Clear Sky Science · en
Oxidative uptake of Ce by oceanic ferromanganese crusts and implications for paleoredox estimates using Ce isotopes
Reading Earth’s Ancient Breath
Earth’s deep oceans slowly record the rise and fall of oxygen through time, but decoding that record is tricky. One promising clue comes from cerium, a metallic element that behaves differently depending on how much oxygen is around. This study explores exactly how cerium sticks to metal-rich crusts on the seafloor and how that process imprints subtle isotopic “fingerprints” that scientists can use to reconstruct the redox (oxygen) history of Earth’s oceans.
Metal Coatings on the Ocean Floor
The seafloor in many parts of the Pacific, Atlantic, and Indian oceans is dotted with slow-growing ferromanganese crusts—thin, metal-rich coatings that build up on hard rock over millions of years. These crusts act like chemical flypaper, capturing trace elements from seawater as it flows past. Cerium is unusual among these elements because it can exist in two oxidation states, a dissolved form (Ce(III)) and an oxidized, particle-bound form (Ce(IV)). The balance between these forms depends on how much oxygen is present, so the way cerium is stored in these crusts has become a standard tool for inferring how oxygenated ancient oceans were.
How Cerium Really Attaches
Earlier lab experiments using synthetic manganese minerals suggested that when cerium is oxidized on their surfaces, it precipitates as a separate solid called ceric hydroxide, Ce(OH)4. However, this model never fully matched signals from natural seafloor deposits. In this work, the authors used highly sensitive X-ray spectroscopy at a synchrotron to probe the structure of cerium atoms directly in natural ferromanganese crusts from all three major oceans. They show that all cerium present is in the oxidized Ce(IV) state, but crucially, it is not forming grains of Ce(OH)4 or CeO2. Instead, individual cerium atoms are bonded directly to the manganese mineral vernadite (δ-MnO2) at specific atomic positions.
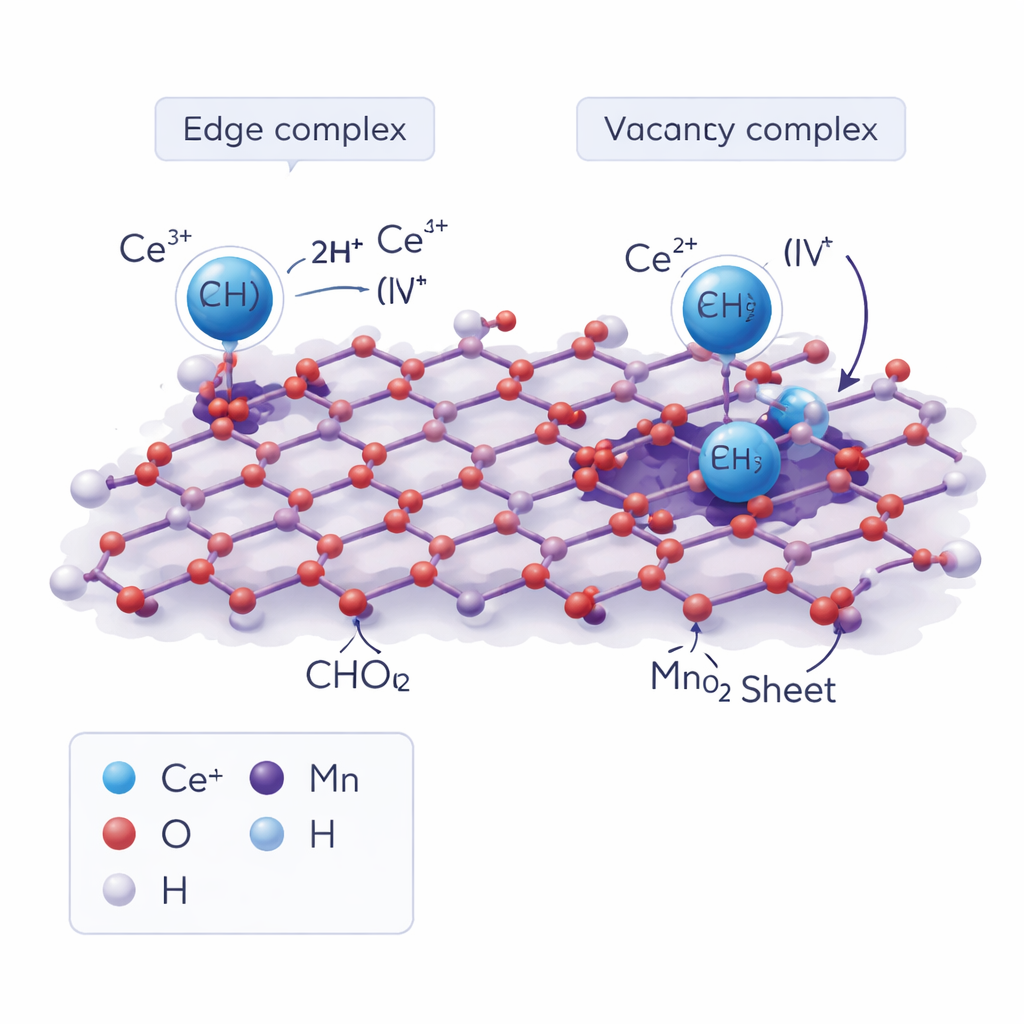
Atomic Anchors on Mineral Surfaces
By combining the X-ray data with quantum mechanical calculations, the team identified two main “anchor points” for cerium on vernadite. At the edges of the mineral layers, cerium forms so‑called double-edge-sharing complexes, where a single Ce atom shares oxygen atoms with neighboring manganese octahedra. Cerium can also occupy positions associated with vacant sites inside the mineral layers, sitting partially in a missing manganese site. When Ce(III) from seawater first binds to the edge, nearby water molecules lose protons—a process called hydrolysis—which in turn promotes the transfer of an electron from cerium to manganese. This step flips cerium from Ce(III) to Ce(IV) and stabilizes it on the surface without forming a separate mineral phase.
Isotope Fingerprints as Oxygen Proxies
Cerium has several stable isotopes, including light 136Ce and heavy 140Ce. The exact way cerium bonds to oxygen atoms changes the stiffness of those bonds, and that, in turn, influences how strongly different isotopes are favored. The authors calculated how cerium isotopes partition between dissolved Ce(III) in seawater and the Ce(IV) complexes on vernadite. They found that when cerium is oxidized and locked into these surface complexes, the product can become significantly enriched in heavy 140Ce relative to 136Ce—by about 1.2–1.3 parts per thousand at room temperature. This is a much larger effect than the small net fractionation seen for the commonly measured 142Ce/140Ce pair, where competing nuclear effects largely cancel each other out.
A Sharper Tool for Reading Earth’s Past
These findings show that in real oceanic crusts, cerium is taken up mainly as isolated Ce(IV) surface complexes, not as bulk Ce(OH)4. That revision matters because it changes how scientists interpret the isotopic signals frozen into seafloor minerals. The work suggests that the 136Ce/140Ce ratio, though more challenging to measure, could serve as a much more sensitive tracer of past oxygen conditions than the traditional 142Ce/140Ce ratio. In practical terms, understanding exactly where and how cerium atoms sit in these deep-sea coatings brings researchers closer to reading Earth’s ancient “oxygen logs” with greater precision, improving reconstructions of how the planet’s surface environment evolved through time.
Citation: Manceau, A., Liao, J., Li, Y. et al. Oxidative uptake of Ce by oceanic ferromanganese crusts and implications for paleoredox estimates using Ce isotopes. Commun Earth Environ 7, 172 (2026). https://doi.org/10.1038/s43247-026-03196-6
Keywords: cerium isotopes, ferromanganese crusts, ocean redox, vernadite, paleoceanography