, the molecular display cases used to present protein fragments to CD4 T cells. When macrophages with DNA damage were mixed with T cells, they drove stronger T cell activation and interferon-gamma production than normal macrophages. Blocking MHC-II or interfering with the DNA damage response reversed much of this effect, and in live animals, antibodies that blocked MHC-II or depleted CD4 T cells reduced kidney inflammation and autoantibody levels.
Self-Proteins from the Nucleus Put on Display
To see exactly what damaged macrophages were showing to T cells, the team isolated the peptides bound to MHC-II and analyzed them by mass spectrometry. Compared with normal or lipopolysaccharide-stimulated cells, DNA-damaged macrophages presented a strikingly different menu of peptides. Instead of mostly membrane and extracellular proteins, their MHC-II molecules were enriched for fragments derived from nuclear and ribosomal proteins, including histones and other components closely associated with DNA. When some of these nuclear peptides were synthesized and used to restimulate immune cells from the engineered mice, they triggered strong T cell responses, suggesting that these self-derived fragments were indeed immunogenic.
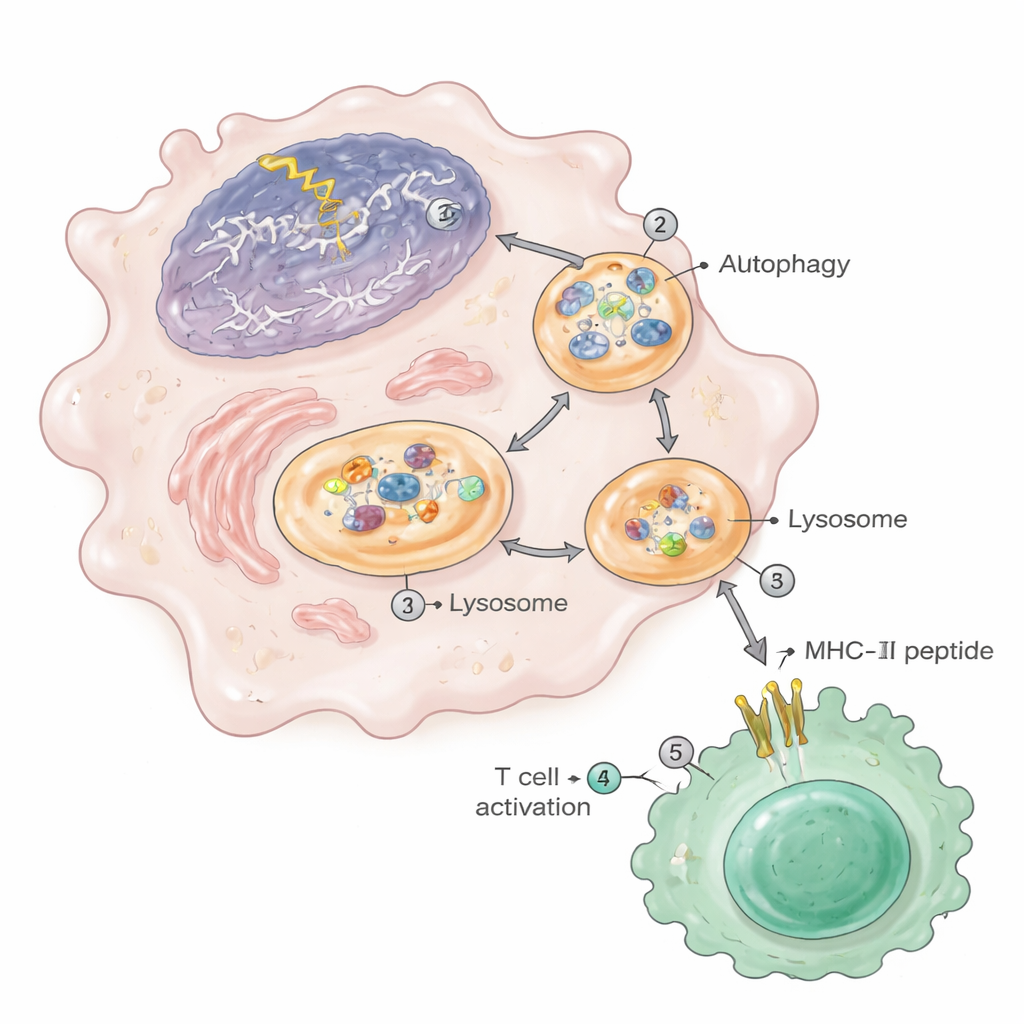
Autophagy: The Cleanup Pathway That Fuels Autoimmunity
The journey of nuclear material to the MHC-II display case depended heavily on autophagy, the cell’s internal recycling system. Under DNA damage, autophagy ramped up and shuttled chromatin fragments—from the cell’s own nucleus—into autophagosomes that then fused with lysosomes, where proteins are broken into peptides. These lysosomes in damaged macrophages contained elevated levels of nuclear and ribosomal proteins. When the researchers blocked autophagy with drugs or genetically removed an essential autophagy gene (Atg5) in the same myeloid cells, the animals showed fewer kidney lesions, fewer activated T cells and plasma cells, and a dramatic reduction in nuclear peptides appearing on MHC-II. Similar chromatin fragments and increased antigen presentation were seen in macrophages from lupus-prone mice, human monocyte cell lines exposed to DNA-damaging drugs, and in macrophages from very old mice.
Why This Matters for Aging and Autoimmune Disease
Taken together, the findings suggest a simple but powerful idea: as we age, DNA damage accumulates in macrophages, which in turn use autophagy to clear nuclear debris. In doing so, they inadvertently chop nuclear material into peptides that are loaded onto MHC-II and shown to T cells as if they were foreign. Over time, this chronic exposure to nuclear self-antigens can train the immune system to react against the body itself, promoting autoimmunity. By pinpointing this pathway—from DNA damage to autophagy to nuclear antigen presentation—the study highlights new strategies to blunt age-related autoimmunity, such as enhancing DNA repair, fine-tuning autophagy, or selectively blocking the presentation of nuclear self-peptides without shutting down the immune system’s vital defenses.
Citation: Niotis, G., Arvanitaki, E.S., Theodorakis, E. et al. DNA damage in macrophages drives immune autoreactivity via nuclear antigen presentation. Nat Aging 6, 393–413 (2026). https://doi.org/10.1038/s43587-025-01053-3
Keywords: aging and autoimmunity, DNA damage, macrophages, autophagy, antinuclear antibodies