Clear Sky Science · en
Exploratory analyses of clinical outcomes from the BIIB080 phase 1b study in mild Alzheimer’s disease
Why this research matters for families
Alzheimer’s disease slowly erodes memory, independence and identity, and today’s drugs offer only modest relief. Most new treatments have focused on clearing one culprit, the amyloid protein, with limited impact on the other key hallmark of the disease: tangled clumps of a protein called tau inside brain cells. This study explored whether an experimental drug, BIIB080, that aims to turn down tau production at its genetic source might safely slow memory loss and daily decline in people with early Alzheimer’s.
A new way to dial down a troublemaking protein
Instead of targeting tau after it has already formed tangles, BIIB080 works one step earlier. It is an antisense oligonucleotide, a short strand of synthetic genetic material designed to bind the message that cells use to make tau. By attaching to this message, BIIB080 instructs cells to destroy it, reducing the overall amount of tau the brain produces, including forms thought to be especially toxic. The hope is that with less new tau being made, existing tangles may shrink and spread more slowly, easing the damage to brain circuits that underlies memory and thinking problems in Alzheimer’s disease.
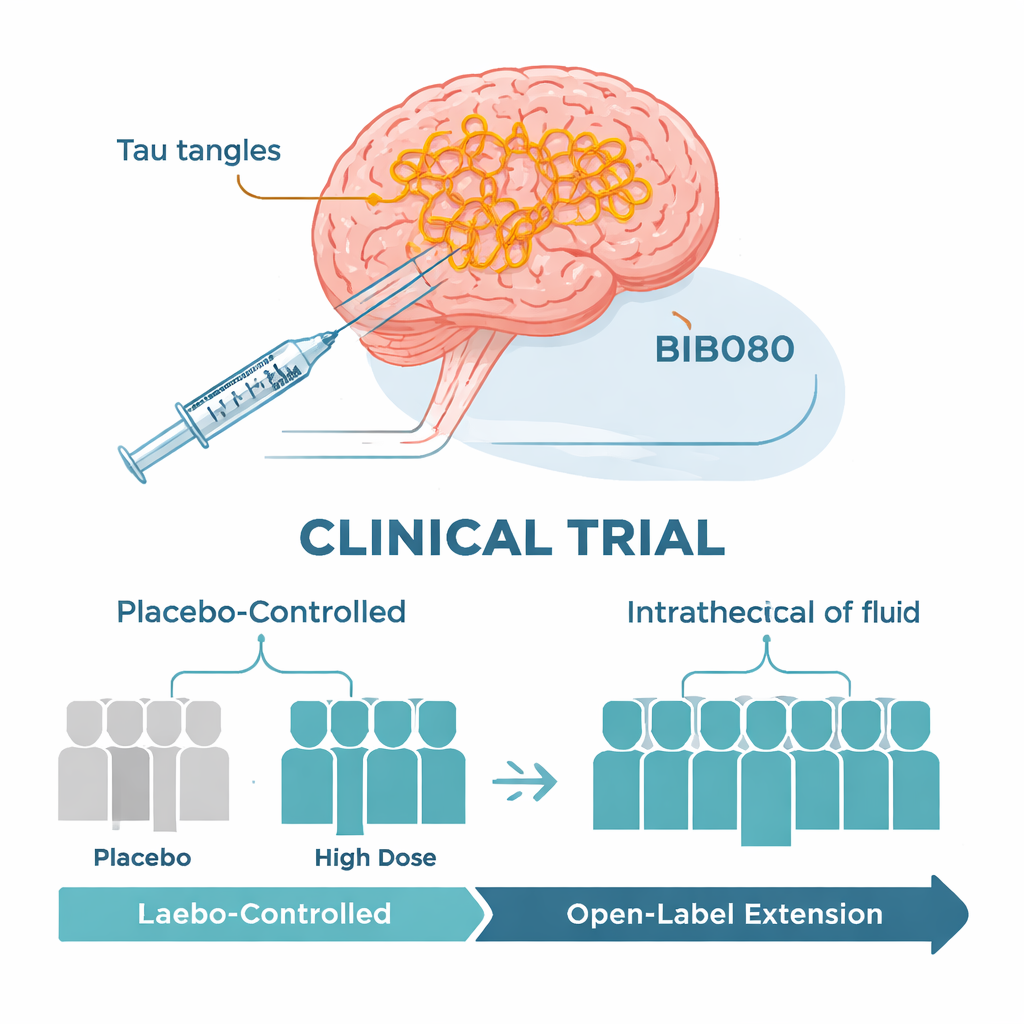
How the early trial was set up
The researchers ran an international, phase 1b clinical trial mainly designed to test safety. Just over 100 people aged 50 to 74 with mild Alzheimer’s, confirmed by spinal fluid tests, were enrolled. In the first part of the study, participants were randomly assigned to receive BIIB080 or a placebo through periodic injections into the fluid surrounding the spinal cord over three months, then followed for several more months. Different groups received gradually higher doses. A long-term extension then followed, in which everyone received what the team considered high-dose BIIB080 for almost a year. Because there was no placebo group in this second phase, the scientists compared treated patients with well-matched patients drawn from other large Alzheimer’s studies.
Signals of slower decline in thinking and daily life
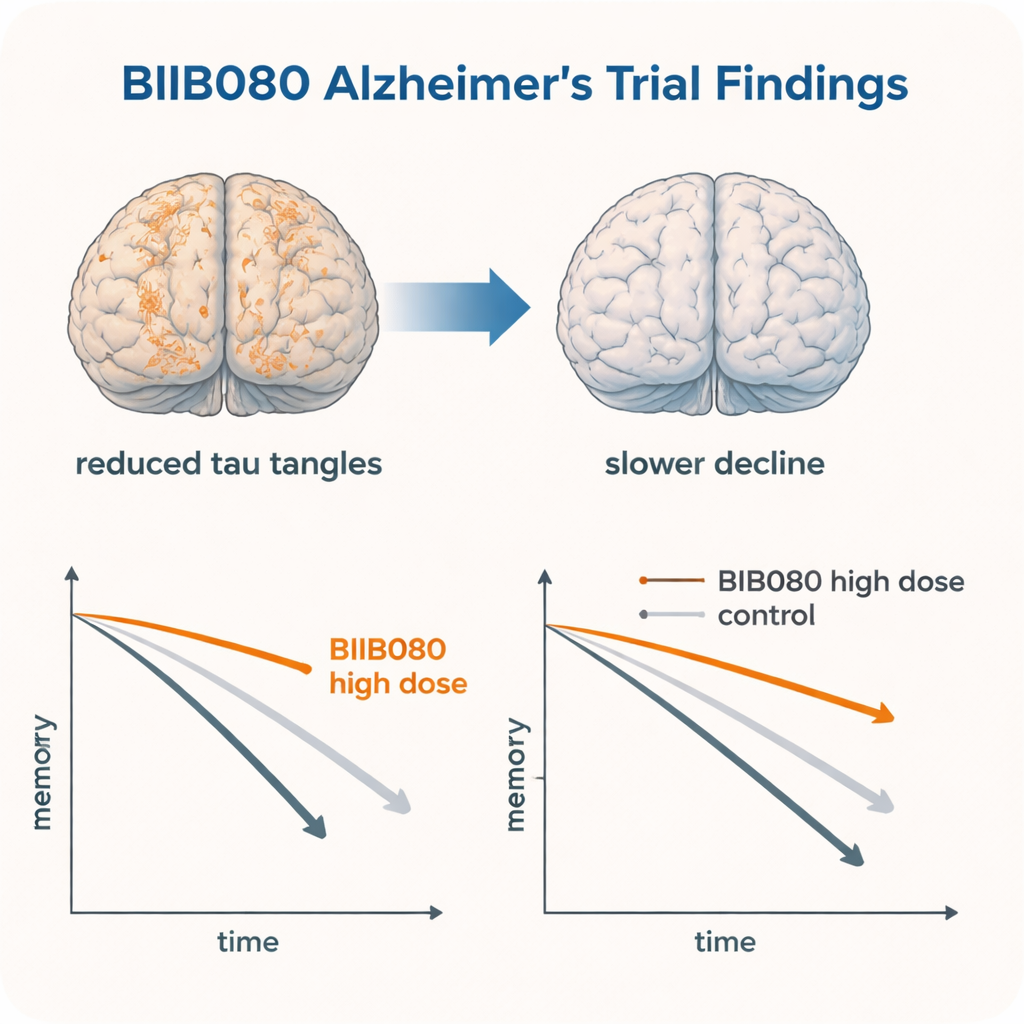
Although the study was small and not built to prove effectiveness, several patterns emerged. People on the higher doses of BIIB080 showed, on average, less worsening on standard tests of thinking (such as the Mini-Mental State Examination), memory, and ability to manage everyday tasks than those on placebo over the first months. In the long-term extension, 16 participants who stayed on high doses throughout were compared with carefully matched “external controls” from another tau-focused trial and from a major Alzheimer’s research database. Across multiple measures—global dementia rating, thinking scores and functional questionnaires—the BIIB080 group tended to decline more slowly than these comparison groups over about two years from the start of treatment.
Changes inside the brain that match the clinical trends
Earlier reports from the same trial had already shown that BIIB080 sharply lowered tau levels in spinal fluid and, strikingly, reduced the amount of tau tangles seen on specialized brain scans across many regions. This study’s exploratory clinical findings line up with those biological changes: where tau tangles fell, thinking and daily function appeared to deteriorate more slowly. In animal models that overproduce tau, similar antisense drugs had not only reduced tau deposits but also lessened seizures, nerve-cell loss and behavior problems. Together, these strands of evidence suggest that dialing down tau at its source might truly modify the course of Alzheimer’s, not just mask symptoms.
Safety, caveats and what comes next
Most side effects from BIIB080 in this trial were mild to moderate and related to the spinal injections themselves, such as headache, back pain or temporary dizziness. A few participants developed more serious problems, including worsening dementia symptoms, and stopped treatment, so careful monitoring remains essential. Importantly, the study was small, used several dose groups with uneven starting characteristics, and relied on comparison with outside data once everyone was on active drug. The authors therefore stress that the encouraging trends could still be due to chance or subtle biases.
What this could mean for future treatment
For people facing Alzheimer’s disease, these findings do not yet offer a new approved therapy, but they point toward a promising path. BIIB080 appears to safely reduce tau tangles in the human brain and is associated with slower clinical decline in this early study, especially at higher doses. Because the trial was exploratory, larger and more rigorous studies are needed to confirm whether the drug truly slows memory loss and preserves independence. Such testing is now underway in a phase 2 trial called CELIA, which will follow more patients for longer. If future results hold up, therapies that lower tau production could join or complement existing amyloid-targeting drugs, bringing a more comprehensive approach to altering the course of Alzheimer’s.
Citation: Shulman, M., Wu, S., Ziogas, N. et al. Exploratory analyses of clinical outcomes from the BIIB080 phase 1b study in mild Alzheimer’s disease. Nat Aging 6, 445–453 (2026). https://doi.org/10.1038/s43587-025-01031-9
Keywords: Alzheimer’s disease, tau protein, antisense therapy, clinical trial, neurodegeneration