Clear Sky Science · en
Lowering the Mo limit for nitrogen fixation by Mo-nitrogenase
Why this ancient chemistry matters today
All life on Earth depends on nitrogen, an ingredient of DNA and proteins. Yet most organisms cannot use the nitrogen gas that makes up most of our air; they rely instead on specialized microbes that “fix” nitrogen into usable forms. For decades, scientists have argued that Earth’s early oceans lacked enough of a key metal, molybdenum, to support this process, potentially slowing the rise of life. This study tests that idea in a modern lake that mimics the chemistry of our planet billions of years ago.
A modern lake as a time machine
Deming Lake in northern Minnesota is a small, permanently layered lake whose waters resemble the ancient ocean in several ways. The surface is oxygen-rich and dominated by photosynthetic cyanobacteria, while deeper layers are dark, low in oxygen, and rich in dissolved iron—conditions scientists call ferruginous. Measurements show that both molybdenum and sulfate, two dissolved components thought to control nitrogen-fixing activity, are extremely scarce here: molybdenum is usually below one-billionth of a mole per liter, and sulfate is less than one-millionth. This makes Deming Lake an ideal natural laboratory for asking whether nitrogen fixation can thrive when molybdenum is vanishingly rare. 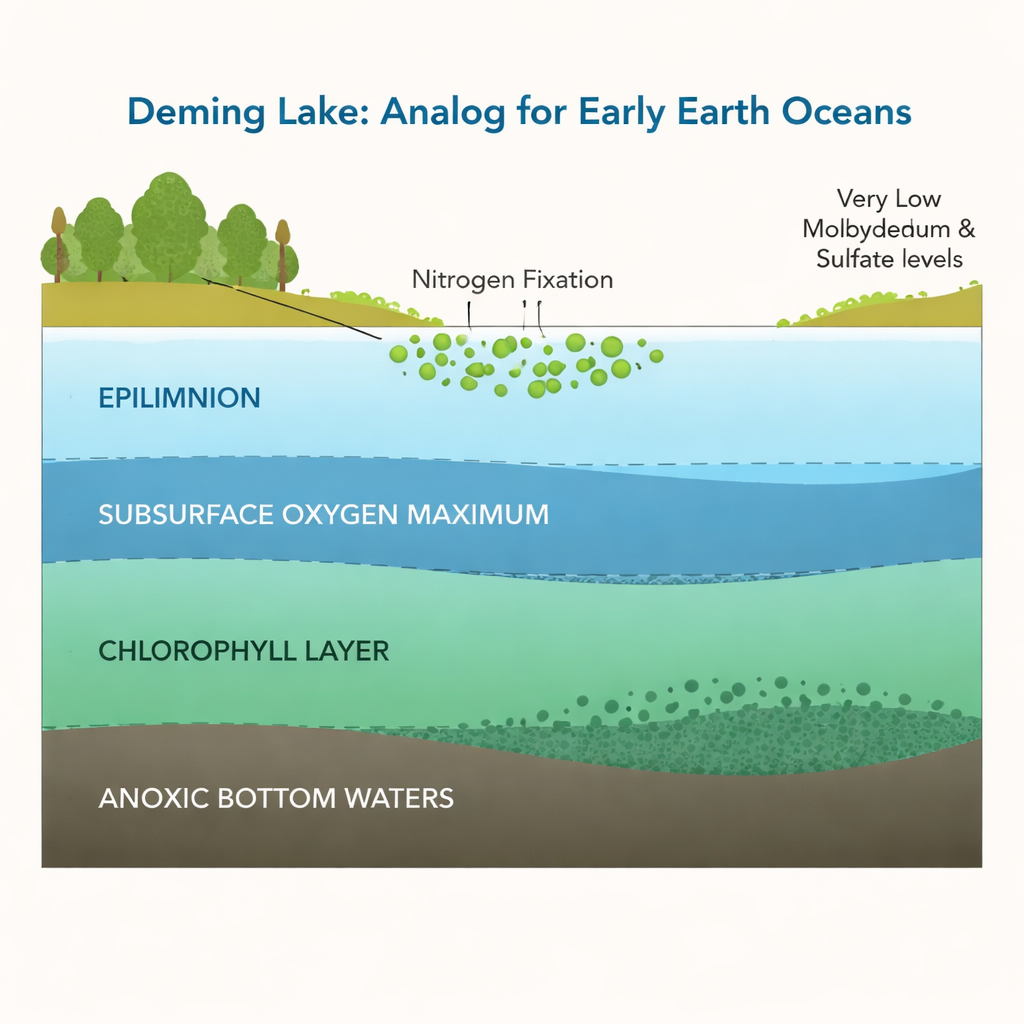
Tracking invisible nitrogen traffic
To see if microbes were still fixing nitrogen under these lean conditions, the researchers combined several lines of evidence. First, they measured how much nitrogen gas disappeared from the water relative to argon, an inert gas, and found signatures of net nitrogen loss where cyanobacteria were most active. They then used an isotope tracer technique, adding a heavy form of nitrogen gas (¹⁵N₂) to bottles of lake water suspended back in the lake. Over 24 hours, the heavy nitrogen built up in particulate matter, revealing that microbes in the sunlit surface and just below it were fixing tens of nanomoles of nitrogen per liter per day—substantial rates for such a nutrient-poor system.
Starving the system of molybdenum—without slowing it down
If molybdenum were truly limiting, adding a bit more should boost nitrogen fixation. The team therefore enriched some bottle incubations with extra molybdenum, bringing concentrations to levels still low by ocean standards but far above the lake’s background. Nitrogen fixation rates, however, did not increase in a statistically meaningful way. At the same depths where fixation was strongest, the process ran just as well without the supplement. This shows that, in Deming Lake, nitrogen-fixing microbes are not bottlenecked by the supply of molybdenum, even when it is more than one hundred times lower than in today’s oceans.
Which molecular tools are doing the work?
To identify the machinery behind this robust nitrogen fixation, the authors sequenced DNA and RNA from microbes at various depths. They focused on the genes that build nitrogenase, the enzyme complex that turns nitrogen gas into biologically useful forms, and on genes that transport molybdenum into cells. Every nitrogenase gene set they detected encoded the classic molybdenum–iron version of the enzyme; the alternative versions that use only iron or vanadium were absent. A cyanobacterium related to Synechococcus stood out as especially abundant and transcriptionally active, and it carried both molybdenum-based nitrogenase genes and high-affinity transport systems that can scavenge trace amounts of molybdenum. The lake’s very low sulfate levels likely further ease competition between sulfate and molybdate at these transport sites, allowing microbes to harvest molybdenum efficiently. 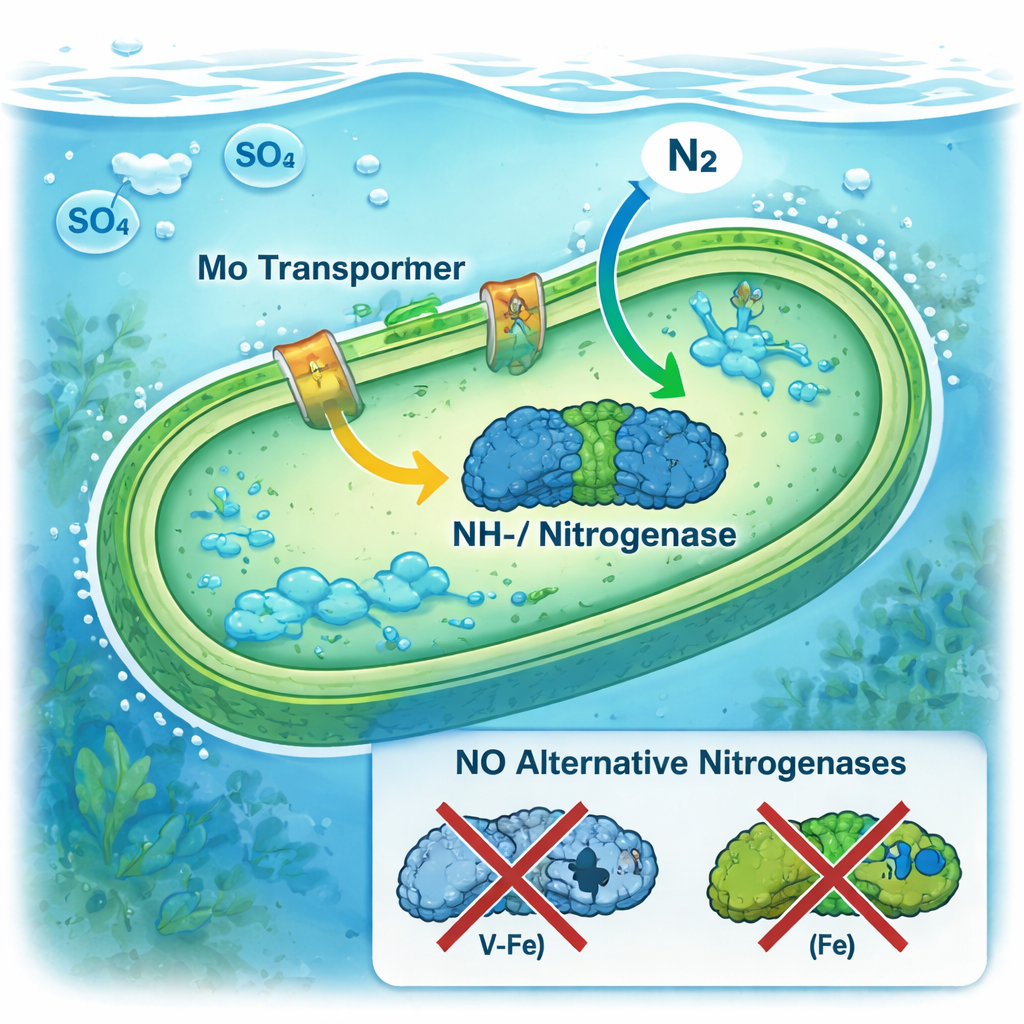
Rethinking early Earth’s nitrogen engine
The study’s central message is that molybdenum-based nitrogen fixation can flourish even when molybdenum concentrations fall below one nanomolar, provided sulfate is also scarce and microbes have efficient uptake systems. That finding undercuts the long-standing assumption that early oceans were too molybdenum-poor to support this enzyme, forcing life to rely on alternative metal versions. Instead, it supports geological and evolutionary clues suggesting that the molybdenum-based system was both ancient and dominant. As sulfate levels rose later in Earth’s history, they may have created the very molybdenum stress that favored the evolution of vanadium- and iron-only nitrogenases. In simple terms, this work shows that early life may have gotten by with less molybdenum than we thought, reshaping our picture of how Earth’s nitrogen cycle—and the biosphere it sustains—first took hold.
Citation: Stevenson, Z., Schultz, D.L., Chamberlain, M. et al. Lowering the Mo limit for nitrogen fixation by Mo-nitrogenase. Commun Earth Environ 7, 169 (2026). https://doi.org/10.1038/s43247-026-03193-9
Keywords: nitrogen fixation, molybdenum, cyanobacteria, early Earth oceans, lake ecology