Clear Sky Science · en
Anthropogenic emissions of volatile Cd detected in western tropical North Atlantic surface seawater
Invisible metal in a changing ocean
Cadmium is best known as a toxic heavy metal, but in the open ocean it behaves more like a rare nutrient, quietly cycling between water, tiny drifting plants, and the atmosphere. This study investigates why surface waters in part of the tropical North Atlantic contain surprisingly little cadmium with an unusual chemical fingerprint, and shows that human-made air pollution – not just natural ocean processes – is already reshaping the chemistry of seemingly pristine seas.
Why cadmium matters in seawater
Although cadmium is present in seawater at vanishingly low levels, its distribution closely tracks that of phosphate, a key plant nutrient. Near the surface, phytoplankton strip cadmium from the water as they grow; when they die and sink, cadmium is released again at depth as the organic matter breaks down. Because lighter and heavier forms (isotopes) of cadmium are taken up and recycled in slightly different ways, scientists can use these isotopes to trace how cadmium moves through the ocean and to distinguish internal recycling from outside inputs such as rivers or airborne dust.
Tracing water masses and deep ocean mixing
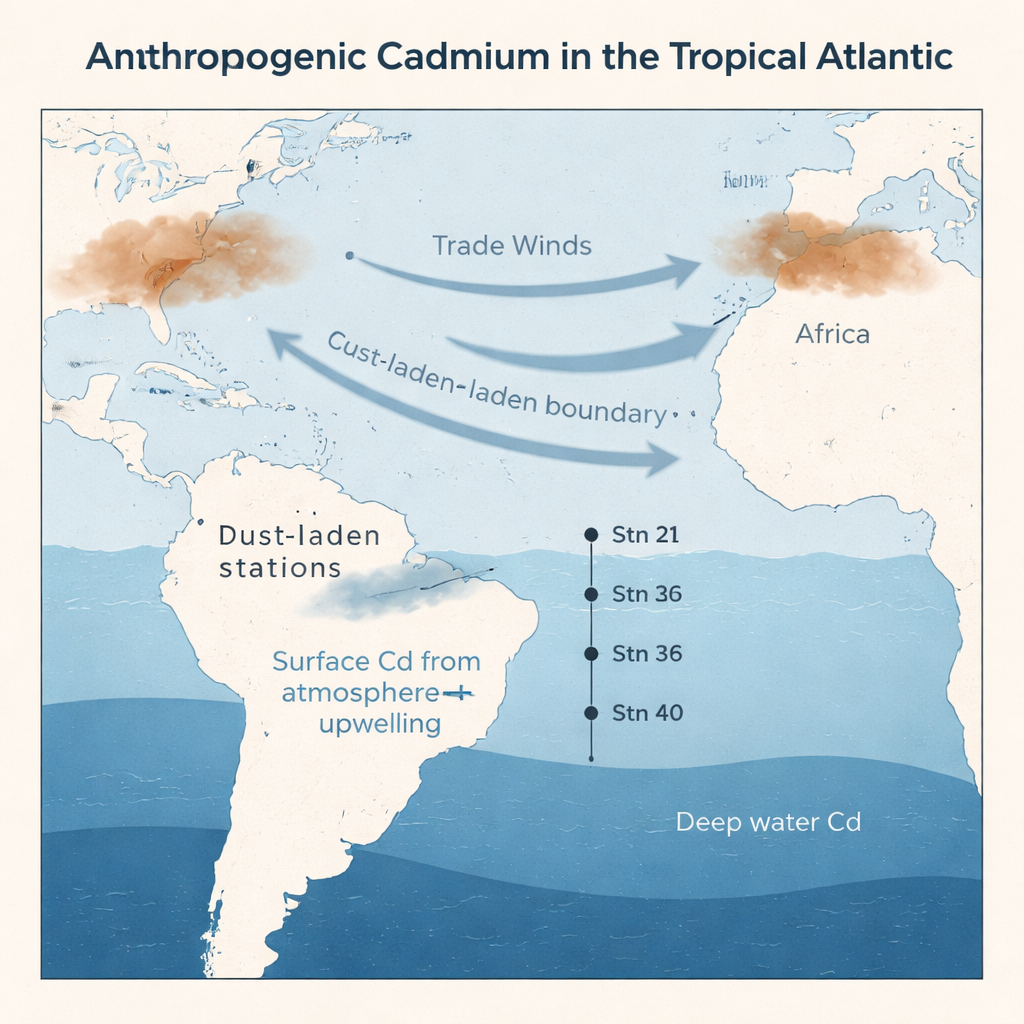
The researchers collected seawater from four stations along the western tropical North Atlantic, between Bermuda and Brazil, measuring cadmium concentrations and isotopes from the surface down to nearly 5,000 meters. Using a detailed water-mass mixing model, they showed that in deep waters, cadmium patterns are largely controlled by the blending of water masses originating in the North and South Atlantic and the Southern Ocean. In these depths, the model could accurately reproduce both how much cadmium was present and its isotope composition, indicating that simple physical circulation dominates cadmium behavior far below the surface.
Life and decay in the mid-depths
At intermediate depths, where oxygen levels are lower and sinking organic matter is decomposed, biology becomes more important. The team compared observed cadmium levels with those expected from water-mass mixing alone and found substantial "extra" cadmium in oxygen-poor layers. This excess, sometimes accounting for up to 80 percent of dissolved cadmium there, is best explained by remineralization—the release of cadmium from decaying biological particles. Yet the isotope signatures in these layers remained very similar to those predicted from mixing, implying that cadmium in the particles and in the surrounding water had nearly the same isotopic makeup. As a result, remineralization changes how much cadmium is present but only subtly alters its isotopic fingerprint at these depths.
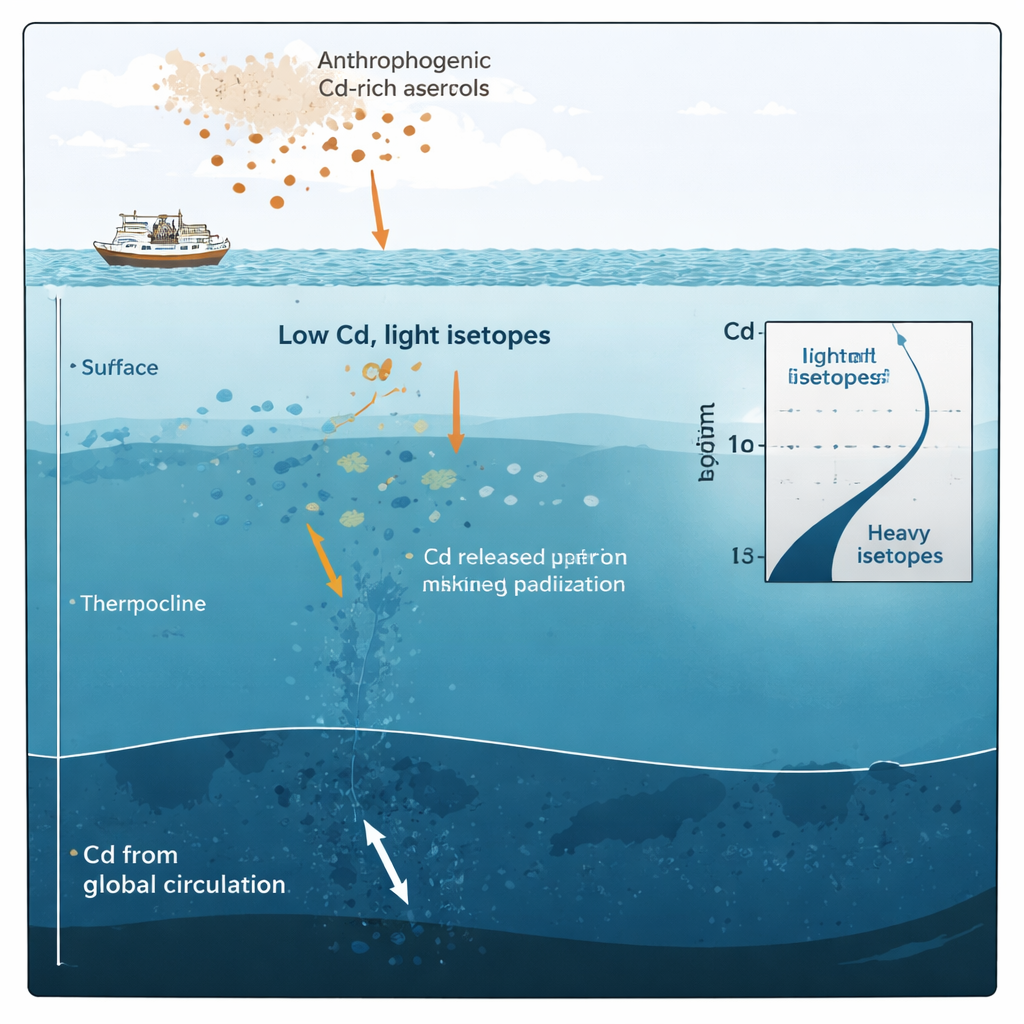
A surprising signal at the sunlit surface
The biggest surprise came from the upper 100 meters. Here, cadmium concentrations were extremely low, as expected in nutrient-poor (oligotrophic) waters where phytoplankton have already stripped out much of the available metal. However, the cadmium left behind was isotopically lighter than in deeper water, the opposite of what would be expected if biology were the only influence. Careful checks ruled out measurement problems, and river input from the nearby Amazon plume was shown to be minimal. This pointed toward a different external source: atmospheric deposition of cadmium-bearing particles, especially those enriched by human activities such as smelting, fossil fuel burning, and waste incineration.
Air pollution reaching remote seas
To test this idea, the authors built a simple "box" model of the surface ocean that balanced inputs of cadmium from upwelling deep water and falling aerosols against losses to sinking particles and downwelling. When they fed in realistic values for wind-blown dust, cadmium solubility, and biological uptake, the model could reproduce the observed light isotope signatures only if a substantial share of surface cadmium came from cadmium-rich, human-influenced aerosols with light isotopic compositions. Their calculations suggest that during the study period, at least about one-fifth and possibly nearly half of the dissolved cadmium in these surface waters originated from anthropogenic emissions carried by the wind.
What this means for people and the planet
For non-specialists, the key message is that industrial pollution is leaving a detectable fingerprint in the chemistry of even the most remote parts of the open ocean. In the western tropical North Atlantic, the cadmium pattern in surface seawater no longer reflects only natural upwelling and biological recycling; it now also carries the mark of smokestacks and burning on land. While the absolute cadmium levels remain very low, this study shows that volatile metals emitted into the air can travel far and alter the subtle balance of trace elements that support marine life. Such findings underscore how tightly connected our atmosphere and oceans are, and how human activities on land can reshape the chemistry of the global sea.
Citation: Xu, H., Rehkämper, M., Huang, Y. et al. Anthropogenic emissions of volatile Cd detected in western tropical North Atlantic surface seawater. Commun Earth Environ 7, 181 (2026). https://doi.org/10.1038/s43247-026-03211-w
Keywords: cadmium in oceans, marine aerosols, trace metal pollution, Atlantic Ocean chemistry, anthropogenic emissions