Clear Sky Science · en
Evidence for diverse anaerobic metabolisms in 3.7-billion-year-old marine detrital sediments
Ancient Clues to Earth’s Earliest Life
Deep in Greenland’s bedrock lies a time capsule from more than 3.7 billion years ago—rocks that hold some of the oldest known traces of life on Earth. This study asks a deceptively simple question with big implications: were those earliest seafloor communities already using a range of “breathing” strategies without oxygen, much like microbes do in today’s muds and sediments? The answer helps us understand how quickly life diversified, how it shaped the young planet’s oceans and atmosphere, and what kinds of biosignatures we might look for on other worlds.
A Quiet Seafloor, Rich in Life’s Debris
The rocks examined come from the Isua Supracrustal Belt in southwest Greenland, one of the oldest surviving pieces of Earth’s crust. Back then, this area was a quiet deep-marine basin where fine particles slowly drifted down from the surface ocean, building up thin, gently layered sediments. Periodically, underwater avalanches of coarser material, called turbidites, swept in from nearby high ground. These sediments are packed with ancient carbon-rich particles, now transformed into graphite, that earlier work has linked to photosynthetic microbes in the surface ocean. In other words, the seafloor here was continually dusted with the remains of an early, surprisingly productive biosphere.
Breathing Without Oxygen: Iron and Methane in Play
Today, much of the organic matter that reaches the seafloor is broken down by microbes that “breathe” oxygen. But once oxygen runs out, microbes switch to other chemical partners, such as nitrate, iron, or sulfate. The authors measured the ratios of carbon isotopes in the Isua sediments to see how that ancient organic matter was processed. They found unusually light carbon signatures in some layers—lighter than expected from simple photosynthesis alone. This pattern fits with organic matter and methane being broken down in the seafloor by anaerobic microbes. In layers where iron was especially abundant, the lightest carbon values lined up with high iron-to-carbon ratios, pointing to microbes that used oxidized iron as their main “oxygen substitute,” reducing it as they consumed organic compounds.
Sulfur Traces of Invisible Microbial Work
Iron was not the only game in town. The team also studied tiny grains of sulfide minerals, such as pyrrhotite and pyrite, that occur mainly in the fine, carbon-rich pelagic layers, not in the coarser turbidites. Petrographic textures—like thin sulfide bands hugging bedding planes and concentric pyrite nodules—suggest that sulfur-bearing minerals formed early, within the sediments, as reactive fluids moved through them. Using precise sulfur-isotope measurements on individual grains, the researchers showed that most of the sulfur carried a distinct atmospheric imprint, having started as elemental sulfur and sulfate produced when sunlight broke apart sulfur dioxide in the anoxic early air. Isotope patterns indicate that microbes likely reduced this elemental sulfur, and at times also reduced a small, locally replenished pool of seawater sulfate, to make hydrogen sulfide that then reacted with iron to form sulfide minerals.
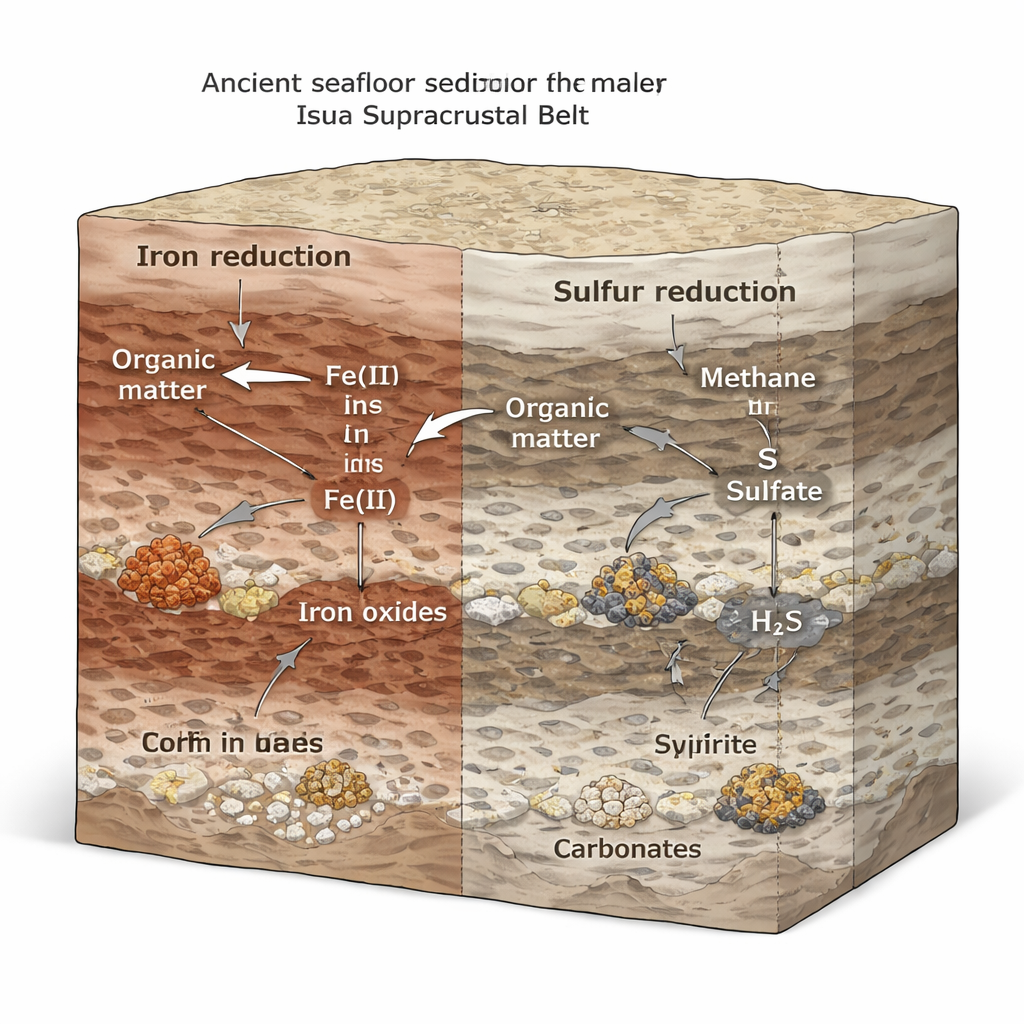
A Layered Landscape of Hidden Microbial Niches
By combining carbon and sulfur isotopes with iron concentration profiles and mineral textures, the study reconstructs a dynamic chemical landscape beneath the ancient seafloor. Iron-rich layers and organic-rich layers frequently sat close together, creating micro-environments where different metabolic strategies could flourish side by side. Where ferric iron was plentiful, iron-reducing microbes appear to have dominated. In zones with slower sediment buildup and greater fluid exchange, sulfur-based respiration and near-complete reduction of scarce sulfate became more important. Methane, produced deeper down by fermenters and methanogens, likely seeped upward and was consumed by other microbes using iron or sulfur as oxidants, further lightening the carbon isotopic signatures.
What This Means for the Story of Early Life
To a non-specialist, the key message is that by 3.7 billion years ago, Earth’s seafloor was not a simple, single-pathway ecosystem. Instead, it already hosted a web of interacting microbial communities that could tap into iron, elemental sulfur, and sulfate to survive without oxygen, while recycling organic matter and methane. These findings push the emergence of complex, diversified microbial metabolism to very early in Earth’s history. That, in turn, suggests that once life gained a foothold, it rapidly evolved a toolkit for exploiting a wide range of chemical energy sources—an encouraging thought for the hunt for life in ancient rocks on Earth and on other planets.
Citation: Boyd, A.J., Harding, M.A.R., Bell, E.A. et al. Evidence for diverse anaerobic metabolisms in 3.7-billion-year-old marine detrital sediments. Commun Earth Environ 7, 166 (2026). https://doi.org/10.1038/s43247-026-03188-6
Keywords: early Earth life, ancient seafloor microbes, anaerobic respiration, iron and sulfur cycles, Isua Supracrustal Belt