Clear Sky Science · en
Senotoxins target senescence via lipid binding specificity, ion imbalance and lipidome remodeling
Why killing “zombie” cells could sharpen cancer treatment
As we age—or undergo harsh treatments like chemotherapy—some cells stop dividing but refuse to die. These “zombie” cells, known as senescent cells, can pump out inflammatory molecules that damage tissues and may help tumors come back. This study explores a surprising new way to selectively eliminate such problem cells using a venom-derived protein, potentially making cancer treatments both stronger and safer.
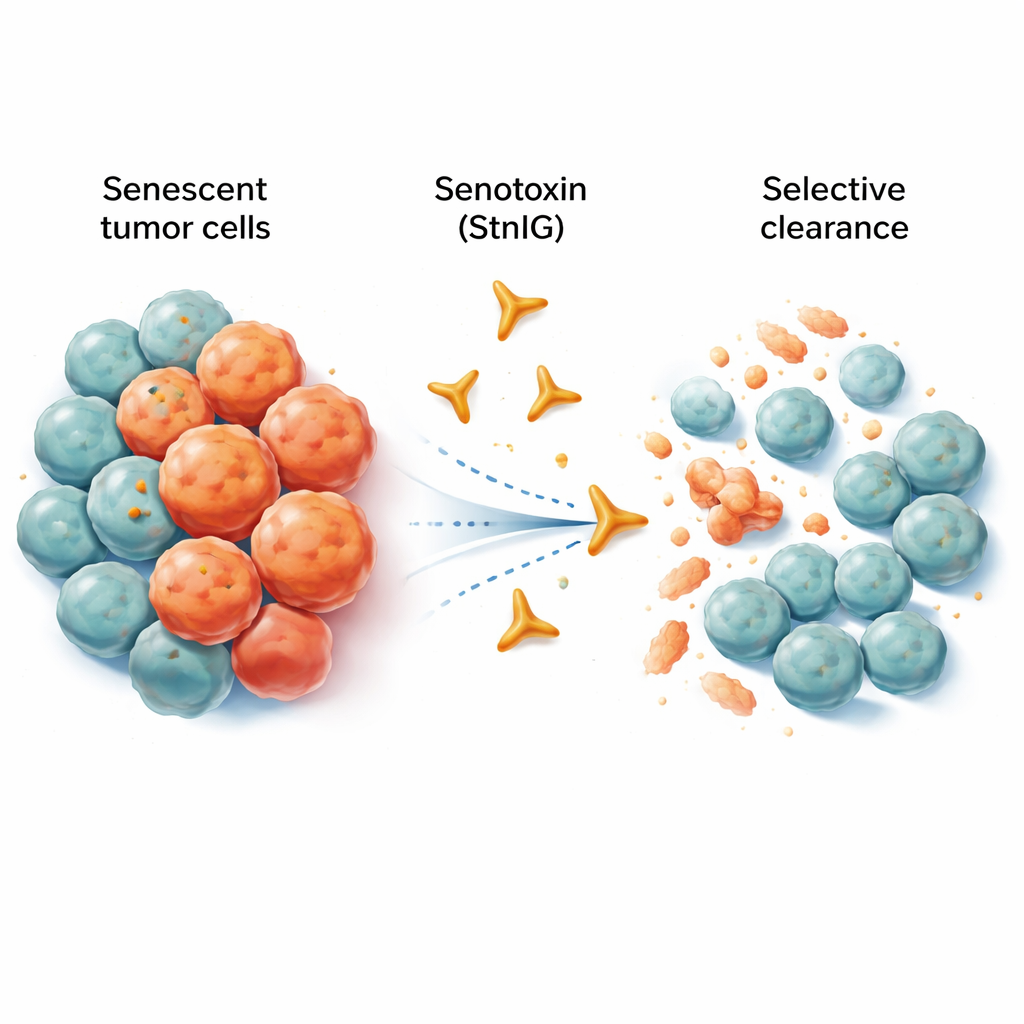
Old cells that won’t quit
Senescent cells are like retired workers who still occupy their desks and disrupt the office. They no longer multiply, but they release a cocktail of inflammatory signals that can weaken nearby tissue, promote aging, and even support tumor growth and spread. Chemotherapy, while killing many cancer cells, often leaves behind a trail of senescent cells in and around the tumor. These lingering cells can fuel chronic inflammation, side effects, and eventually, relapse. Existing experimental drugs called senolytics aim to remove senescent cells, but many of them either hit healthy cells as well, work only at very narrow doses, or are difficult for the body to absorb.
Turning sea anemone venom into a precision tool
The researchers turned to nature’s own specialists in cell-killing: animal venoms. They focused on a pore-forming protein called sticholysin I (StnI), found in a Caribbean sea anemone. This protein can punch tiny holes in cell membranes. The team discovered that StnI, and a refined version they engineered called StnIG, are strikingly better at killing senescent cancer cells than their non-senescent neighbors. Across several types of human cancer cells (melanoma, liver, lung, and nerve-derived tumors) pushed into senescence by chemotherapy drugs, StnI and especially StnIG wiped out the aged cells at much lower doses than needed to affect actively growing cells. StnIG was more selective than a leading experimental senolytic, navitoclax, suggesting that carefully tuned toxins can outperform current approaches.
How altered cell membranes become an Achilles heel
Senescent cells do not just change on the inside; their outer membrane—the fatty skin that surrounds each cell—also remodels. In healthy cells, certain lipids sit mainly on the inner side of the membrane, hidden from the outside world. In senescent cells, this balance breaks down, and these lipids become exposed. Using detailed chemical analysis and computer simulations, the team showed that StnIG recognizes and sticks to this altered lipid landscape. Its structure is especially good at latching onto specific lipids that senescent cells expose on their surface. Once attached, StnIG forms pores that allow charged atoms, or ions, to flow in and out in a highly unbalanced way.
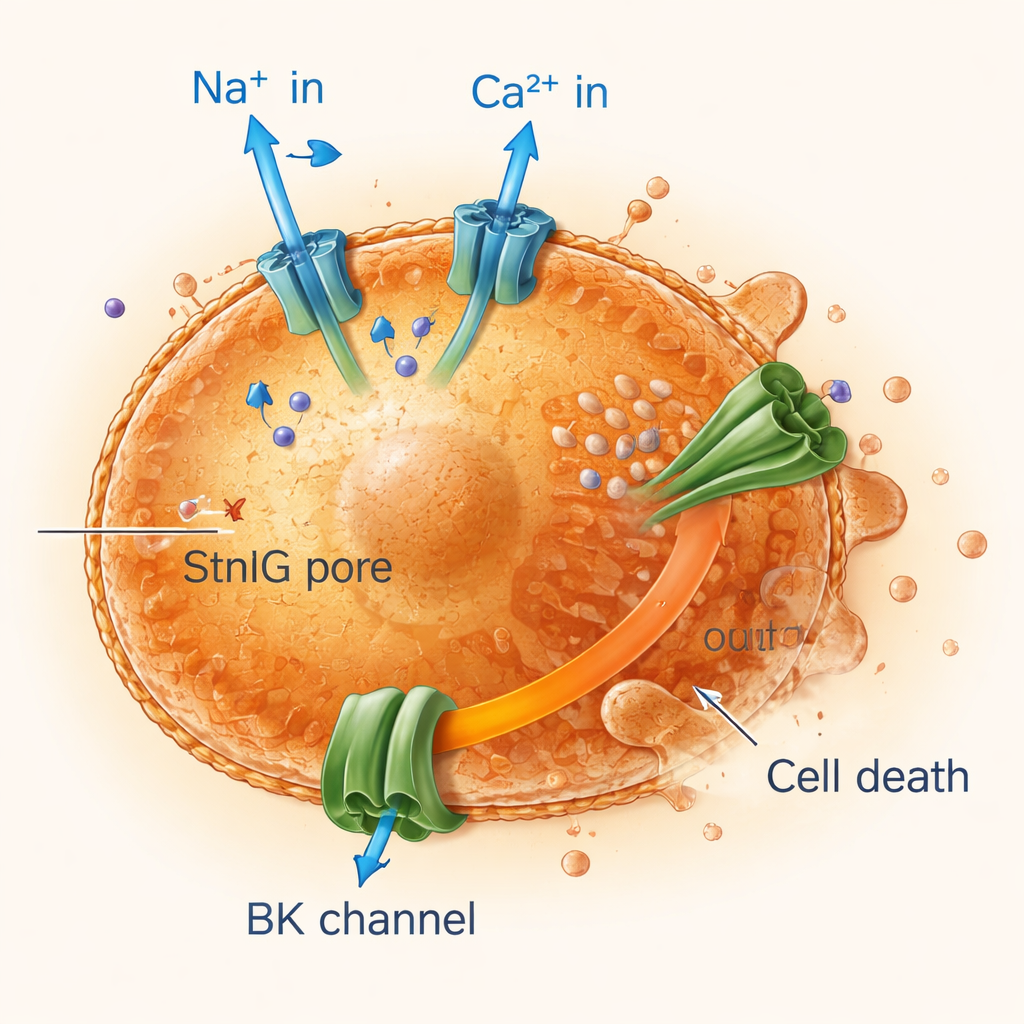
Short-circuiting aging cells with ion imbalance
By tracking electricity and ion movement across cell membranes, the researchers found that StnIG pores let sodium and calcium ions rush into senescent cells while potassium leaks out continuously. The calcium surge switches on large potassium channels, amplifying the loss of potassium, which is crucial for maintaining cell volume and survival. Senescent cells, already less flexible and more fragile than healthy ones, cannot cope with this sustained ion disruption. Their mitochondria—the cell’s power plants—lose their electrical charge, energy production dives, and the cells undergo programmed forms of death, including apoptosis and a fiery inflammatory death called pyroptosis. Blocking calcium entry or potassium channels protected senescent cells, confirming that this ion disturbance is central to the toxin’s action.
From cell dishes to living animals
The team then tested whether this strategy works in living organisms. In zebrafish embryos carrying human senescent melanoma cells, low doses of StnIG reduced the number of these cells as effectively as navitoclax, without obvious toxicity. In mouse models of melanoma and liver cancer, chemotherapy with a cell-cycle drug (palbociclib) slowed tumor growth by driving many cancer cells into senescence. When the researchers added StnIG intermittently, tumors shrank or went into remission more effectively than with chemotherapy alone, and comparable to the chemotherapy–navitoclax combination. Importantly, StnIG-treated mice showed limited side effects and no clear damage in major organs, though blood potassium rose in animals with senescent tumors—likely reflecting the massive release of potassium as tumor cells were destroyed.
What this means for future cancer care
This work introduces “senotoxins”: engineered venom-inspired proteins that exploit the unique membrane and ion-handling quirks of senescent cells. By homing in on altered lipids and forcing fatal ion imbalances, StnIG can selectively clear harmful senescent cells while sparing most healthy tissue. Used alongside chemotherapy, such senotoxins might one day help sweep away the zombie cells that fuel relapse and chronic inflammation, making cancer treatments more durable and potentially easing some long-term side effects.
Citation: Moral-Sanz, J., Fernández-Carrasco, I., Ramponi, V. et al. Senotoxins target senescence via lipid binding specificity, ion imbalance and lipidome remodeling. Nat Aging 6, 349–367 (2026). https://doi.org/10.1038/s43587-025-01030-w
Keywords: cellular senescence, senolytics, cancer therapy, venom-derived drugs, ion channels