Clear Sky Science · en
Aperture and roughness govern iron oxide passivation in olivine fractures during carbon mineralization
Why tiny cracks in rocks matter for climate solutions
Turning carbon dioxide into stone deep underground is one of the most permanent ways to keep this greenhouse gas out of the atmosphere. This study looks at what happens inside the tiny cracks of a common volcanic mineral called olivine when it reacts with pressurized CO2. By zooming in on how rough or smooth those crack surfaces are, and how wide the cracks are, the researchers uncover hidden details that could make or break the effectiveness of this natural carbon-locking process.
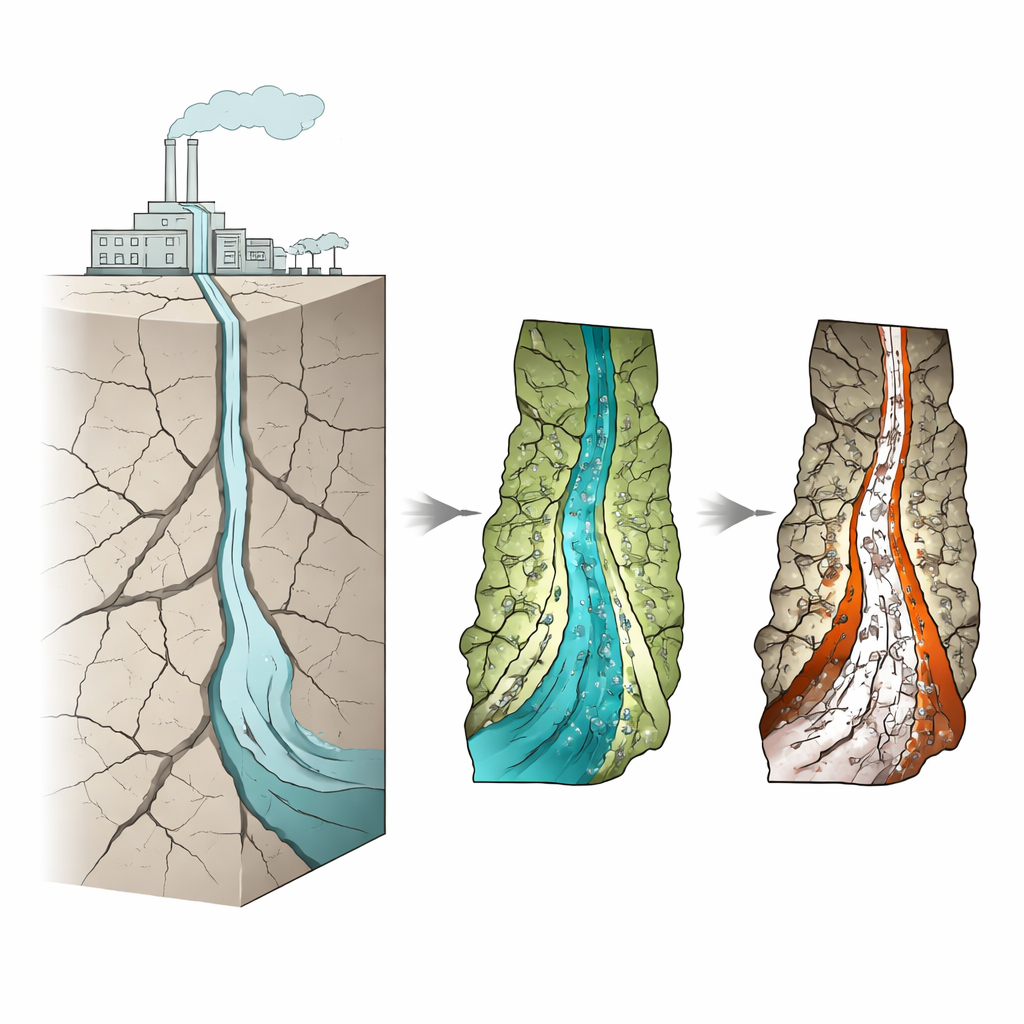
Locking away carbon in volcanic rocks
Engineers are exploring ways to inject captured CO2 into deep rock formations where it can react with minerals and form solid carbonates, effectively turning gas into rock. Basalts and related rocks rich in olivine are especially promising because they contain magnesium and iron, elements that readily form stable carbonate minerals. But these rocks are not like open caves; most fluid movement and reactions happen in narrow fractures. In these dead-end cracks, CO2-rich fluids linger, providing ideal conditions for mineral reactions—if the crack surfaces stay reactive.
Building controlled cracks to watch reactions
To understand how crack shape controls carbon storage, the team prepared artificial “fractures” in slices of forsteritic olivine. Each fracture had one rough side and one smooth side, and the gap between the two sides (the aperture) was carefully set to be either relatively small or larger, mimicking tight and wider natural cracks. These rock sandwiches were then exposed for two weeks to hot, high-pressure, CO2-rich water under conditions similar to those planned for industrial carbon storage. Afterward, the researchers used microscopes, Raman spectroscopy (a light-based tool for identifying minerals), surface profiling, and chemical analyses of the fluids to map which new minerals formed where, and how much of the original olivine was eaten away.

Rough surfaces help and hinder at the same time
The team found a striking pattern in the smaller fractures. Both rough and smooth areas grew a magnesium carbonate mineral called magnesite, which is the desired product for locking up CO2. However, rough surfaces also strongly encouraged the formation of iron oxide coatings, while smooth areas mostly avoided them. These iron-rich layers act like a protective skin: they cover the olivine and slow further reaction, a process known as passivation. Surface measurements showed that smooth regions in small fractures lost more material overall, meaning they kept dissolving and reacting, whereas rough regions lost less, consistent with their surfaces becoming shielded. In other words, extra roughness increases the area that can react but also creates microenvironments where passivating iron oxides build up and choke off the reaction over time.
Wider cracks change the balance
When the fracture aperture was larger, the influence of roughness faded. In these wider cracks, iron oxides appeared on both rough and smooth sides, and carbonate crystals tended to be bigger and more abundant. The larger opening allowed more rapid exchange between the CO2-rich bulk fluid and the rock surface, supplying more reactive ingredients and raising the overall concentration of dissolved ions. This environment favored both continued carbonate growth and widespread iron oxide formation. As a result, wider fractures initially enhanced reaction but also promoted more uniform passivation across the surfaces. Computer models that included realistic surface roughness and coatings reproduced these trends, showing that simply increasing surface area does not guarantee faster or more complete carbon mineralization if passivating layers form.
Designing better underground carbon storage
To a non-specialist, the core message is that not all rock fractures are equally good at turning CO2 into stone. Tiny differences in how rough the crack walls are and how wide the cracks open can determine whether carbon-absorbing reactions keep going or stall out behind a film of iron oxide. In tight, rough fractures, carbonates can form but may be limited by quick passivation. In wider fractures, reactions are more vigorous but can still slow as coatings spread. The study shows that future carbon storage projects need to account for these microscopic details in fracture networks when predicting how much CO2 will truly become locked away as minerals over decades to centuries.
Citation: Yang, Y., Boampong, L.O., Nisbet, H. et al. Aperture and roughness govern iron oxide passivation in olivine fractures during carbon mineralization. Commun Earth Environ 7, 210 (2026). https://doi.org/10.1038/s43247-026-03235-2
Keywords: carbon mineralization, geologic carbon storage, olivine, rock fractures, iron oxide passivation